Clear Sky Science · en
The N-Myc MB0-MBI region interacts specifically and dynamically with the N-lobe of Aurora kinase A
Why this matters for cancer treatment
Many aggressive childhood cancers rely on a protein called N-Myc to keep tumor cells growing. N-Myc itself is hard to block with drugs because it behaves more like a floppy piece of spaghetti than a rigid lock a key can fit into. This study reveals, in unprecedented detail, how N-Myc grips another protein, the enzyme Aurora kinase A (AurA), to boost its activity and help cancer cells survive. By mapping this partnership, the researchers uncover a realistic way to break it apart with small molecules.
A flexible troublemaker finds a solid partner
N-Myc belongs to the MYC family of master switches that control cell growth, metabolism, and division. When overproduced, these switches become powerful cancer drivers, especially in tumors of the nervous system such as high-risk neuroblastoma in children. Unlike many enzymes or receptors, N-Myc does not fold into a fixed 3D shape on its own; it is an “intrinsically disordered” protein that constantly wiggles and reshapes. This flexibility makes it adaptable but also notoriously difficult to target with drugs. AurA, by contrast, is a well-structured enzyme that helps organize cell division. Previous work showed that AurA helps stabilize N-Myc in tumor cells and that the two proteins physically interact, but the exact nature and importance of the contact points remained unclear.
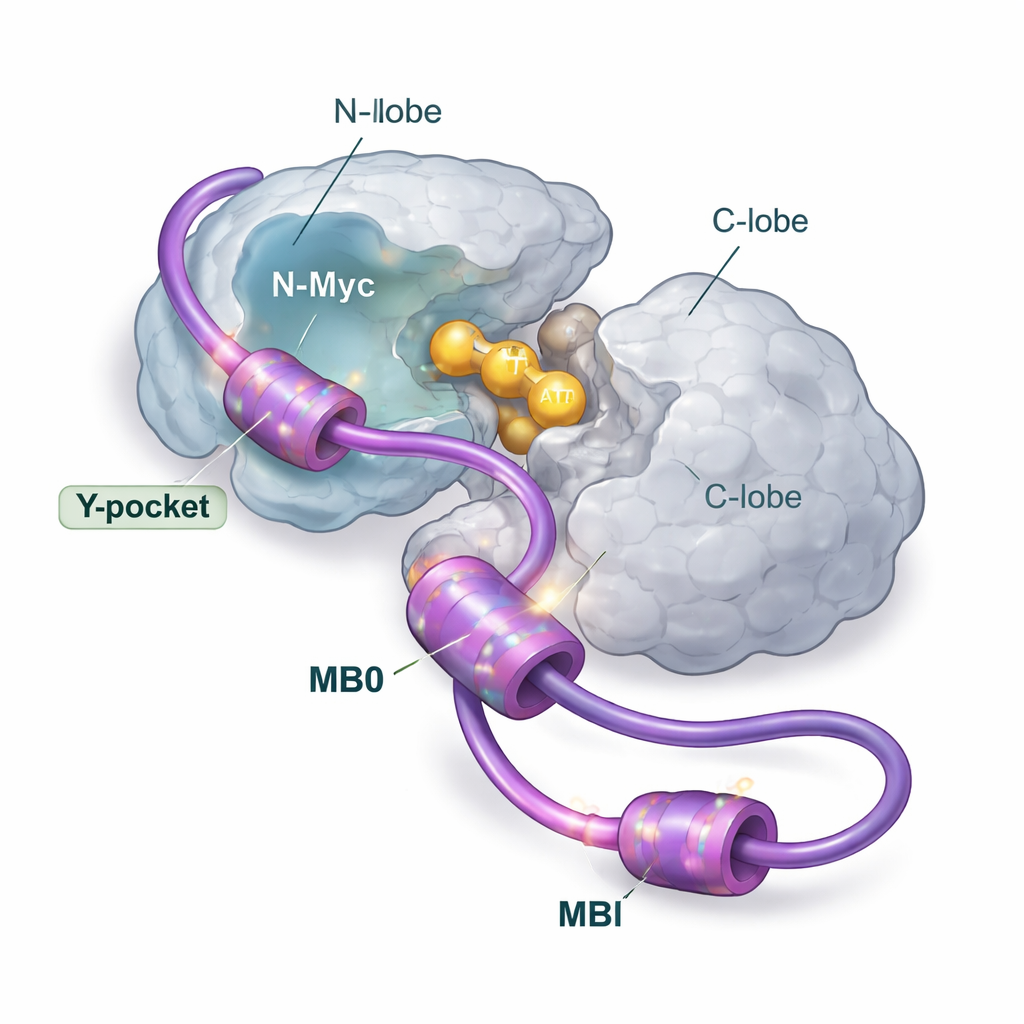
Multiple gripping points, one main landing pad
Using a broad toolkit of structural and biophysical methods, including nuclear magnetic resonance, X-ray scattering, mass spectrometry, calorimetry, and computer modeling, the authors dissect how N-Myc and AurA come together in solution. They focused on N-Myc’s front end, which contains two conserved regions called MB0 and MBI. These short segments are rich in aromatic building blocks (such as phenylalanine, tyrosine, and tryptophan) that tend to stick to greasy pockets on partner proteins. The team shows that N-Myc binds AurA mainly as a 1:1 pair, with MB0 and MBI acting as the primary contact zones. Another N-Myc stretch that had drawn attention in earlier crystal structures turns out to play only a minor role when the full MB0–MBI region is present.
