Clear Sky Science · en
Amorphous/crystalline interwoven multipods with high Co/Ni activity for wide-temperature-range sodium-sulfur batteries
Why Better Batteries Matter in All Weather
Modern life runs on rechargeable batteries, but many of them struggle when the weather turns icy or sweltering. Sodium–sulfur batteries are an appealing, low-cost option for storing renewable energy, yet they tend to lose power in the cold and age quickly in the heat. This study introduces a new battery material that keeps sodium–sulfur cells working efficiently from well below freezing to a hot summer’s day, bringing us closer to robust, all‑climate energy storage. 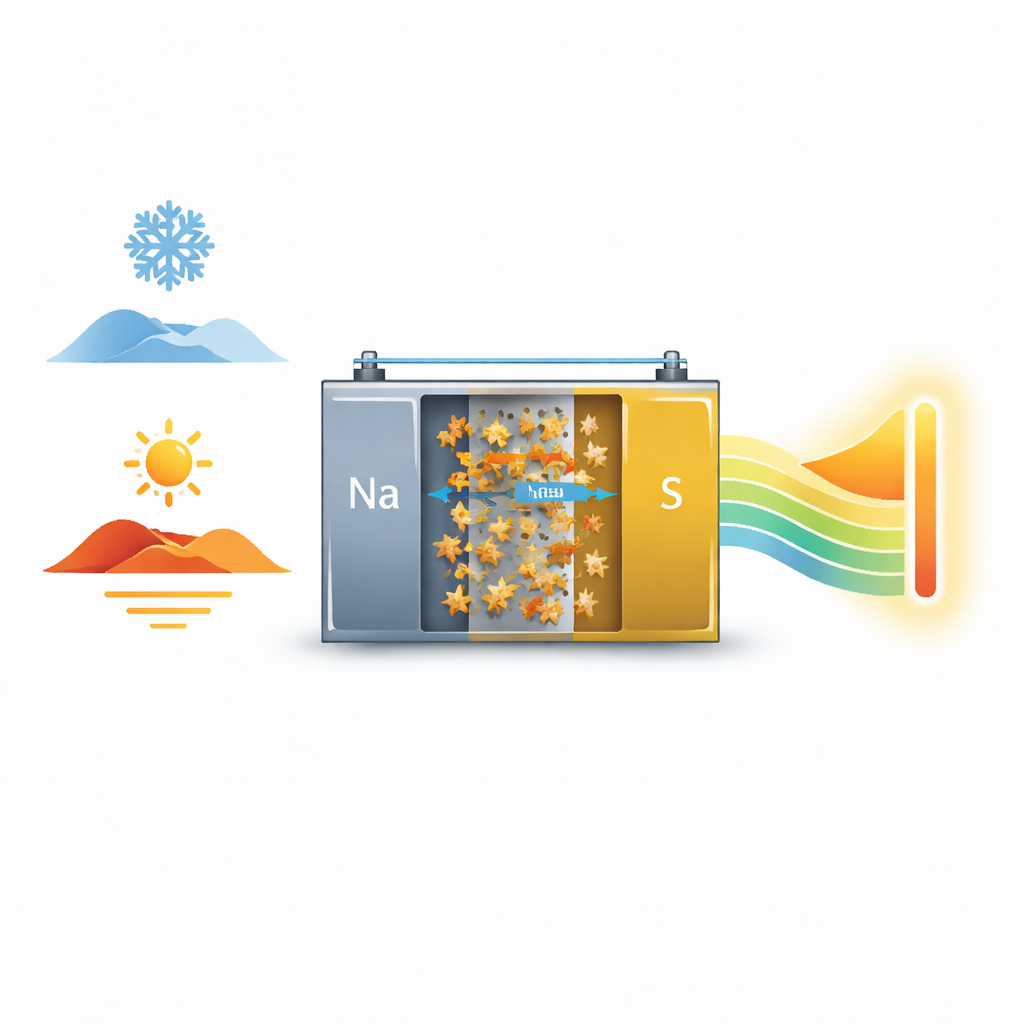
A Simple Idea Behind a Complex Battery
Sodium–sulfur batteries use abundant elements: metallic sodium on one side and sulfur on the other. During charging and discharging, sodium and sulfur pass through a complex sequence of chemical steps involving many electrons. In theory this makes the battery very high in capacity, but in practice it slows everything down and forms intermediate compounds, called sodium polysulfides, that can dissolve and drift away inside the battery. The result is sluggish performance, fading capacity, and especially poor behavior when it is very cold or very hot.
Designing a Tiny Star-Shaped Helper
The researchers tackled these limitations with a specially designed catalyst that sits in the sulfur electrode and helps steer the reactions. They built tiny “multipods” – star‑like particles made of cobalt and nickel sulfide – and subtly changed their structure by adding a small amount of tin during synthesis. This tin addition disrupts crystal growth, producing a curious mix: regions where atoms are neatly ordered (crystalline) interwoven with regions where the arrangement is more disordered (amorphous). These multipods are grown on thin sheets of a conductive material called MXene, which acts like a scaffold and highway for electrons.
