Clear Sky Science · en
Simultaneous promotion of photocatalytic CH4 conversion and H2O2 production via nanopore water confinement
Turning a greenhouse gas into useful liquids
Methane, the main ingredient in natural gas, is both a valuable resource and a powerful greenhouse gas. Converting it gently into liquid chemicals and fuels could help reduce emissions while making everyday products like solvents and disinfectants. This study shows a way to use light, water, and carefully engineered nanoparticles to upgrade methane into useful oxygen‑rich liquids while at the same time producing hydrogen peroxide, a common disinfectant and green oxidant.
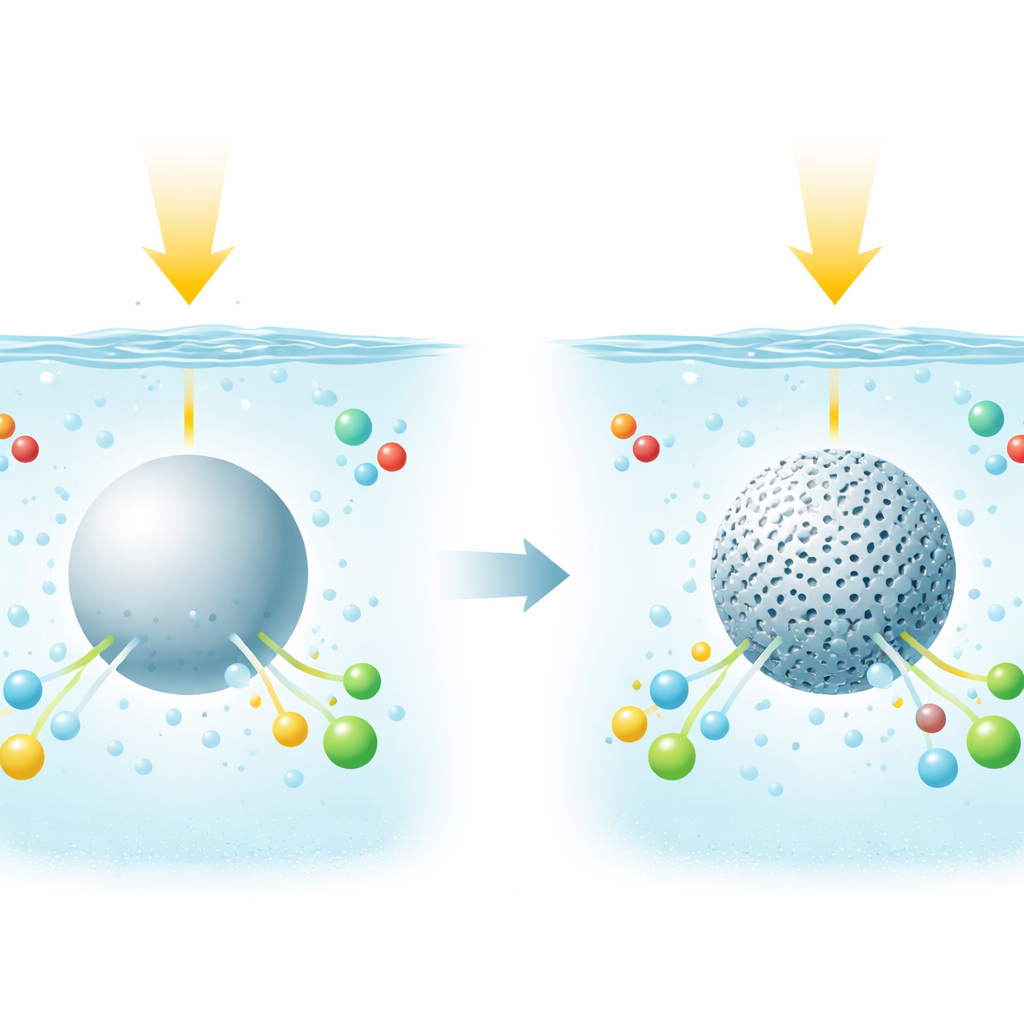
Why water structure matters
Many clean‑energy reactions happen in water, where electrons and protons must move together in tightly choreographed steps. In ordinary liquid water, molecules are held in a constantly shifting hydrogen‑bond network, which quietly governs how easily charges and atoms can move. The authors wondered: what if you could gently squeeze water into tiny spaces so that this network changes? Would that make it easier for light‑excited catalysts to guide methane toward desired products instead of burning it completely to carbon dioxide?
A tiny cage around an active core
To test this idea, the team built core–shell particles. At the center sits a well‑known photocatalyst, titanium dioxide decorated with tiny metal particles such as gold or platinum. Around this core they grew a thin shell of transparent silica pierced with nanometer‑scale pores filled with water. By tuning the pore size down to about 1.7 nanometers—just a few water molecules across—they created a confined water layer hugging the catalyst surface. Importantly, light absorption and basic catalyst properties remained nearly unchanged; what changed was how water sat and moved in these tiny channels.
From methane and oxygen to liquids and peroxide
When these particles were illuminated in water under methane and oxygen, the confined‑water design dramatically improved performance. Compared with the same catalyst without the porous shell, methane conversion roughly tripled and hydrogen peroxide output increased about twenty‑two‑fold. The process produced liquid oxygen‑containing products such as methanol and related molecules with high selectivity, meaning far less wasteful over‑oxidation to carbon dioxide. The effect was robust: it held under different light sources, persisted over many reaction cycles, and could be reproduced using other metals and even other semiconductor cores, showing that the strategy is broadly applicable rather than a one‑off trick.
