Clear Sky Science · en
Drug-caged drugs enable photocatalytic dual decaging of nitric oxide and anesthetics for antibacterial analgesia
Fighting germs and pain at the same time
Bacterial infections do not just threaten our health; they also hurt—a lot. Standard treatments usually target the germs but leave patients relying on separate painkillers, which may wear off quickly or cause side effects. This study introduces a light-controlled medicine that aims to kill bacteria and ease pain in one coordinated step, offering a glimpse of smarter, more comfortable infection care.
A two-in-one medicine idea
The researchers built their approach around tetracaine, a familiar local anesthetic used to numb skin and nerves. They chemically attached nitric oxide, a tiny gas molecule known for its strong antibacterial and anti-inflammatory actions, to tetracaine. This created a “prodrug” called TTC-NO that is largely inactive until triggered. TTC-NO was then packed, together with a light-sensitive helper molecule, into tiny polymer bubbles called micelles that can travel in watery environments such as blood or tissue fluid. 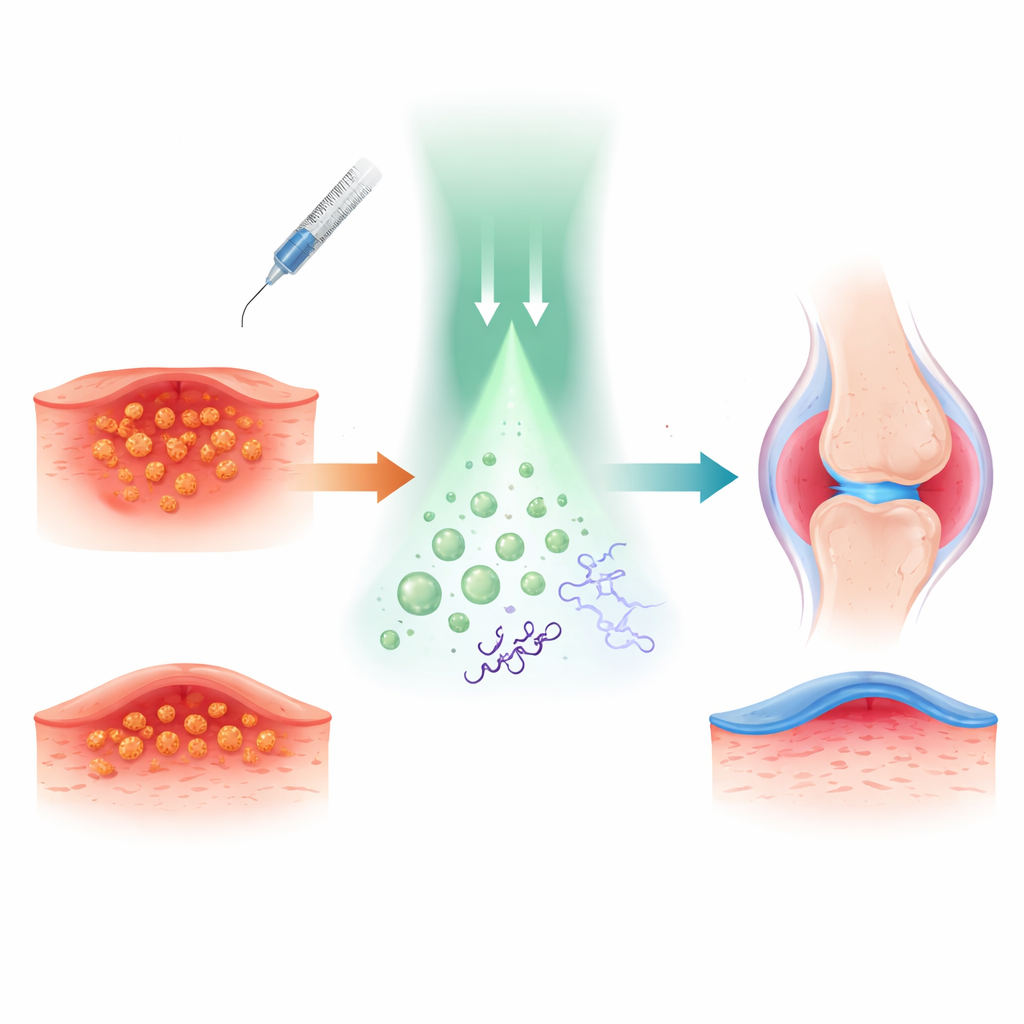
Turning on the medicine with gentle light
To understand how this light switch works, the team studied the chemistry in solution. They showed that a photocatalyst based on iridium can transfer an electron to TTC-NO when illuminated with green light. This weakens a specific bond, causing nitric oxide to break free and restoring tetracaine to its active form. Measurements using spectroscopy and chromatography confirmed that TTC-NO is almost completely converted to tetracaine and nitric oxide under these conditions, while staying essentially unchanged in the dark. When the two components were encapsulated in micelles, the particles remained stable, about 80 nanometers across, and only released their contents when irradiated, proving that the “drug-caged drug” concept can be controlled from outside the body with light.
Helping infected wounds heal faster
The team next moved to mice with skin wounds deliberately infected with methicillin-resistant Staphylococcus aureus (MRSA), a problematic hospital bug. Some wounds received standard treatments such as an antibiotic or growth factor, while others were treated with the TTC-NO micelles, with or without light. When the light was turned on, the TTC-NO system sharply reduced bacterial counts, calmed inflammation, encouraged new blood vessel growth, and boosted collagen deposition, all of which are essential for proper repair. Wounds treated in this way closed faster and more completely than those given tetracaine alone or micelles kept in the dark, and their performance was comparable to a frontline antibiotic. Importantly, safety checks showed no major damage to organs or blood chemistry.
