Clear Sky Science · en
TCL1A mediates DNA methylation defects in recurrent hydatidiform mole with NLRP7 pathogenic variants
Why this matters for women’s health
Some pregnancies go wrong very early, turning into a mass of abnormal placental tissue instead of a developing baby. This condition, called a hydatidiform mole, can recur in some women and sometimes progress to cancer. This study digs into one major genetic cause of these rare but serious pregnancies and uncovers how a single faulty safeguard in the egg can derail the chemical “labels” that guide healthy development.
A pregnancy problem rooted in the egg
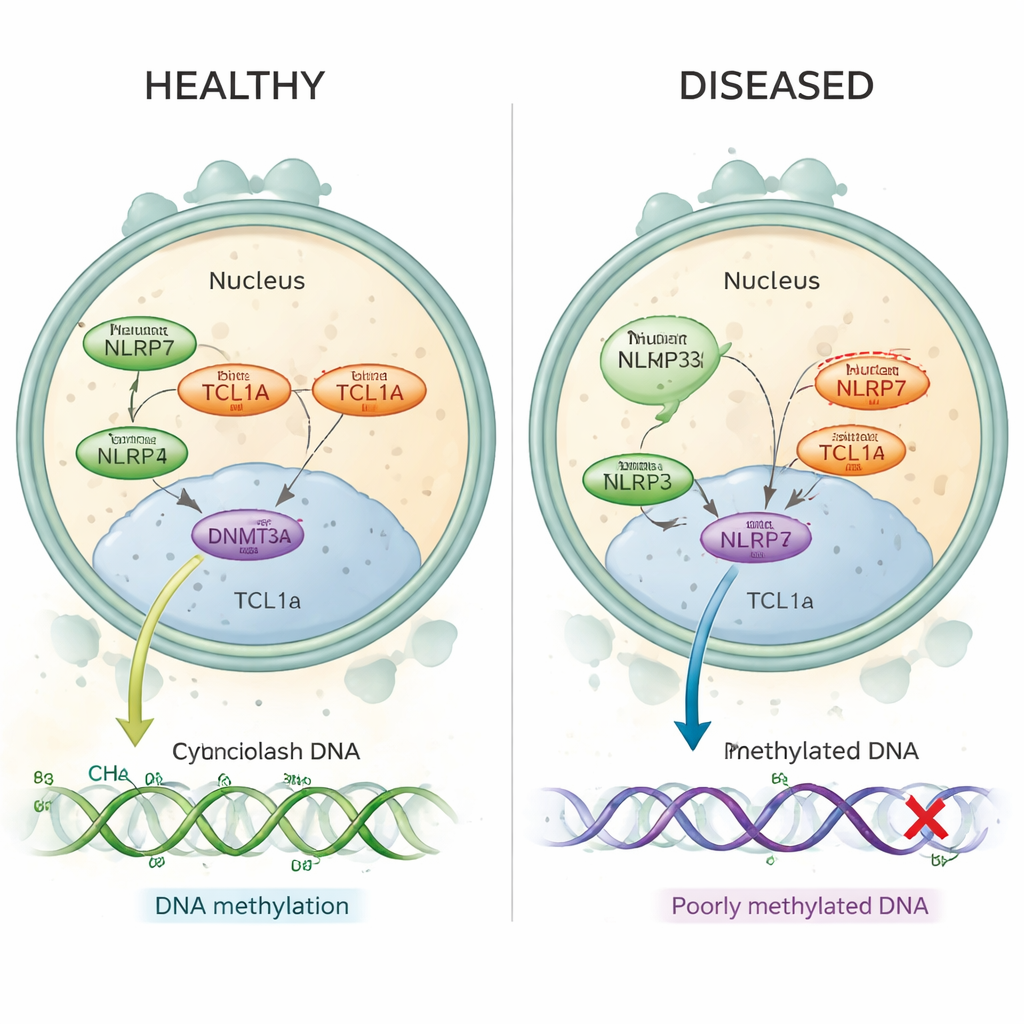
Recurrent hydatidiform mole is a disorder in which women experience repeated molar pregnancies, often without any normal pregnancies in between. Many such cases are now known to be caused by harmful changes in a gene called NLRP7, which is active in the egg before and just after fertilization. In these women, key stretches of DNA that should carry “imprints” from the mother lack normal methylation marks—small chemical tags that help switch genes on or off at the right time. Until now, scientists did not understand how a protein that sits in the egg’s cytoplasm, like NLRP7, could control methylation that happens on DNA packaged inside the nucleus.
