Clear Sky Science · en
Efficient methanol upcycling to ethylene glycol and glycolaldehyde via divergent C−C coupling synthesis
Turning a Simple Fuel into Useful Building Blocks
Methanol, a simple alcohol that can be made from carbon dioxide, natural gas, coal, or biomass, is already a key fuel and chemical. This study shows how beams of light and carefully designed nanomaterials can transform methanol into more complex, higher-value molecules—while simultaneously releasing hydrogen gas as a clean energy carrier. The work reveals a new way to steer a single starting material into two different useful products simply by changing how individual metal atoms are arranged on a tiny catalyst surface. 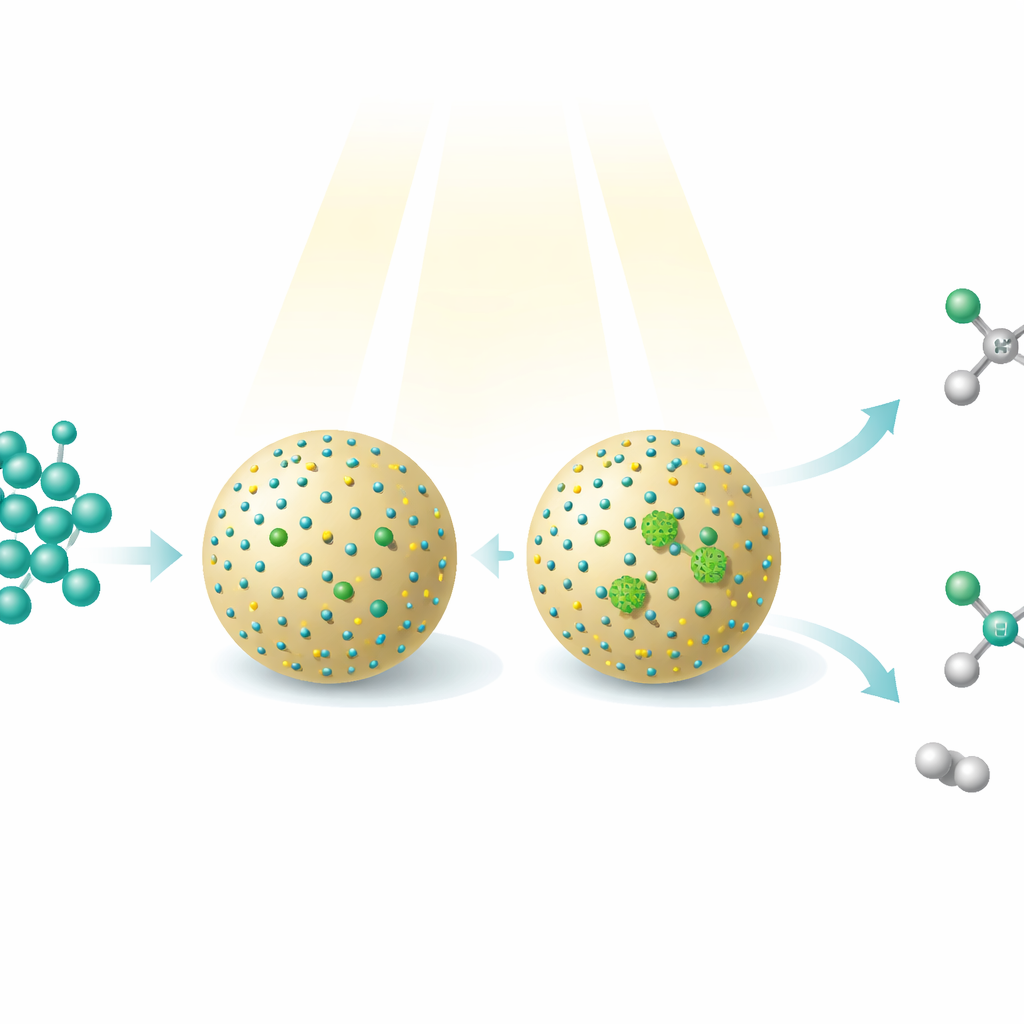
Why Upgrading Methanol Matters
As the world searches for alternatives to oil, methanol has emerged as an attractive liquid that can store energy and carbon. However, most industrial processes that turn methanol into larger molecules are blunt tools: they need high temperatures, depend on fossil-based routes, and often generate many unwanted by-products. Chemists would like to couple methanol molecules together with high precision so they can selectively make just the compounds they want. Doing this under mild conditions using light instead of heat would make it easier to integrate chemical production with renewable energy.
Light-Activated Nanospheres Do the Work
The researchers built a photocatalyst—literally a light-driven catalyst—by decorating tiny cadmium sulfide quantum dots onto spherical particles of silica. These quantum dots absorb light and create energetic electrons and holes that can tear hydrogen atoms off methanol, forming highly reactive fragments. Nickel atoms were then deposited onto the quantum dots in two distinct ways. In one material, the nickel existed mostly as isolated single atoms; in the other, the nickel formed very small clusters of several atoms. Although the overall composition barely changed, this subtle difference in nickel arrangement dramatically altered which reactions occurred on the surface.