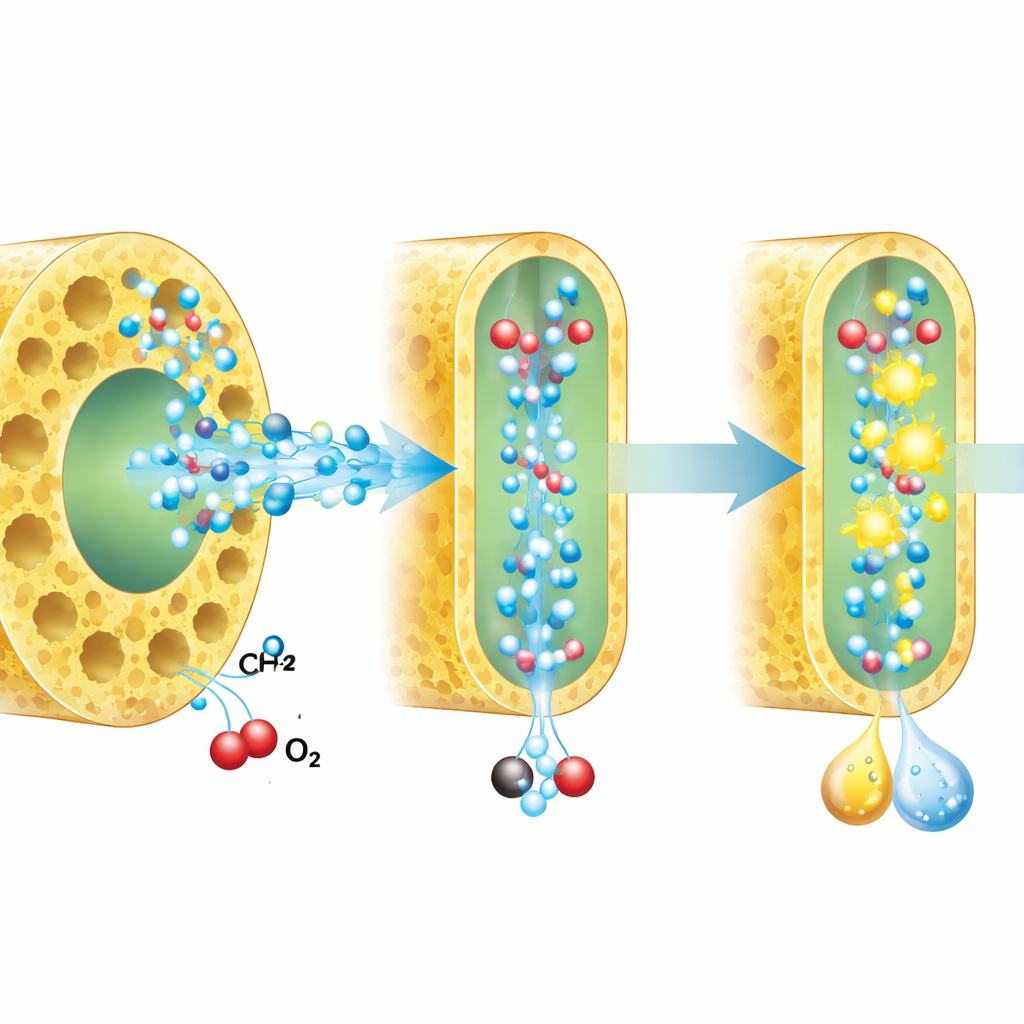
How squeezed water changes the reaction path
To understand why confinement helps, the researchers combined spectroscopic measurements, radical‑trapping experiments, isotope labeling and computer simulations. They found that confined water forms a weaker, more linear hydrogen‑bond network than bulk water. In this altered environment, key reactive species—short‑lived oxygen‑bearing radicals that attack methane—are generated more efficiently and live longer near the catalyst surface. At the same time, the pathway that reduces oxygen is steered toward directly making hydrogen peroxide rather than other, less useful intermediates. Isotope studies, where hydrogen was replaced by deuterium or oxygen atoms were relabeled, confirmed that proton movement becomes more central to the slow, controlling steps of both water oxidation and oxygen reduction once water is confined.
A new handle for cleaner chemistry
In everyday terms, the silica shell acts like a carefully designed sponge that forces water into narrow corridors around the active catalyst, subtly changing how it holds together and how easily protons and electrons can move. This rearranged micro‑environment makes it easier for light‑excited charges to carve methane into valuable liquids and to transform oxygen into hydrogen peroxide, instead of simply burning the fuel. The work suggests that tuning the "feel" of water near solid surfaces—without changing the active material itself—could become a powerful design tool for cleaner chemical processes, from converting greenhouse gases to producing green oxidants and fuels.
Citation: Lv, F., Wei, S., Wu, X. et al. Simultaneous promotion of photocatalytic CH4 conversion and H2O2 production via nanopore water confinement. Nat Commun 17, 2119 (2026). https://doi.org/10.1038/s41467-026-69719-z
Keywords: photocatalytic methane oxidation, nanoporous core-shell catalysts, confined water, hydrogen peroxide production, proton-coupled electron transfer