Clear Sky Science · en
Limitations of serial cloning in mammals
Why Copying Mammals Isn’t as Simple as It Sounds
For decades, the idea of copying animals—right down to their DNA—has captured the public imagination, from Dolly the sheep to dreams of reviving vanished species. But can mammals actually sustain themselves through cloning alone, generation after generation, the way some plants and simple animals do? This study followed one mouse family produced entirely by cloning over nearly 20 years to find out. The results reveal why, despite our technical advances, nature still leans heavily on sex to keep mammals healthy over the long haul.
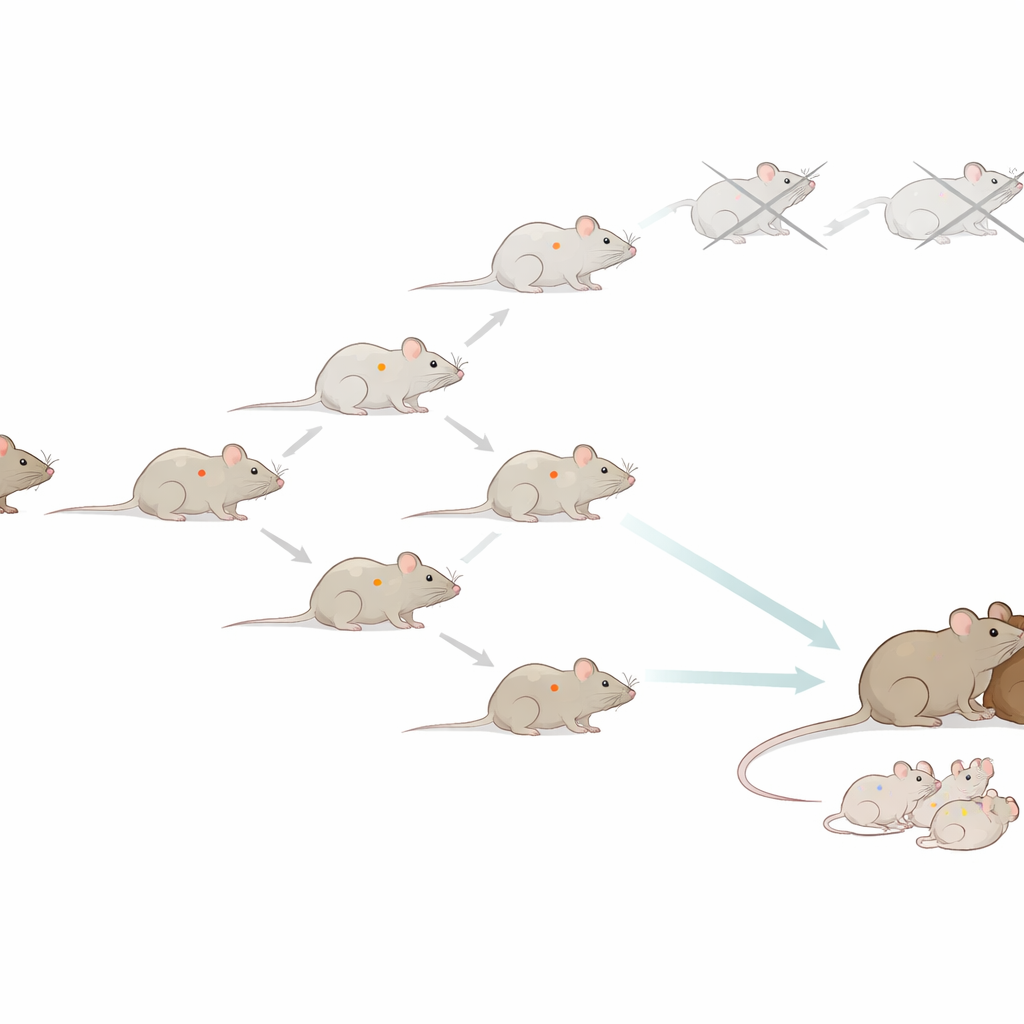
Following One Mouse Line for Twenty Years
The researchers began with a single female mouse and used its body cells to create a cloned mouse. Once that first clone grew up, they took cells from it to make the next clone, and repeated this process again and again. In total, they attempted more than 30,000 nuclear transfer procedures and produced over 1,200 cloned animals, reaching 58 generations from the original donor. Many of these mice looked and behaved like ordinary animals, with normal lifespans of about two years and similar body weights. At first, the success rate of cloning even improved with each generation, suggesting that serial cloning of mammals might be practical in the long run.
Healthy Bodies, But Hidden Damage
On the surface, the cloned mice seemed fine. Their internal organs and especially their placentas showed the usual quirks known from cloning experiments, such as being larger than those of naturally conceived mice, but these abnormalities did not worsen with each generation. The team also checked how well embryos developed in the lab and examined the patterns of chemical marks on DNA-packaging proteins—features that often go wrong in cloning. Early embryos from late-generation clones looked much like those from the first cloned generation. This suggested that the known "reprogramming" problems of cloning were not steadily piling up over time.
