Clear Sky Science · en
Synergistic sulfur-chlorine battery chemistry towards efficient energy storage
Why squeezing more from every watt matters
As the world leans heavily on solar panels and wind farms, a surprising amount of that green electricity is quietly wasted when it is stored. Today’s large batteries often give back much less energy than they take in, meaning thousands of terawatt-hours are effectively thrown away each year. This paper describes a new kind of rechargeable battery that wastes almost nothing: it can return up to 99.5% of the energy put into it. For anyone who cares about cutting costs, shrinking carbon footprints, or powering devices in harsh places like polar regions or deep sea, such ultra-efficient storage could be a game changer.
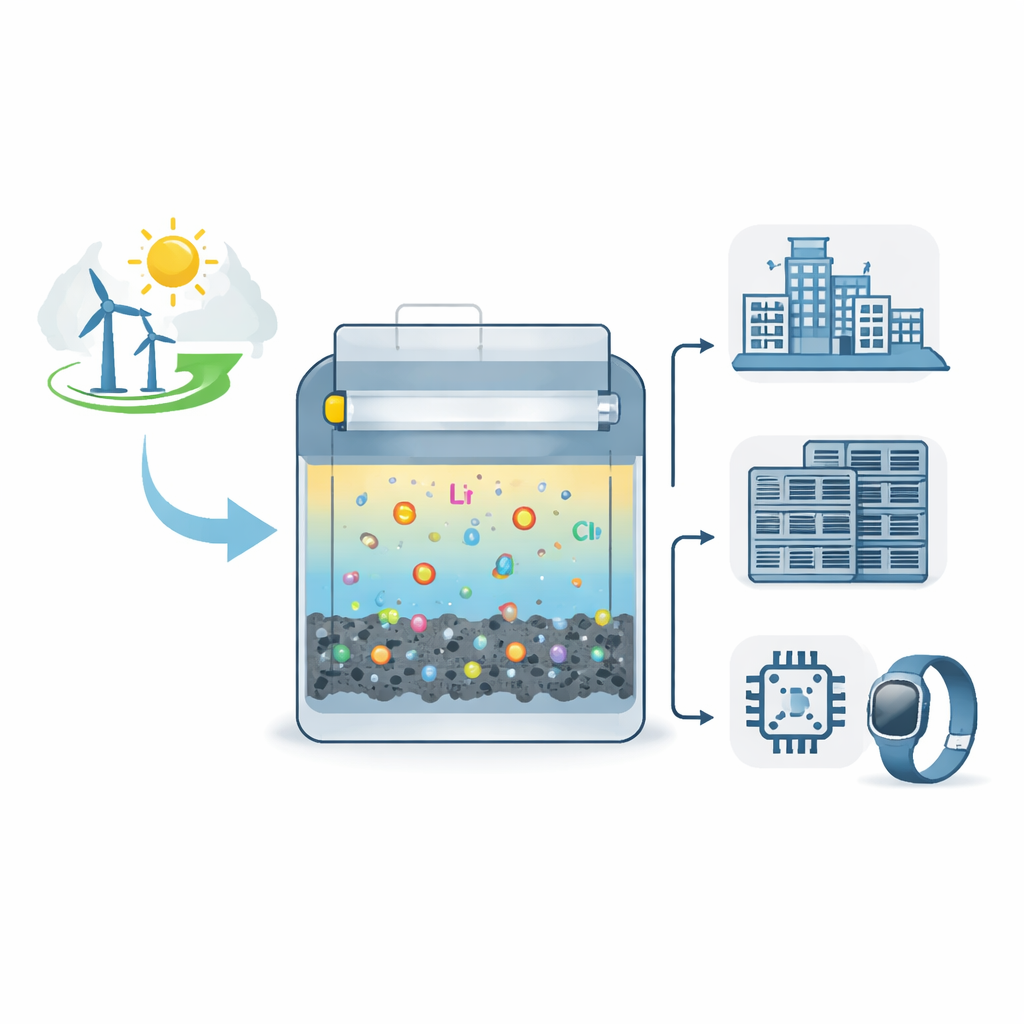
A new twist on common battery ingredients
Most familiar batteries, like those in phones and electric cars, move lithium ions in and out of solid materials. Another class, called conversion-type batteries, instead transforms one set of molecules into another during charging and discharging. These systems can be cheap and energy-dense but usually suffer big energy losses and sluggish reactions. The authors tackled this problem by designing a lithium battery that uses a liquid called sulfuryl chloride (SO2Cl2) together with chlorine chemistry at the positive side of the cell. In their design, the liquid serves both as the solvent and as an active energy-storing material, while a simple porous carbon acts as the support where reactions take place.
