Clear Sky Science · en
LOAd703-induced tumor microenvironment gene engineering in combination with atezolizumab in metastatic malignant melanoma: a phase I/II trial
Why this new melanoma study matters
For people living with advanced skin melanoma that no longer responds to today’s powerful immunotherapies, treatment options are limited and often harsh. This study tests a different idea: using a gene‑engineered virus injected directly into tumors, together with an existing immune‑boosting drug, to wake up the body’s defenses and regain control over cancer. The early results suggest this approach can be given safely and may help a meaningful share of patients whose disease had previously resisted standard care.
A stubborn skin cancer that outsmarts treatment
Melanoma is one of the most common serious skin cancers in Western countries, and its incidence has risen sharply over recent decades. Modern immune checkpoint drugs—which take the brakes off T cells—have transformed outcomes, lifting five‑year survival in advanced melanoma to about half of patients. Yet many tumors eventually stop responding. Often, these resistant cancers are surrounded by a “cold” neighborhood of cells and molecules that keep immune cells out or shut them down. Finding ways to rewire this tumor microenvironment so that T cells can once again recognize and attack the cancer is a major research priority.
A virus designed to rewire the tumor’s neighborhood
The treatment tested here, called LOAd703, is a modified adenovirus—a type of common cold virus—engineered to do two jobs. First, it prefers to replicate in and destroy tumor cells, a strategy known as oncolytic virotherapy. Second, it carries genetic instructions for two powerful immune‑stimulating signals placed on infected cells. These signals, CD40L and 4‑1BBL, help professional immune sentinels (dendritic cells) mature and present tumor fragments, and they strengthen and expand cancer‑fighting T cells and natural killer cells. Patients in the trial received LOAd703 injected directly into one or more tumors every three weeks, along with atezolizumab, an established antibody drug given by vein that blocks the PD‑L1 brake on T cells. The hope was that the virus would turn tumors into in‑body vaccine factories, while the antibody kept reinvigorated T cells active.
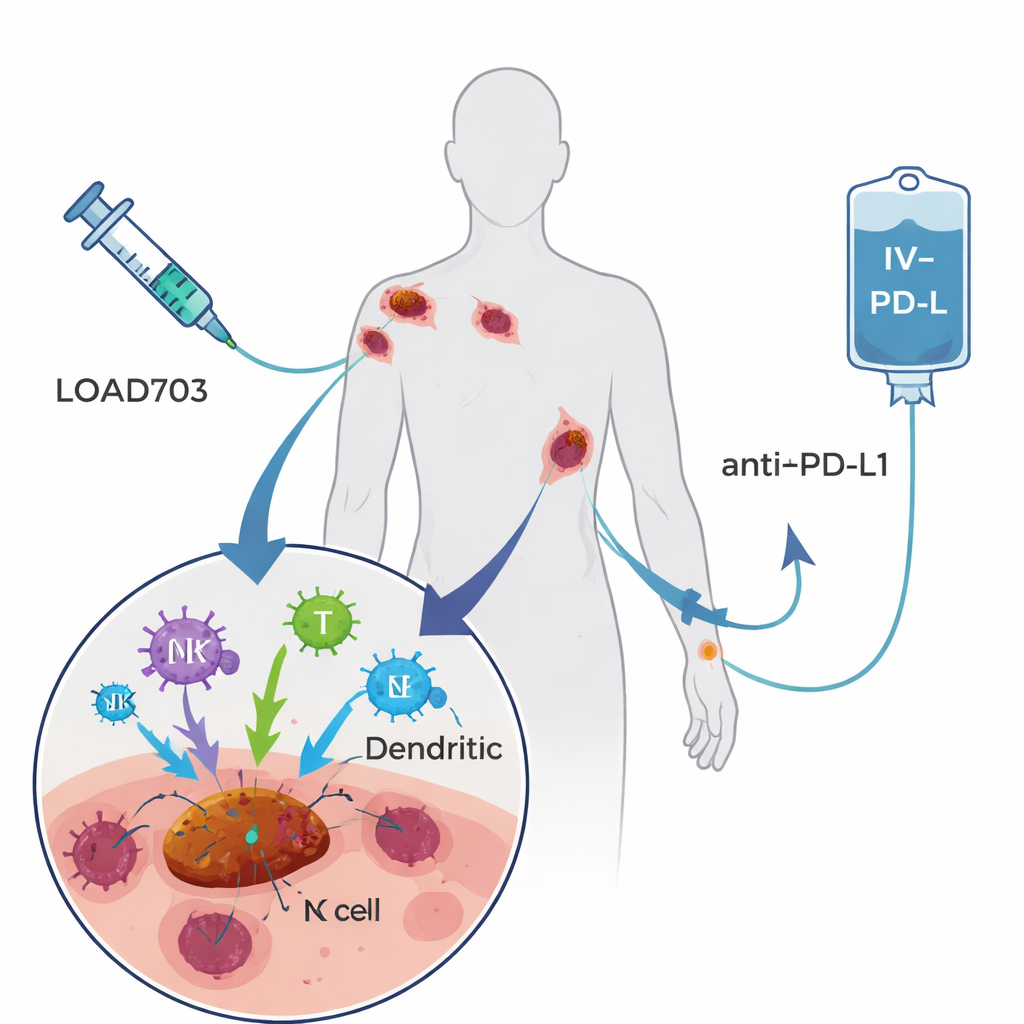
Who was treated and how they fared
The phase I/II trial enrolled 24 people with stage IV melanoma that had clearly progressed despite at least one prior PD‑1–blocking treatment; many had received several lines of immunotherapy and targeted drugs. Two virus dose levels were tested. Overall, the combination was well tolerated. The most common side effects linked to the virus—fever, chills, nausea and flu‑like symptoms—were mostly mild and temporary. Only two patients had more serious but reversible reactions at the higher dose, and no one had to stop treatment because of virus‑related problems. When researchers looked at tumor responses, four patients (17%) had measurable shrinkage of their cancers, and over half had at least stable disease for a time. At two years after entering the study, nearly half of the participants were still alive, including some whose scans showed long‑lasting control or complete clearing of detectable disease after finishing therapy.
