Clear Sky Science · en
Interface-driven energy-independent charge extraction in GaN photocatalysts
Turning Sunlight into Fuel
Sunlight can, in principle, power the production of clean fuels like hydrogen, but today’s materials waste many of the energized electrons they create. This study looks at a promising semiconductor, gallium nitride (GaN), and shows how adding tiny platinum (Pt) islands to its surface creates a sort of “express lane” for electrons. By guiding charges more efficiently and preventing them from getting stuck at flaws, the researchers greatly improve how well GaN turns light into chemical energy. 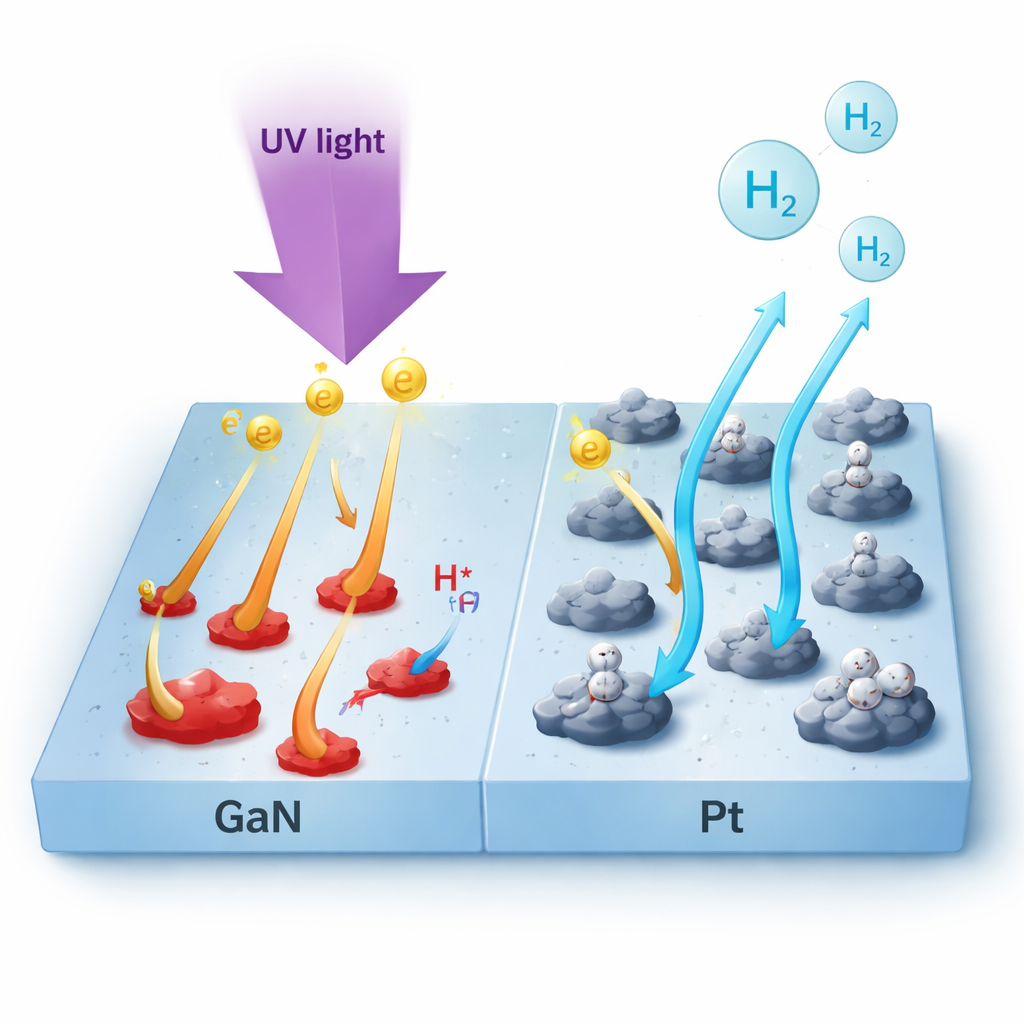
Why Gallium Nitride Matters
GaN is already famous in electronics and LED lighting, and it is also attractive for solar-driven chemistry: its electronic structure can power demanding reactions like splitting water, reducing carbon dioxide, or making hydrogen from ammonia. The challenge is that when sunlight hits GaN, the energized electrons and holes lose their extra energy and often fall into microscopic defects at or near the surface long before they can drive chemistry. Only those charges that keep enough energy and reach the right surface sites in time can help produce fuel. Understanding how electrons move and lose energy in the first trillionths of a second after light absorption is therefore crucial to designing better photocatalysts.
