Clear Sky Science · en
Non-chelation control in allylations of α-oxy ketones using group-14 allylatranes
Building Molecular Shapes with Greater Precision
Many medicines and natural products work only when their atoms are arranged in just the right three‑dimensional shape. Chemists therefore spend enormous effort learning how to "push" new pieces onto a molecule from one side or the other. This article describes a new way to attach allyl groups—short three‑carbon chains—to a common class of molecules, giving access to mirror‑image structures that have been very hard to make until now.
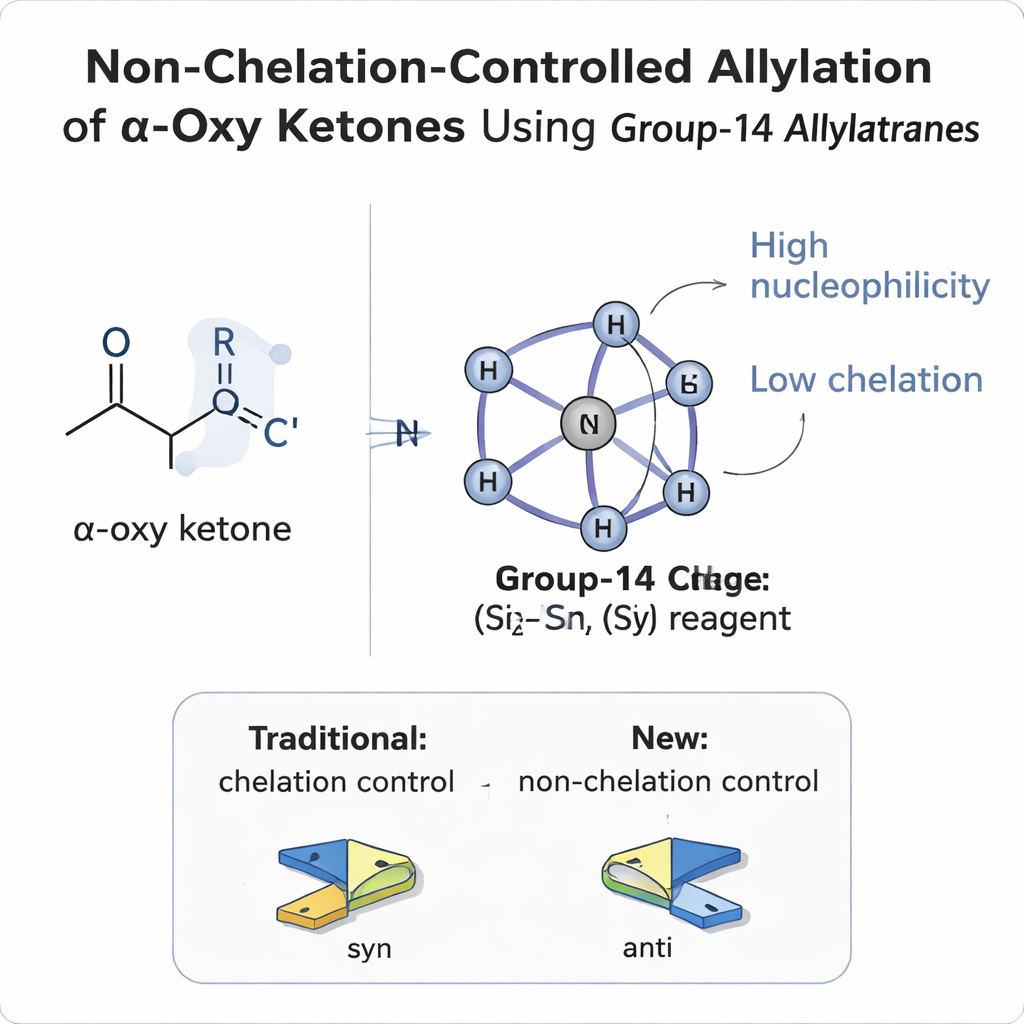
Why Controlling Sides of a Molecule Is So Hard
When a new group is added to a flat, planar carbon–oxygen double bond (a carbonyl), it can attack from either face, like a ball hitting a coin from above or below. If there is already a neighboring substituent at the carbon next door (the α‑position), the result is two possible three‑dimensional products, called diastereomers. For decades, chemists have relied on simple models—Felkin–Anh, polar Felkin–Anh, Cram, and chelation models—to predict which side a reagent will prefer. In α‑oxy carbonyl compounds, where that neighboring substituent is an oxygen‑containing group (such as an ether or ester), the oxygen usually acts like a claw that grabs onto metal reagents. This "chelation" locks the molecule into one conformation and almost always leads to so‑called syn products, where the newly created alcohol groups end up on the same side of the carbon chain.