Signals that the immune system was re‑engaged
To see what was happening inside tumors and in the bloodstream, the team analyzed tissue biopsies and blood samples taken before treatment and about nine weeks after starting. In the injected tumors, they observed higher activity of genes linked to type 1 immune responses, including markers of activated T cells and natural killer cells, and molecules that help these cells enter tumors from nearby blood vessels. Signals associated with antigen presentation—the process by which tumor fragments are displayed to T cells—also increased, suggesting that the viral therapy was turning the tumor microenvironment from suppressive to more immune‑friendly. In blood, several immune‑related proteins, including PD‑L1 itself, rose after therapy, consistent with a broad activation of immune pathways. Patients who survived longer tended to show gene patterns tied to robust T cell function and antiviral protection, while those with shorter survival showed patterns associated with suppressive cell types and tissue‑remodeling signals.
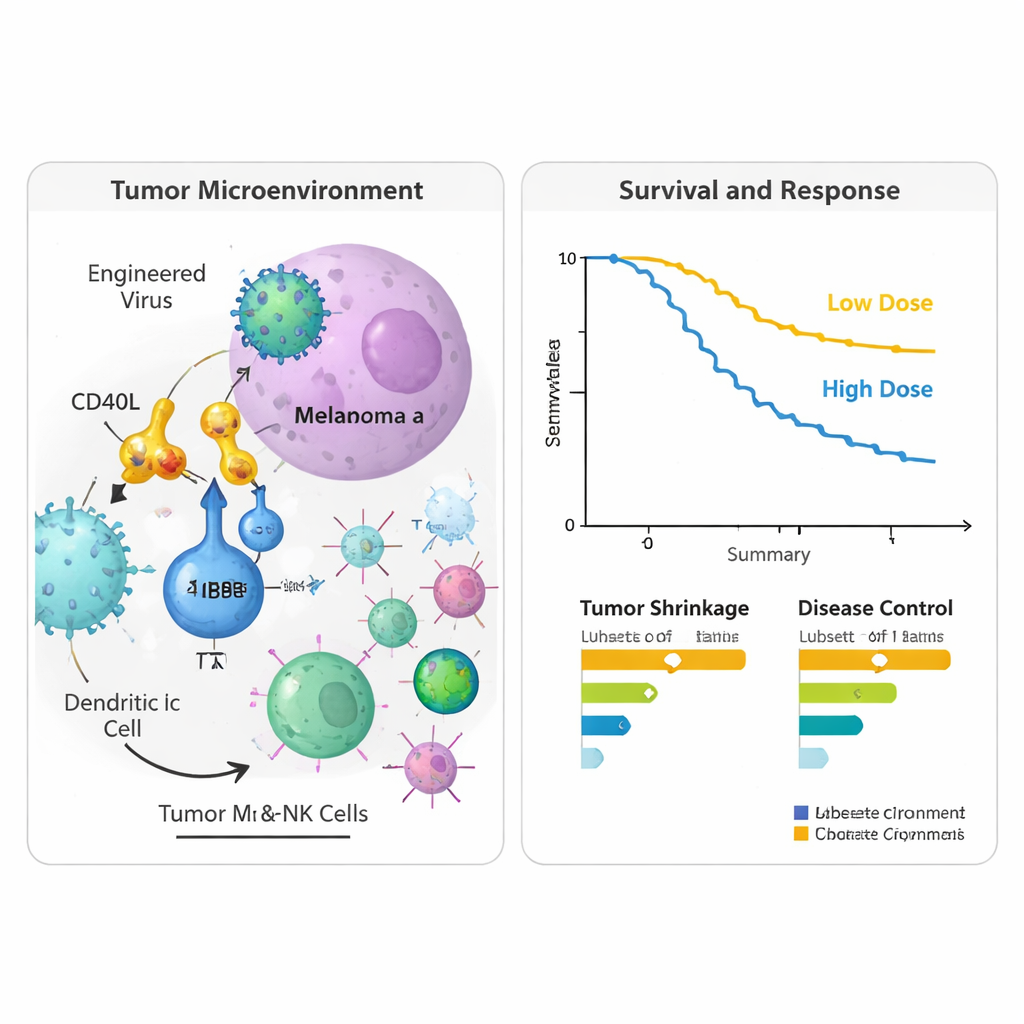
What this could mean for future care
This was a small, single‑arm study without a comparison group, so its survival and response figures must be interpreted cautiously. Still, the combination of good tolerability, immune activation in tumors, and encouraging disease control in heavily pretreated patients hints that gene‑engineered oncolytic viruses like LOAd703 could help re‑sensitize melanoma to checkpoint blockade—or even hold tumors in check on their own. The results support larger, controlled trials, potentially pairing the virus with additional immune‑targeting drugs, to clarify which patients benefit most and how best to integrate this strategy into melanoma treatment.
Citation: Hamid, O., Ekström-Rydén, V., Mehmi, I. et al. LOAd703-induced tumor microenvironment gene engineering in combination with atezolizumab in metastatic malignant melanoma: a phase I/II trial. Nat Commun 17, 1760 (2026). https://doi.org/10.1038/s41467-026-69629-0
Keywords: melanoma, immunotherapy, oncolytic virus, tumor microenvironment, checkpoint inhibitor resistance