Relieving pain and quieting nerve signals
Because infection and tissue injury strongly activate pain pathways, the scientists carefully measured pain-related behaviors in the same wound model. Traditional tetracaine briefly raised the force required to trigger a pain response but wore off within a few hours. By contrast, TTC-NO micelles activated by light provided relief for up to 12 hours. Microscopic studies of nerve clusters near the spinal cord and activity recordings in the brain’s sensory cortex revealed that the combined treatment dampened the firing of pain-sensing neurons long after pure tetracaine had lost its effect. Further experiments suggested that the extra duration comes from lowering the bacterial load and inflammation with nitric oxide, not just from numbing the nerves.
Protecting painful infected joints
The researchers also tested the system in mice with septic arthritis, a severe MRSA infection inside the knee joint that causes swelling, bone loss, and intense pain. Light-activated TTC-NO micelles reduced joint width, cut bacterial numbers, and protected bone structure as seen by high-resolution scans. Levels of harmful inflammatory molecules in the blood dropped, while protective signaling molecules rose. Behaviorally, treated mice could tolerate stronger mechanical pressure on the affected paw and moved more freely in an open arena, again showing longer-lasting relief than tetracaine alone. Nerve and brain measurements mirrored these behavioral gains, indicating broad dampening of pain pathways without obvious local or systemic toxicity. 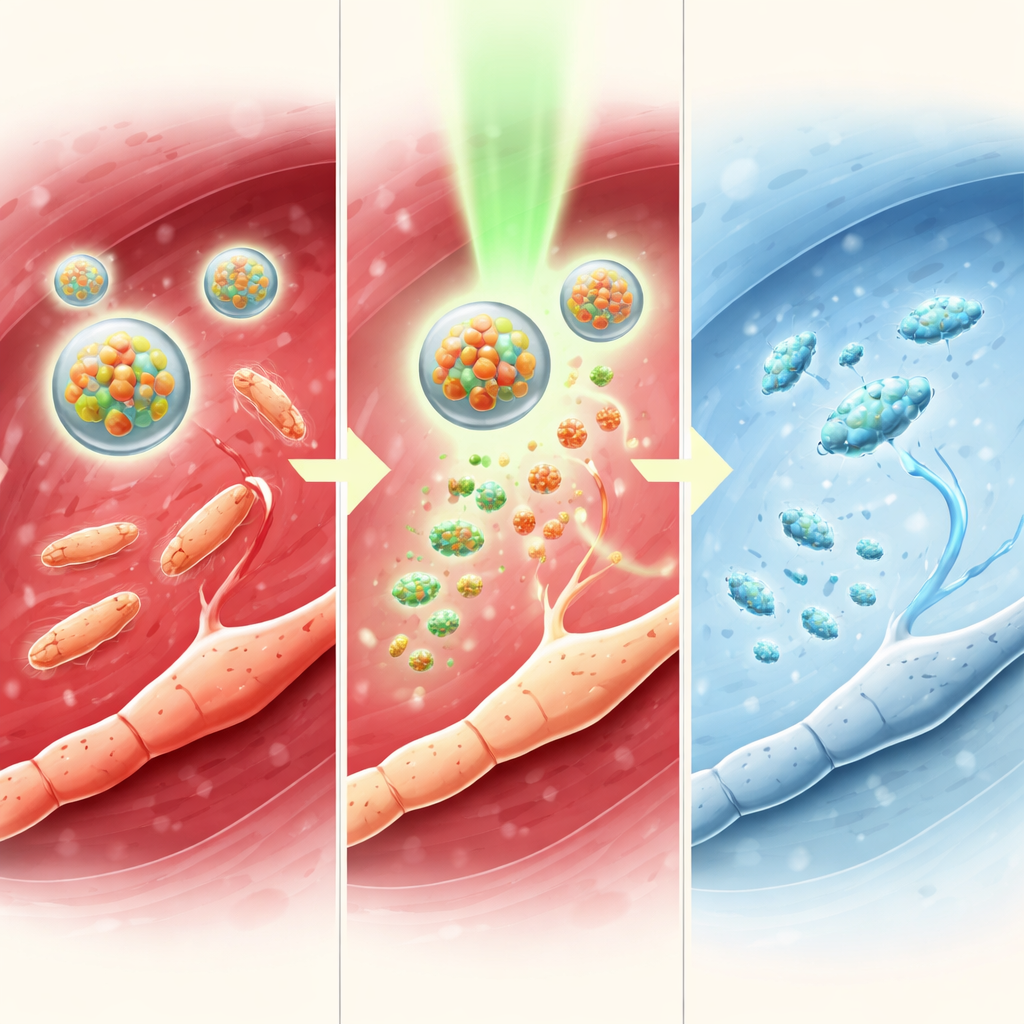
What this could mean for patients
Taken together, the work demonstrates a single, light-activated nanosystem that can both clear stubborn bacterial infections and significantly reduce associated pain. By chemically “caging” an anesthetic inside a nitric-oxide–releasing structure and packaging it into micelles, the authors achieve on-demand, localized release of two helpful agents using mild visible light. In animal models of skin wounds and joint infections, this design speeds healing and extends pain relief beyond what a conventional local anesthetic can offer. While much testing remains before human use, the study highlights how smart, switchable medicines may one day make infection treatments both more effective and more comfortable.
Citation: Zhang, J., Gan, G., Cao, C. et al. Drug-caged drugs enable photocatalytic dual decaging of nitric oxide and anesthetics for antibacterial analgesia. Nat Commun 17, 2843 (2026). https://doi.org/10.1038/s41467-026-69624-5
Keywords: nitric oxide therapy, light-activated drugs, antibacterial analgesia, nanomedicine, MRSA infections