The Long‑Standing Problem with Oxygen Neighbors
While the chelation pathway is very useful, it is also limiting: it strongly biases reactions toward syn products and makes it very difficult to obtain the opposite, anti arrangement, where the two alcohol groups point to opposite sides. For α‑oxy aldehydes—molecules where the carbonyl carbon is attached to at least one hydrogen—a few clever tricks using special silicon reagents and carefully chosen Lewis acids have managed to deliver anti products. But for α‑oxy ketones, which are less reactive and more common in complex targets, the challenge is greater. Stronger nucleophiles tend to be more Lewis acidic, which in turn encourages exactly the chelation the chemist wants to avoid. Thus, balancing "reactive enough" with "not too sticky" toward oxygen has been a central unsolved problem.
A Cage‑Like Reagent that Breaks the Rule
The authors introduce a new family of reagents called allylatranes, built around group‑14 elements—silicon, germanium, and tin—held in a rigid, cage‑like framework. In these molecules, an internal nitrogen atom reaches across the cage to bond to the central atom, creating a highly coordinated, almost encapsulated metal center. This design has two crucial effects. First, it boosts the nucleophilicity of the attached allyl group, making it eager to form a new carbon–carbon bond. Second, it tames the Lewis acidity of the central atom, so it is much less inclined to bind strongly to the oxygen atoms of the substrate. Quantum‑chemical calculations and nuclear magnetic resonance data confirm that charge is localized on the allyl fragment while electronic communication with the silicon center is reduced, explaining how the reagent can be both powerful and yet reluctant to chelate.
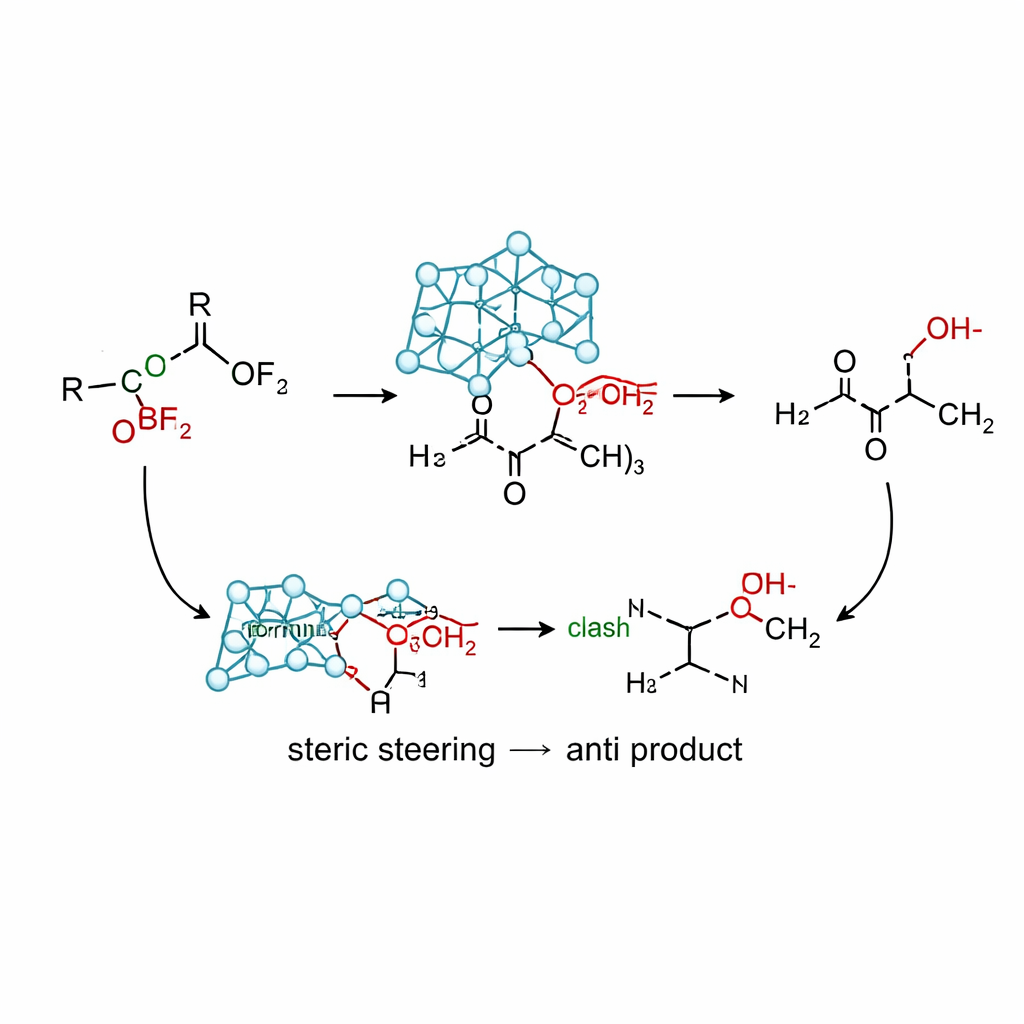
How the New Reaction Works in Practice
Using the silicon version of this reagent, allylsilatrane, together with a mild Lewis acid (boron trifluoride), the team developed conditions that give high yields of anti‑homoallylic alcohols from a wide range of α‑oxy ketones. Benchmark experiments show that many traditional allyl sources—based on tin, indium, magnesium, lithium, and simple silanes—either favor the syn product, give mixtures, or decompose. In contrast, allylsilatrane routinely delivers anti/syn