A dynamic handshake that switches the enzyme on
Instead of snapping into a single locked pose, N-Myc remains largely disordered when bound to AurA. The data point to a “fuzzy” complex: several N-Myc segments take turns contacting different patches on the AurA N-lobe, especially a groove known as the Y-pocket and a nearby loop that links signals to the active site. Despite this looseness, the effect is clear—binding of MB0 and MBI stabilizes the AurA N-lobe and increases its kinase activity, meaning it more efficiently transfers phosphate groups to its targets. When the researchers systematically replaced key aromatic residues in MB0 and MBI with alanine, the N-Myc–AurA complex became weaker, the enzyme was less stabilized, and its activity dropped. This shows that specific aromatic “anchors” in the otherwise floppy N-Myc chain are essential for turning AurA up.
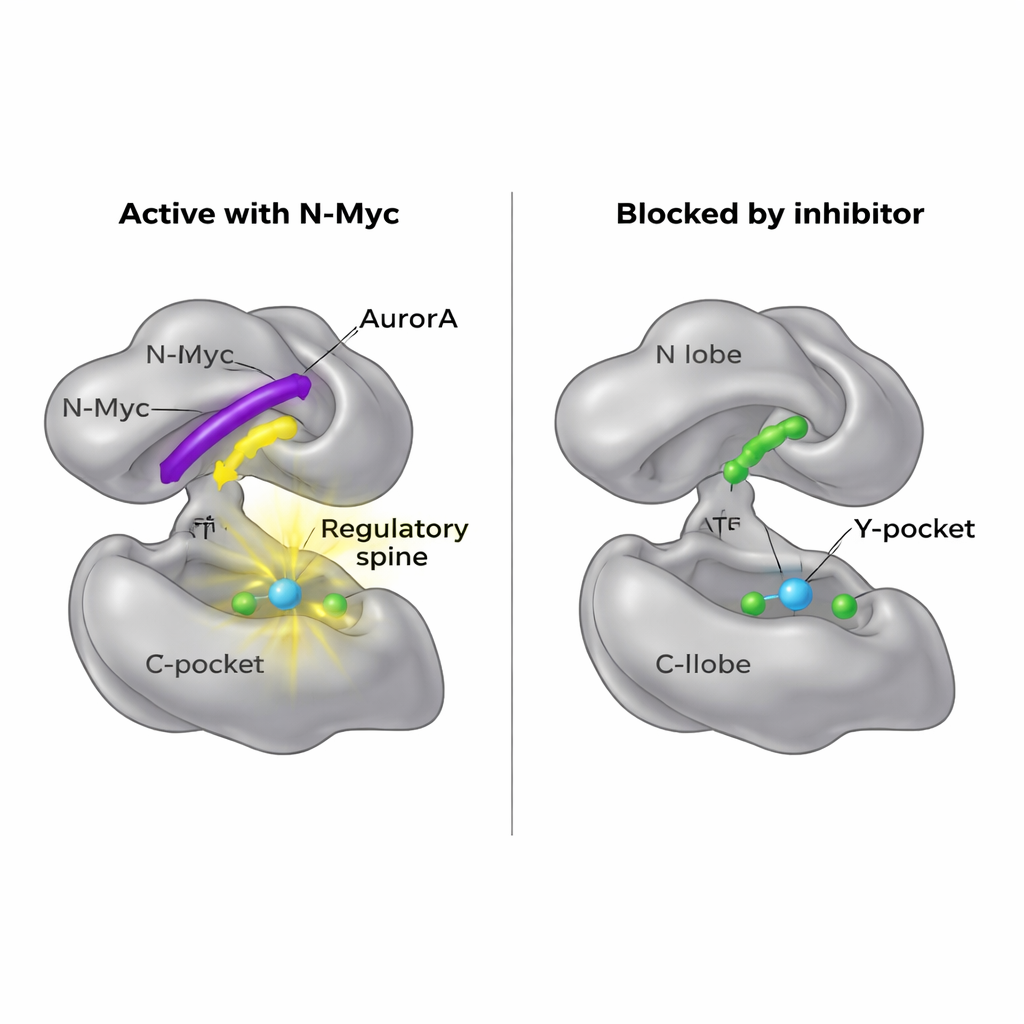
Cracking open a new drug entry point
Because N-Myc itself is hard to drug, the researchers asked whether they could instead block its docking site on AurA. Their structural maps revealed that MB0 and MBI occupy the same general region of the N-lobe that another partner protein, TPX2, uses to activate AurA. A small molecule called AurkinA is already known to wedge into the Y-pocket and displace TPX2. In binding experiments, AurkinA effectively competed with N-Myc for AurA’s N-lobe as well, preventing both N-Myc1–69 and N-Myc1–100 fragments from attaching. This result demonstrates that the N-lobe surface, and especially the Y-pocket, is a druggable hotspot that controls not only classic regulators like TPX2 but also disordered oncoproteins such as N-Myc.
What this means for future therapies
This work reframes the N-Myc–AurA partnership as a dynamic, multi-contact embrace centered on the AurA N-lobe, rather than a single rigid binding site. For non-specialists, the key message is that even a shapeless cancer protein like N-Myc can be tackled indirectly by targeting a solid docking patch on its partner enzyme. By showing that existing N-lobe–binding compounds can push N-Myc away from AurA, the study opens a clear path toward drugs that selectively disrupt this interaction. Such agents could weaken a critical support for N-Myc in high-risk childhood tumors and, with further development, offer a new treatment route for cancers long considered “undruggable.”
Citation: Hultman, J., Morad, V., Tanner, E. et al. The N-Myc MB0-MBI region interacts specifically and dynamically with the N-lobe of Aurora kinase A. Nat Commun 17, 2016 (2026). https://doi.org/10.1038/s41467-026-69725-1
Keywords: N-Myc, Aurora kinase A, intrinsically disordered proteins, neuroblastoma, protein–protein interaction inhibitors