Clear Sky Science · en
Arginase 1 promotes hepatic lipogenesis by regulating ERK2/PPARγ signaling in a non-canonical manner
Why liver fat matters to everyday health
Many people carry extra weight around the waist, but few realize that hidden fat in the liver can quietly drive diabetes, heart disease, and even liver cancer. This study explores why some livers pack on fat while others stay lean, and uncovers an unexpected role for a long-known liver enzyme, arginase 1 (Arg1). By showing how Arg1 helps the liver make and store fat, and how blocking a specific protein handshake can reverse this process in mice, the work points toward new treatment ideas for obesity and fatty liver disease.
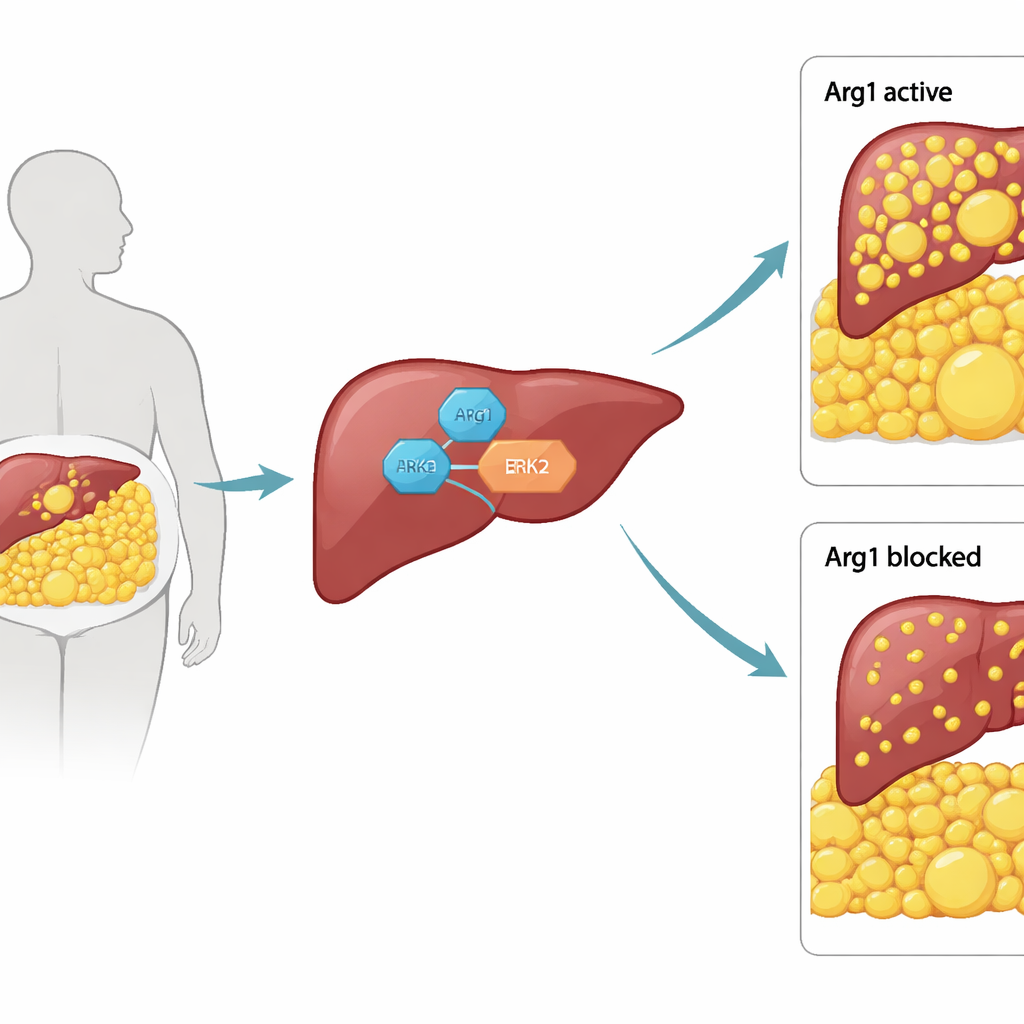
A surprising job for a familiar liver enzyme
Arg1 is best known as a workhorse of the urea cycle, a pathway the liver uses to detoxify ammonia by turning the amino acid arginine into urea. Because of that classic role, most researchers assumed Arg1 simply supported basic metabolism. The authors instead asked whether Arg1 might also influence how the liver handles fat. They began by removing Arg1 only from liver cells in mice. These animals ate normally, but they were smaller, had lighter livers, and showed dramatically less fat in both liver and body fat stores. Blood and tissue measurements confirmed lower triglycerides and free fatty acids, and genes that normally drive fat production, especially those controlled by the master regulator PPARγ, were turned down.
From enzyme loss to better metabolic health
To separate Arg1’s role from the toxic effects of completely losing urea-cycle function, the team created an adult-onset Arg1 knockout and also used a chemical Arg1 blocker. In multiple mouse models of severe obesity and fatty liver—genetic obesity, high-fat diet, and a special diet that rapidly injures the liver—reducing Arg1 activity consistently led to less weight gain, smaller fat pads, and strikingly less fat and inflammation in the liver. These benefits appeared without major changes in food intake or overall energy use, suggesting that Arg1 directly shapes how the liver makes and exports fat, rather than simply changing how much the animals eat or burn.