How the Mixed Structure Speeds and Controls Reactions
By examining the material with advanced microscopes and spectroscopic tools, the team showed that the multipods truly intertwine ordered and disordered regions. The ordered parts offer fast pathways for electrons, while the disordered parts provide abundant “landing spots” where sodium polysulfides can stick and react. The tin‑induced structure also tweaks the electronic environment of cobalt and nickel atoms, creating more sulfur vacancies and strengthening their bonds to the polysulfides. Computer simulations back this up, revealing that key reaction steps – especially the conversion of short sulfur species into the final solid product – require less energy on this mixed material than on a fully crystalline version, meaning the process can run faster and more smoothly. 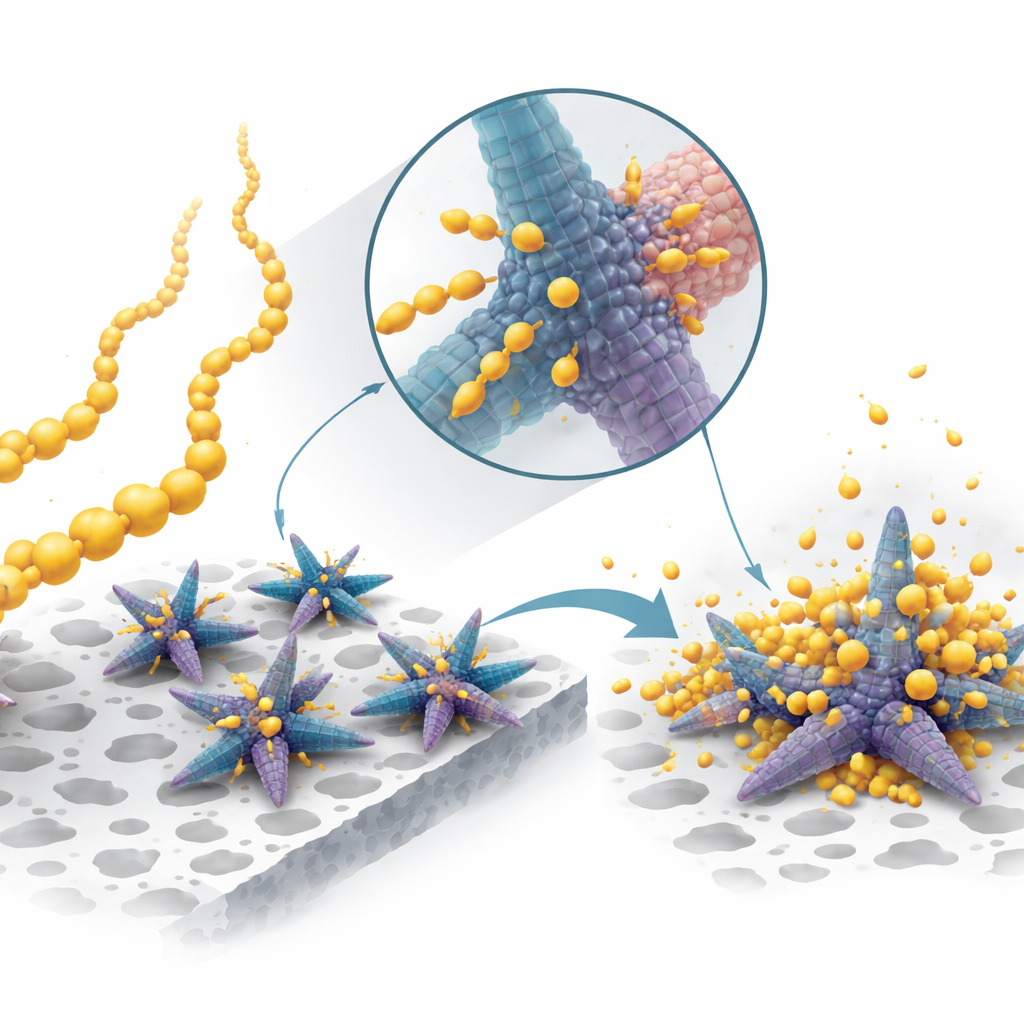
Proving Performance from Freezing to Hot
To test whether this design actually improves a real battery, the researchers built sodium–sulfur cells using their multipod catalyst loaded with sulfur. At room temperature, these cells delivered very high capacity and maintained it over more than a thousand charge–discharge cycles, with only tiny losses each time. At –20 °C, where usual sodium–sulfur batteries suffer from sluggish chemistry, the new cells still provided strong capacity and stable cycling at demanding current levels. At 50 °C, where dissolved polysulfides normally run rampant and damage the cell, the batteries kept most of their capacity over hundreds of cycles. Measurements of electrical resistance and ion movement confirmed that the mixed structure keeps reactions fast even in the cold, while adsorption tests showed it grabs and holds polysulfides effectively, limiting the internal “shuttle” that drains performance in the heat.
What This Means for Future Energy Storage
In everyday terms, the study demonstrates a smart way to make sodium–sulfur batteries both powerful and resilient, regardless of the season. By weaving together ordered and disordered regions inside a tiny catalyst particle and fine‑tuning the local atomic environment, the researchers lowered the barriers that slow the battery reactions and trapped the troublesome intermediate species that usually cause trouble. This approach to engineering interfaces within materials could be applied to many kinds of batteries, offering a path toward cheaper, high‑capacity storage that can reliably support renewable energy grids in cold winters, hot summers, and everything in between.
Citation: Xiao, T., Fang, Z., Ran, N. et al. Amorphous/crystalline interwoven multipods with high Co/Ni activity for wide-temperature-range sodium-sulfur batteries. Nat Commun 17, 2333 (2026). https://doi.org/10.1038/s41467-026-69749-7
Keywords: sodium–sulfur batteries, energy storage, battery catalysts, wide-temperature operation, amorphous–crystalline interfaces