ratios above 95:5 across substrates bearing methoxy, isopropoxy, phenoxy, acetoxy, silyloxy, and aminoxy groups, as well as various aromatic rings and cyclic ketones. Computations suggest a non‑chelation pathway in which the bulky atrane framework steers the approach of the allyl group into a conformation resembling a Cram model: the oxygen substituent lies opposite the carbonyl while steric repulsion between the cage and nearby phenyl rings favors the trajectory that leads to the anti product.
Implications for Drug and Natural Product Synthesis
The method also works for more rigid cyclic systems, where the bulk of the allylsilatrane encourages attack from less hindered positions, and for specialized derivatives that transfer substituted allyl groups with similarly high anti selectivity. Because the resulting anti‑1,2‑diols with a homoallylic fragment are common motifs in biologically active molecules, including candidates for enzyme modulators, this transformation offers chemists a reliable way to access structural variants that previously required multistep workarounds or were simply impractical. Importantly, the same substrates can be directed to give either syn or anti products by switching between a chelating tin system and the new non‑chelating silatrane system, granting precise control over molecular shape.
What This Means in Simple Terms
In essence, the researchers have built a smart allyl‑delivery tool that hits the target carbon from the "unfavored" side without being trapped by nearby oxygen atoms. By carefully engineering a cage around silicon, they decoupled strength and stickiness: the reagent is strong enough to forge a new bond but not so sticky that it grabs surrounding atoms and ruins the desired geometry. For non‑specialists, this means chemists now have a much better handle on sculpting three‑dimensional molecular structures that underpin many drugs and natural products, expanding the design space for future medicines and complex synthetic molecules.
Citation: Tsutsui, Y., Shiga, K., Konishi, A. et al. Non-chelation control in allylations of α-oxy ketones using group-14 allylatranes. Nat Commun 17, 2019 (2026). https://doi.org/10.1038/s41467-026-69732-2
Keywords: stereoselective allylation, alpha-oxy ketones, allylsilatrane, non-chelation control, homoallylic alcohols