How a protein partnership steers fat production
Diving into the molecular machinery, the researchers discovered that Arg1 acts in a completely different way than expected. Instead of working through its usual chemistry on arginine, Arg1 physically binds to a signaling protein called ERK2 at a small structural pocket. This contact uses a flexible "S-shaped" tail on Arg1 and interferes with other proteins that usually dock at the same site. When Arg1 is present, it promotes tagging of ERK2 for breakdown, reducing ERK2 levels. That shift turns up two major pathways, involving AKT, mTOR, and transcription factors like Elk1 and c-Fos, which ultimately raise PPARγ activity and push liver cells to make and store more fat. When Arg1 is removed or blocked, ERK2 accumulates and rebalances these pathways in a way that restrains PPARγ and lipogenesis.
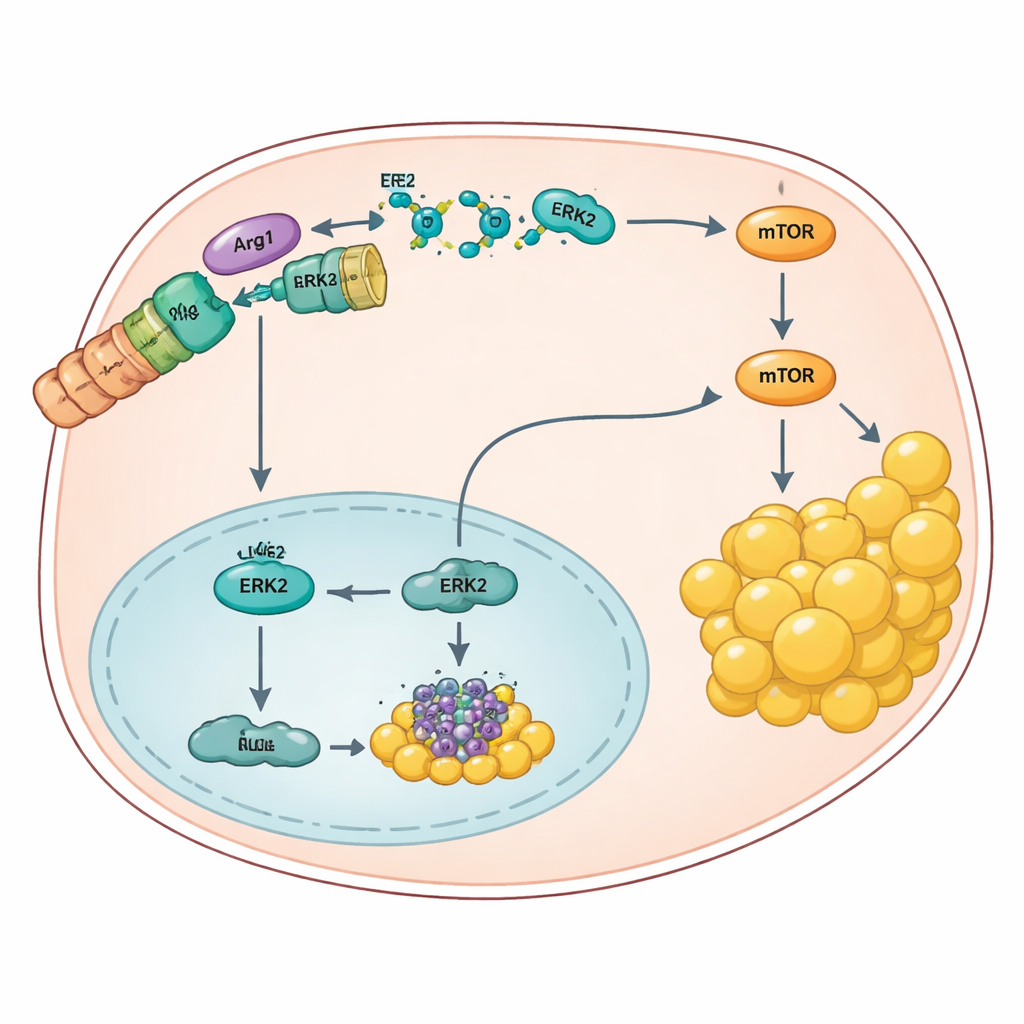
Designer peptides that uncouple the harmful link
Armed with this mechanism, the team engineered short protein fragments, or peptides, that mimic ERK2’s binding pocket. These peptides slip into liver cells and bind Arg1’s S-shaped motif, preventing Arg1 from gripping ERK2. In obese mice fed a high-fat or liver-injuring diet, daily peptide treatment shielded ERK2 from degradation, lowered liver enzymes that signal injury, and significantly reduced fat accumulation in both liver and body fat depots. Importantly, this strategy did not cause the dangerous buildup of arginine that can occur when Arg1’s classical enzyme activity is completely lost, hinting that the physical interaction with ERK2 can be targeted separately from the urea cycle.
What this means for future therapies
To a non-specialist, the takeaway is that Arg1 wears two hats: one for detoxifying nitrogen, and another for quietly turning up liver fat production through a specific protein partnership with ERK2. By disrupting only that partnership—without fully shutting down Arg1’s core job—the study shows it is possible, at least in male mice, to ease obesity-related fatty liver and improve metabolic health. While much work remains before such peptides or similar drugs could be tested in people, the findings reveal a precise new target at the crossroads of liver chemistry and cell signaling that could one day help treat obesity and metabolic liver disease.
Citation: Shao, M., Cao, X., Chen, Y. et al. Arginase 1 promotes hepatic lipogenesis by regulating ERK2/PPARγ signaling in a non-canonical manner. Nat Commun 17, 2903 (2026). https://doi.org/10.1038/s41467-026-69731-3
Keywords: fatty liver, obesity, arginase 1, liver metabolism, signal pathways