Finding a missing partner
To solve this puzzle, the researchers examined discarded human eggs and very early embryos from fertility clinics. They pulled down NLRP7 and its known partners from these cells and identified other proteins that come along for the ride. One stood out: TCL1A, already known in blood cancers as a protein that can move into the nucleus and interfere with DNA methylating enzymes called DNMT3A and DNMT3B. TCL1A is unusually abundant in human eggs, suggesting it plays an important role there. Detailed interaction tests showed that TCL1A binds tightly and specifically to NLRP7, but not to closely related proteins, and that this complex is wired into a larger egg-specific structure called the subcortical maternal complex.
Seeing the molecular embrace
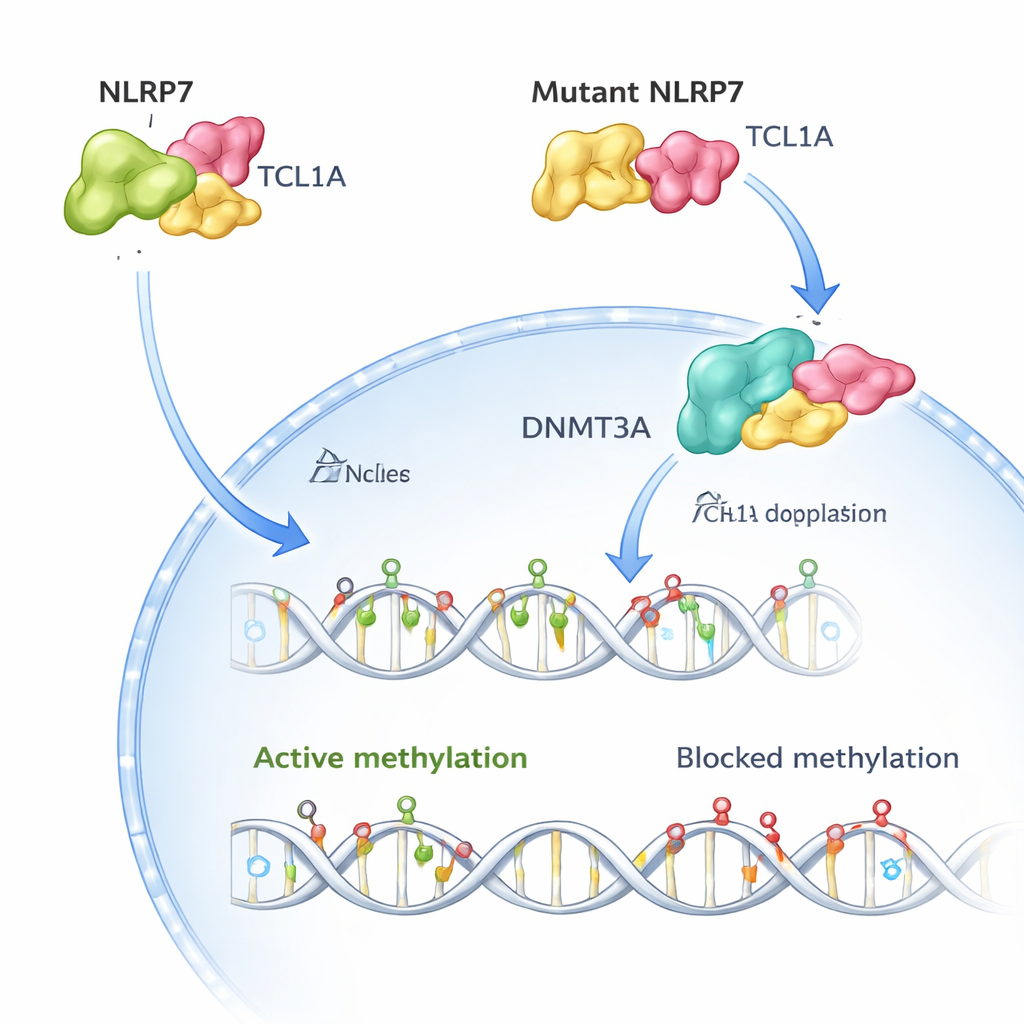
The team then used cryo–electron microscopy to visualize the three‑dimensional structure of the NLRP7–TCL1A complex. They found that two NLRP7 molecules pair up and each one grips a TCL1A dimer along a curved surface of repeated motifs. This arrangement explains why many disease‑causing changes in NLRP7 cluster along that surface: altering key contact points weakens or destroys the embrace with TCL1A. When the researchers recreated more than 50 known patient variants in cells, most of the versions linked to recurrent molar pregnancies either destabilized NLRP7 or sharply reduced its ability to bind TCL1A.
How misplaced proteins unsettle DNA marks
In healthy human eggs, both NLRP7 and TCL1A are mainly found in the cytoplasm, with only a small amount of TCL1A reaching the nucleus. The authors show that NLRP7 effectively acts as a gatekeeper: when it can hold TCL1A, TCL1A stays out of the nucleus. When NLRP7 is mutated and can no longer bind well, TCL1A leaks into the nucleus. There it attaches to DNMT3A, the main enzyme that lays down new methylation marks in eggs, and dampens its activity. In stem‑cell models that normally gain methylation