Clear Sky Science · en
3D dynamic structure of a Pt nanoparticle on SrTiO3 (001) during in-situ heating atomic-resolution ADF STEM imaging
Why tiny metal particles matter
Catalysts are the quiet workhorses of modern life, helping to clean car exhaust, make fertilizers, and split water into hydrogen fuel. Many of the best catalysts rely on tiny clusters of precious metals, such as platinum, sitting on the surface of another material. Scientists know that only a small fraction of the atoms in these nanoparticles actually do most of the chemical work, but pinning down exactly where those “hot spots” are—and how they shift while the catalyst is operating—has been extremely difficult. This study shows, atom by atom, how a single platinum nanoparticle changes in three dimensions at elevated temperature, and links those details directly to where catalytic activity is likely to occur.

Seeing atoms in three dimensions
The researchers focused on platinum nanoparticles just under two billionths of a meter wide, grown on a carefully prepared, atomically flat crystal of strontium titanate (SrTiO3). Using an advanced form of electron microscopy called annular dark-field scanning transmission electron microscopy (ADF-STEM), they recorded images in which brighter spots correspond to heavier atoms like platinum. Because these images are so sensitive to atomic number, the intensity of each bright spot can be used to estimate how many platinum atoms sit on top of a given column of substrate atoms. By combining a single high-quality image with sophisticated statistical analysis, the team was able to reconstruct the full three-dimensional arrangement of 263 platinum atomic sites in one nanoparticle, including how it attaches to the oxide surface.
Tracking moving atoms in a hot environment
Real catalysts operate at high temperatures and often in reactive gases, where atoms do not sit still. To mimic such conditions without damaging the sample, the team heated the platinum–oxide system to about 210 °C in a very clean, low-pressure environment inside the microscope. They rapidly collected dozens of images of the same nanoparticle and averaged them to boost signal while preserving signs of motion. Subtle changes in brightness at certain atomic positions revealed that some platinum atoms hop between nearby sites during the experiment. Instead of treating this as noise, the scientists interpreted those in-between brightness levels as “partial occupation,” meaning that a given site is occupied only part of the time. This allowed them to build not just a static 3D model, but a picture of where atoms are most mobile on the nanoparticle’s surface.
Rough surfaces and special atomic neighborhoods
The reconstructed nanoparticle looks like a tiny metallic dome sitting on the oxide crystal. Many atoms in the interior have 10 to 12 neighboring platinum atoms, similar to bulk metal, but nearly half of the atoms lie at or near the surface and have fewer neighbors. The researchers quantified this by counting the “coordination number” of each atom—the number of close platinum neighbors. They found that about one fifth of the atoms are highly under-coordinated, with as few as two to six neighbors, reflecting a rough, defect-rich surface rather than a perfectly smooth shape. The partially occupied, most mobile sites almost always correspond to these low-coordination positions, and they form connected paths or networks across certain facets of the nanoparticle. This suggests that, under operating conditions, catalytic activity may be concentrated along these atomic-scale networks of flexible, under-bonded atoms.