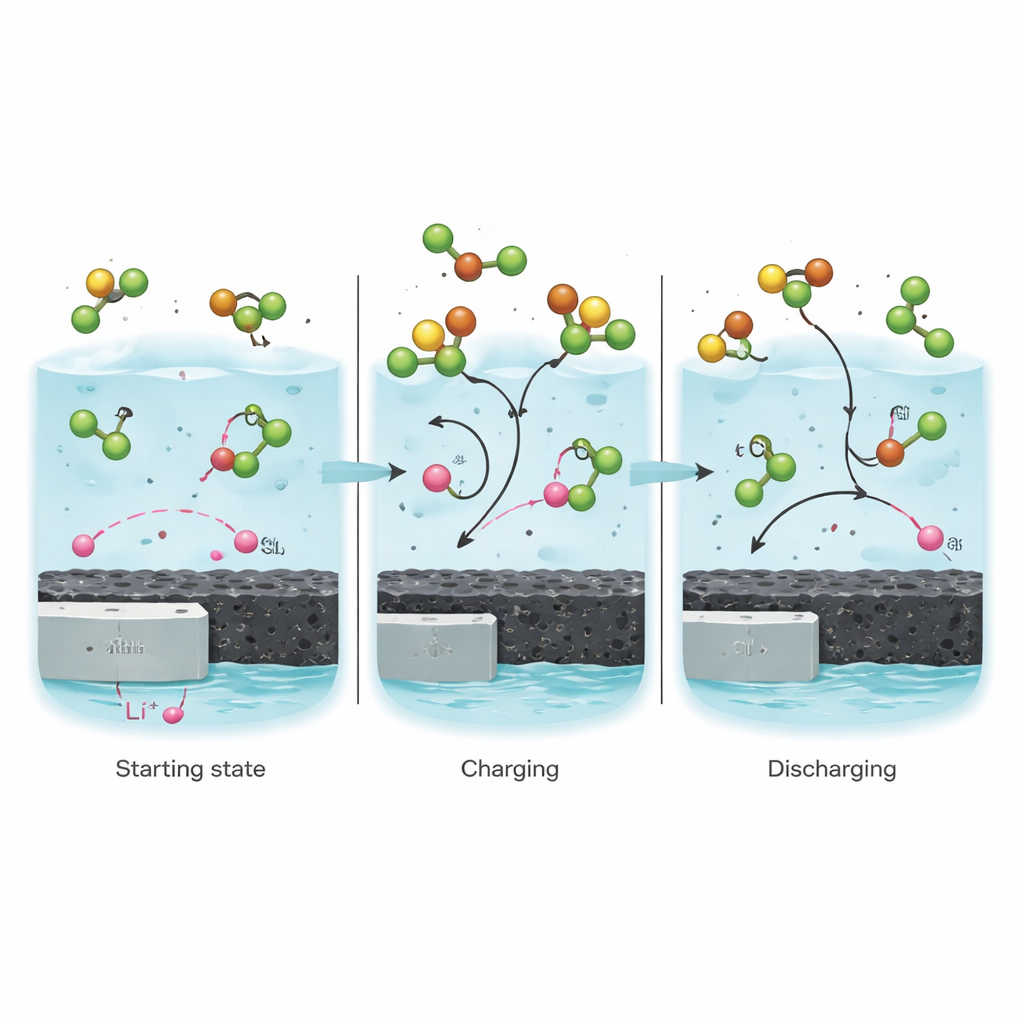
How sulfur and chlorine work together
Inside this battery, sulfur and chlorine atoms do not act alone; they participate in a tightly linked reaction network that the authors call synergistic S–Cl chemistry. When the battery discharges along its preferred pathway, sulfur in the liquid is partially reduced and lithium chloride forms on the carbon, while the lithium metal on the negative side is consumed. During charging, chlorine gas is generated in situ and plays a crucial mediating role: it helps drive a highly reversible back-and-forth conversion between sulfur dioxide (SO2) and sulfuryl chloride (SO2Cl2). Using advanced tools such as X-ray absorption and mass spectrometry, the team shows that this chlorine-assisted loop lowers reaction barriers, so the chemistry proceeds quickly and cleanly with only tiny voltage losses.
Breaking records for efficiency and speed
Because the reactions move so easily, the battery operates with an exceptionally small gap—only about 9 millivolts—between its charge and discharge voltages under typical conditions. That translates into an energy storage efficiency of up to 99.5%, much higher than most existing conversion-type batteries, which usually reach only 59–95% and waste far more energy as heat. The system maintains very high efficiencies, generally 93–97%, even when pushed to demanding conditions: high capacities, rapid cycling, and low temperatures down to –20 °C. The fast sulfur–chlorine interplay also allows very large currents, with demonstrated discharge current densities up to 400 milliamperes per square centimeter—one to three orders of magnitude higher than many comparable designs—without forming dangerous, needle-like lithium deposits.
From tiny chips to large-scale storage
Beyond demonstrating basic performance in lab cells, the researchers built several practical prototypes. A 250 milliampere-hour pouch cell using the same chemistry achieved over 96% energy efficiency at realistic loading levels, indicating that the concept can be scaled. They also made a millimeter-scale microbattery that powered a chip capable of measuring temperature and pressure and sending the data wirelessly, and a flexible fiber-shaped battery suitable for wearable devices, both benefiting from the chemistry’s high power and the nonflammable electrolyte. The system’s long shelf life and robust low-temperature behavior suggest it could serve in emergency backup supplies, space missions, and deep-sea instruments where replacing or recharging batteries is difficult.
What this means for future clean energy
In simple terms, this work shows that smartly combining sulfur and chlorine reactions can all but eliminate energy loss in a rechargeable battery while keeping power output high. By using chlorine formed inside the cell to steer the sulfur chemistry along an easier, faster path, the authors achieve near-perfect round-trip efficiency and very rapid charge and discharge. This not only points to better batteries for grids, electronics, and wearables, but also offers a design blueprint: pairing elements that assist one another at the molecular level can dramatically improve how well we store renewable electricity.
Citation: Zhao, X., Liao, M., Geng, S. et al. Synergistic sulfur-chlorine battery chemistry towards efficient energy storage. Nat Commun 17, 3088 (2026). https://doi.org/10.1038/s41467-026-69748-8
Keywords: high-efficiency batteries, sulfur chlorine chemistry, energy storage, lithium conversion batteries, renewable power storage