as they mature, overproducing TCL1A caused a dramatic loss of methylation across the genome, while co‑expressing NLRP7 partially rescued this defect. Together, these findings support a simple picture: normal NLRP7 keeps a methylation “brake” (TCL1A) locked in the cytoplasm so DNMT3A can properly label DNA; faulty NLRP7 lets that brake slip into the nucleus and block the process.
From mechanism to diagnosis
Beyond explaining how recurrent molar pregnancies arise, the study suggests a practical way to judge whether a newly discovered NLRP7 change in a patient is truly harmful. The authors compare three approaches—laboratory tests of NLRP7–TCL1A binding, computer‑based predictions, and standard genetic scoring tools—and show that loss of TCL1A binding aligns closely with variants that cause disease. They also uncover a previously unrecognized harmful variant, L766R, in families with recurrent moles, confirming that it both weakens the protein and misdirects TCL1A into the nucleus.
What this means in simple terms
This work reveals a molecular chain reaction behind a rare but devastating pregnancy disorder. In essence, eggs from affected women carry a broken “bodyguard” protein, NLRP7, that fails to keep its partner TCL1A out of the nucleus. Once inside, TCL1A interferes with the enzyme that writes vital chemical tags on DNA. Without those tags, the early placenta grows abnormally and a baby cannot form. By tracing this pathway step‑by‑step, the study clarifies why certain genetic changes in mothers can repeatedly sabotage pregnancies and points toward more precise genetic counseling and diagnosis for women with recurrent hydatidiform moles.
Citation: Gao, Z., Liu, Q., Li, L. et al. TCL1A mediates DNA methylation defects in recurrent hydatidiform mole with NLRP7 pathogenic variants. Nat Commun 17, 2160 (2026). https://doi.org/10.1038/s41467-026-69744-y
Keywords: DNA methylation, recurrent hydatidiform mole, NLRP7, TCL1A, genomic imprinting