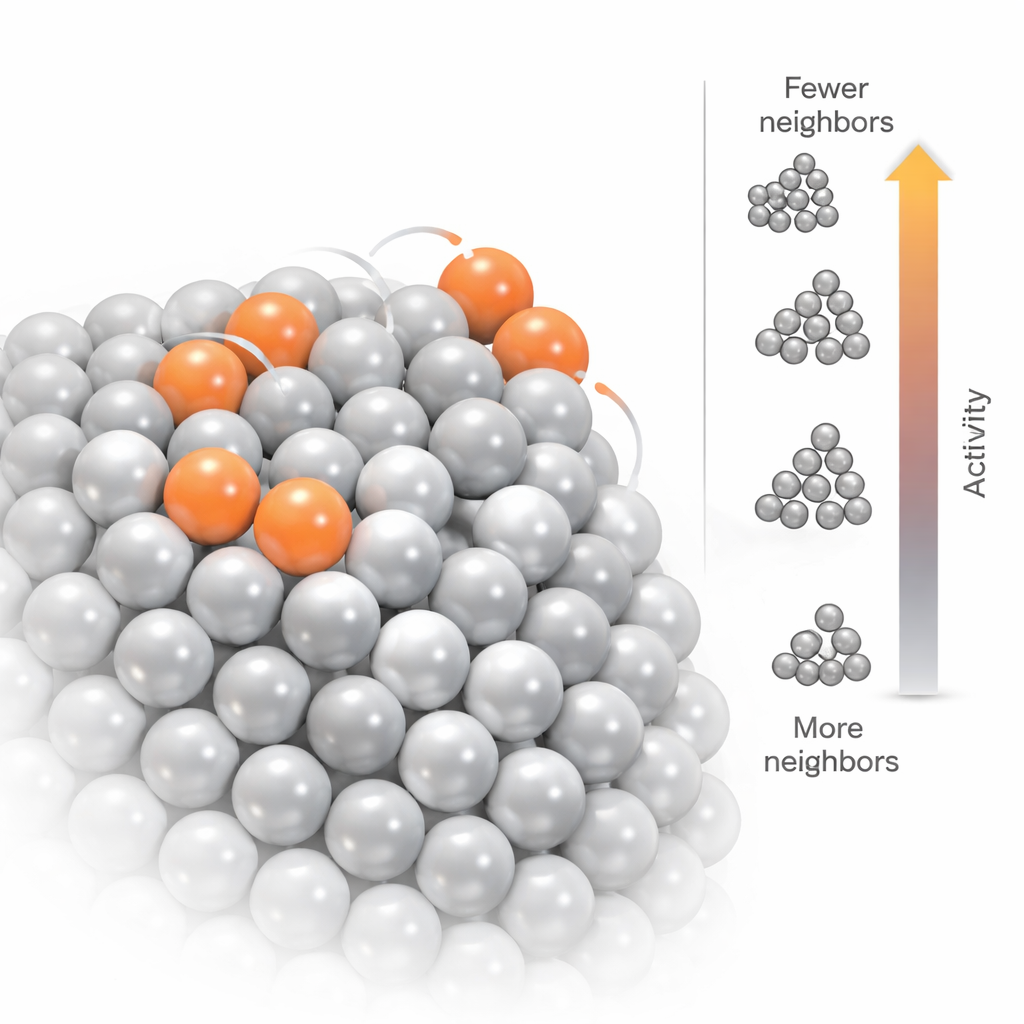
Linking charge and activity to atomic structure
To see how this detailed structure affects chemistry, the team used quantum mechanical calculations based on density functional theory. They started from the experimentally determined 3D model and let the atoms relax slightly to their lowest-energy positions. The calculations show that the nanoparticle as a whole carries a small negative charge, drawn from the oxide support, and that this excess charge piles up at the low-coordination surface atoms. Using a standard “d-band” model of catalytic behavior, they also found that these same under-coordinated atoms have electronic states that bind molecules more strongly, indicating higher catalytic activity. In other words, the very atoms that move the most and have the fewest neighbors are also those most likely to grab and transform reactant molecules.
What this means for better catalysts
For a non-specialist, the key outcome is that catalysts cannot be fully understood as rigid, ideal shapes. This work shows that even a single nanoparticle has a complex, constantly shifting landscape of atomic sites, and that the most active spots are mobile, under-bonded atoms carrying extra negative charge. By directly connecting detailed 3D atomic maps with models of electronic structure and reactivity, the study provides a blueprint for designing better catalysts: tailor the support material and particle shape to create more of these special sites, stabilize them, and control how they move under reaction conditions.
Citation: Ishikawa, R., Kubota, R., Kawahara, K. et al. 3D dynamic structure of a Pt nanoparticle on SrTiO3 (001) during in-situ heating atomic-resolution ADF STEM imaging. Nat Commun 17, 1860 (2026). https://doi.org/10.1038/s41467-026-69767-5
Keywords: platinum nanoparticle catalysis, oxide-supported catalysts, atomic-scale imaging, active sites, nanoparticle dynamics