Watching Electrons in Extreme Slow Motion
To follow these ultrafast events, the team used time-resolved two-photon photoemission spectroscopy, a technique that works like an ultrafast camera for electrons. Short laser pulses first excite electrons inside GaN; a second pulse then kicks some of these electrons out of the material so that their energies and arrival times can be measured. By varying the delay between pulses and tuning their colors, the researchers built up a movie of how the electron energy landscape evolves at bare GaN surfaces and at GaN surfaces decorated with Pt nanoislands. This allowed them to separate what happens in the bulk of the crystal, at defects, and across the metal–semiconductor interface. 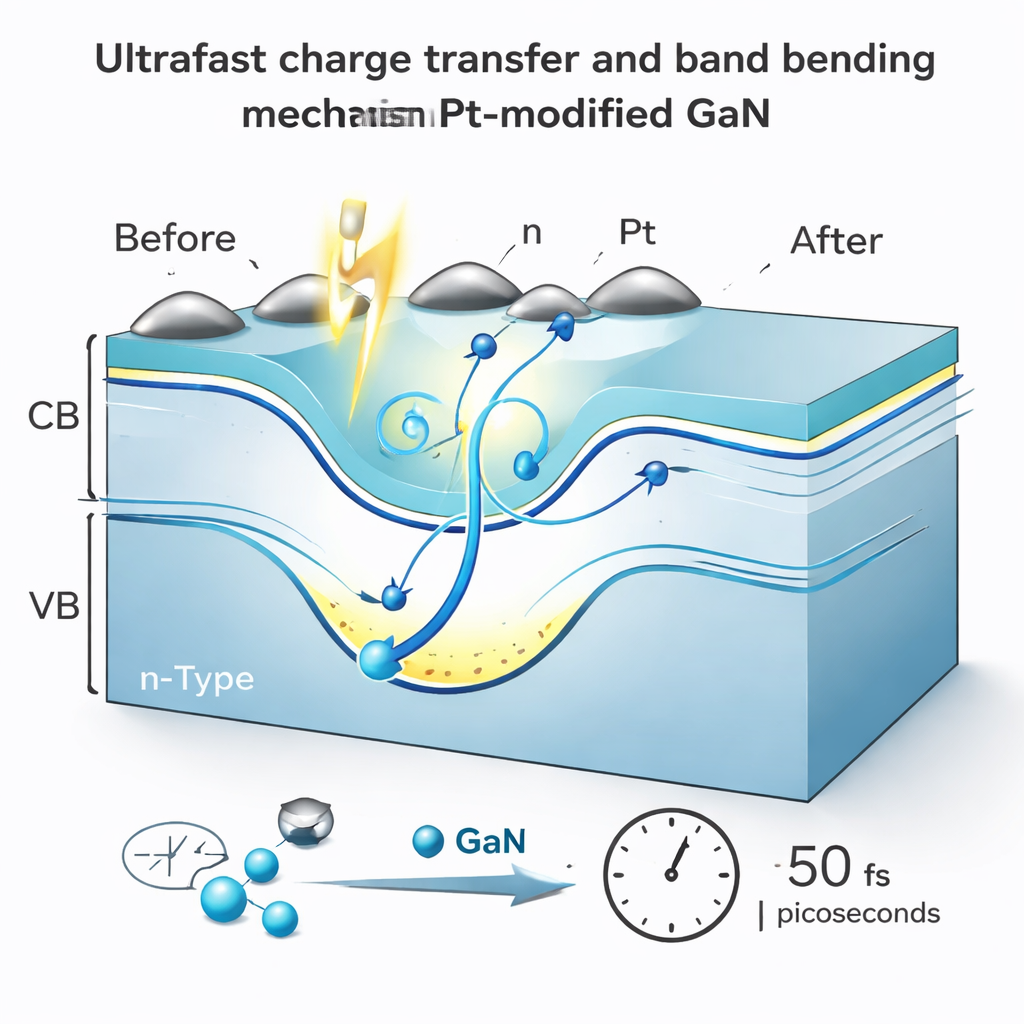
How Platinum Changes the Electron Pathways
On clean GaN, excited electrons quickly slide down to the edge of the conduction band and are then trapped in defect states, many related to missing nitrogen atoms or misplaced magnesium dopants. These traps capture electrons in less than a trillionth of a second and hold them for much longer, effectively removing them from useful chemistry and also disturbing the electric field at the surface. When the surface is covered with ultrathin Pt islands, this behavior changes dramatically. The long-lived defect signal nearly disappears, and instead electrons of many different energies are seen to transfer into Pt within about 50 femtoseconds, with almost no dependence on how energetic they started. In other words, Pt provides a very fast and nearly energy-blind escape route for electrons before they can be lost to traps.
Pulling Electrons from Deep Inside the Crystal
Beyond simply catching surface electrons, Pt also influences how electrons flow from the interior of GaN to its surface. The measurements reveal a slow component attributed to electrons diffusing from the bulk toward the Pt-coated surface over a few trillionths of a second. Because Pt rapidly removes electrons that arrive at the surface, it helps prevent charge buildup there. This, together with a light-induced change in the surface voltage known as surface photovoltage, temporarily flattens the bending of the energy bands near the surface. The result is that electrons find it easier to travel from deeper in the crystal to the surface, increasing the population of useful charges available for reactions by roughly half compared with bare GaN.
From Ultrafast Physics to Better Hydrogen Production
To connect these microscopic dynamics with real-world performance, the authors used Pt-coated GaN as a photocathode to drive hydrogen evolution from water in a neutral salt solution. Compared with bare GaN, the Pt/GaN electrode turned on hydrogen production at much more favorable voltages, produced about 6.6 times higher photocurrent, and maintained stable operation with nearly all photogenerated electrons ending up as hydrogen gas. To a non-specialist, the key message is that carefully engineering the interface between a semiconductor and a metal cocatalyst does more than just cover up defects: it reshapes how and how fast electrons move and lose energy from the very first moments after light absorption, which in turn determines how efficiently sunlight can be converted into chemical fuel.
Citation: Gao, Y., Xie, Y., Höhn, C. et al. Interface-driven energy-independent charge extraction in GaN photocatalysts. Nat Commun 17, 1853 (2026). https://doi.org/10.1038/s41467-026-69683-8
Keywords: gallium nitride, photocatalysis, ultrafast spectroscopy, hydrogen evolution, metal semiconductor interface