Two Nickel Designs, Two Clean Products
When the catalyst with single nickel atoms was exposed to light in methanol, it favored pairing up two identical methanol-derived fragments, leading mainly to ethylene glycol—a two-carbon diol widely used in antifreeze and plastics. This route reached about 90% selectivity, meaning almost all the converted methanol ended up as this one product, while hydrogen gas was produced in matching amounts from the removed hydrogen atoms. By contrast, the catalyst with nickel clusters channeled the same methanol fragments along a different path. Here, one portion of methanol was further oxidized to a short-lived formaldehyde-like intermediate, which then combined with another fragment to form glycolaldehyde, another two-carbon compound with many uses in fine chemicals and potentially in bio-based processes. This pathway delivered glycolaldehyde with 96% selectivity, again alongside hydrogen. The silica support helped the quantum dots harvest light more efficiently and made the particles more robust over many cycles of use. 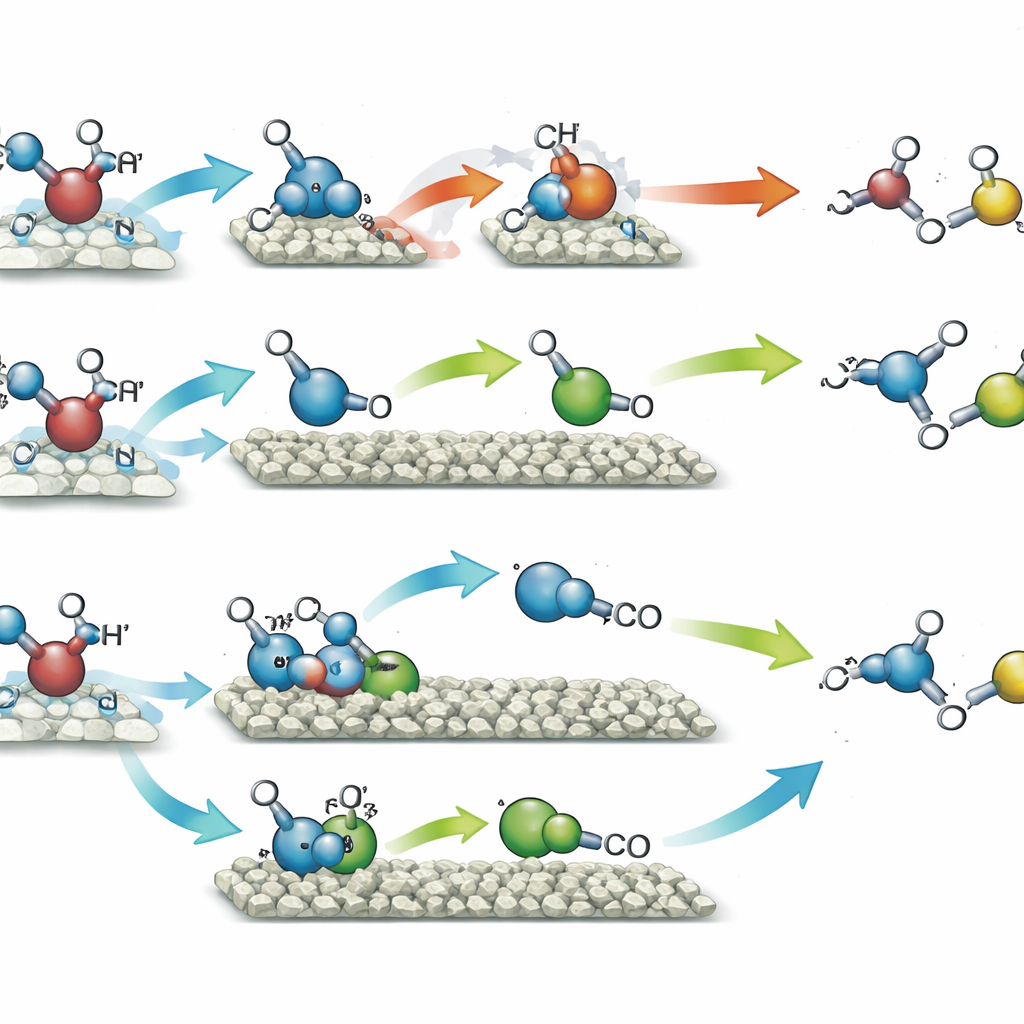
Peeking Under the Hood of the Reaction
To understand why the two nickel arrangements behaved so differently, the team combined a suite of advanced measurements with computer simulations. Electron paramagnetic resonance experiments showed that both catalysts generated methanol-based radicals under light, but the cluster catalyst also promoted breakage of the oxygen–hydrogen bond, leading to more diverse reaction fragments and to the formaldehyde intermediate. Time-resolved tests confirmed that this intermediate rose and then fell in concentration as it was consumed to make glycolaldehyde. Quantum-chemical calculations mapped out the energy costs of each tiny step. On single nickel atoms, directly coupling two identical radicals to form ethylene glycol was the easiest route. On nickel clusters, that same coupling bound the product too tightly, making its release difficult, whereas the pathway that first formed the asymmetric intermediate and then glycolaldehyde became energetically preferred.
A New Lever for Cleaner Chemical Making
In everyday terms, this work shows that "where" each nickel atom sits on a nanometer-scale surface can decide "what" molecule methanol becomes when illuminated by light. By switching between single atoms and small clusters of nickel, the researchers can flip the main product from ethylene glycol to glycolaldehyde, both made efficiently and cleanly along with hydrogen fuel. This strategy of architecting catalysts atom by atom offers a promising route to upgrade simple, renewable feedstocks like methanol into a range of useful chemicals without relying on petroleum, harsh conditions, or wasteful side reactions.
Citation: Qi, MY., Tan, CL., Tang, ZR. et al. Efficient methanol upcycling to ethylene glycol and glycolaldehyde via divergent C−C coupling synthesis. Nat Commun 17, 2835 (2026). https://doi.org/10.1038/s41467-026-69656-x
Keywords: methanol upcycling, photocatalysis, nickel single atoms, ethylene glycol, glycolaldehyde