Mutations Creep In with Every Copy
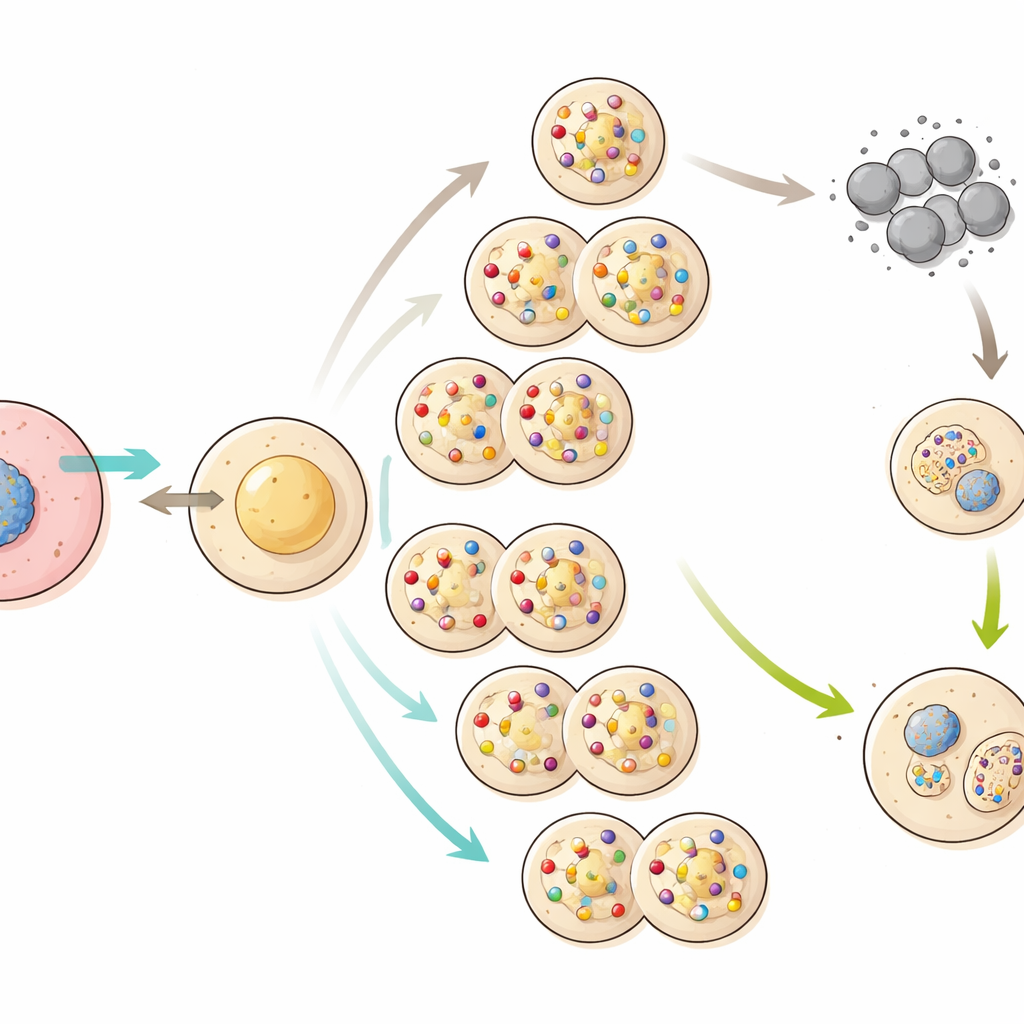
The real story emerged when the scientists sequenced the genomes of mice from different cloning generations. With each round of copying, new changes in the DNA appeared: on average about 70 single-letter mutations and one to two structural alterations per generation, including large rearrangements and chromosome losses. Many of these changes resembled natural mutations seen in ordinary reproduction, but the key difference was that, in cloning, there is no reshuffling of chromosomes through mating to help weed out harmful variants. Over time, particularly after about the 25th generation, damaging mutations—such as the loss of an entire X chromosome or breaks and swaps between chromosomes—began to accumulate. The proportion of mutations likely to disrupt important genes nearly doubled in later generations.
Egg Cells Reveal the Breaking Point
To see how this hidden damage affected reproduction, the team focused on egg cells from late-generation cloned females. When these eggs were triggered to develop without sperm, almost none grew into healthy early-stage embryos, indicating that many carried lethal combinations of mutations. Fertilization with sperm from normal males improved development somewhat, but success still dropped sharply in later generations. Experiments that swapped the nuclei and surrounding material between eggs from normal and late-generation cloned mice showed that both the genetic material and the egg cytoplasm had become compromised. Eventually, by the 58th generation, all cloned offspring died shortly after birth and the cloning line could not be continued.
Sexual Reproduction as a Repair System
When the scientists allowed late-generation cloned mice to mate naturally with normal males, a different pattern appeared. Although these cloned mothers had small litters, some of their offspring—and especially their grandchildren—were much healthier. The next generation’s placentas shrank back toward normal size, and overall development improved. This suggests that the mixing and sorting of chromosomes during egg and sperm formation, followed by fertilization, can break up damaging mutation combinations and filter out many of the worst changes. In other words, sex acted as a built-in clean-up system that pure cloning lacks.
What This Means for the Future of Cloning
By carefully tracking a single cloned mouse lineage across 57 successful generations, this work shows that mammals cannot rely on cloning alone to sustain a lineage indefinitely. Visible health and normal lifespans can mask a slow genetic decline, as mutations quietly build up until reproduction fails. While cloning remains a powerful tool—for rescuing endangered animals, multiplying valuable livestock, or preserving genetic resources—it is unlikely to replace sexual reproduction as a long-term strategy for mammalian survival. The study provides strong experimental support for the idea that, in complex animals, sex is not just about making offspring—it is essential housekeeping for the genome.
Citation: Wakayama, S., Ito, D., Inoue, R. et al. Limitations of serial cloning in mammals. Nat Commun 17, 2495 (2026). https://doi.org/10.1038/s41467-026-69765-7
Keywords: animal cloning, genetic mutations, mouse reproduction, asexual reproduction, evolution of sex