Clear Sky Science · en
T cells engineered against Dickkopf-1-A2 complex can be used to treat HLA-A2+ solid and hematologic cancers
New Hope for Tough-to-Treat Cancers
Many powerful new immunotherapies work well for blood cancers but have stumbled against solid tumors like pancreatic, lung, and breast cancers. This study introduces a specially engineered T cell that homes in on a molecular “flag” found on a wide range of cancers but largely absent from normal tissues, offering the possibility of one treatment that could safely target both blood and solid tumors in patients who carry a common immune type.
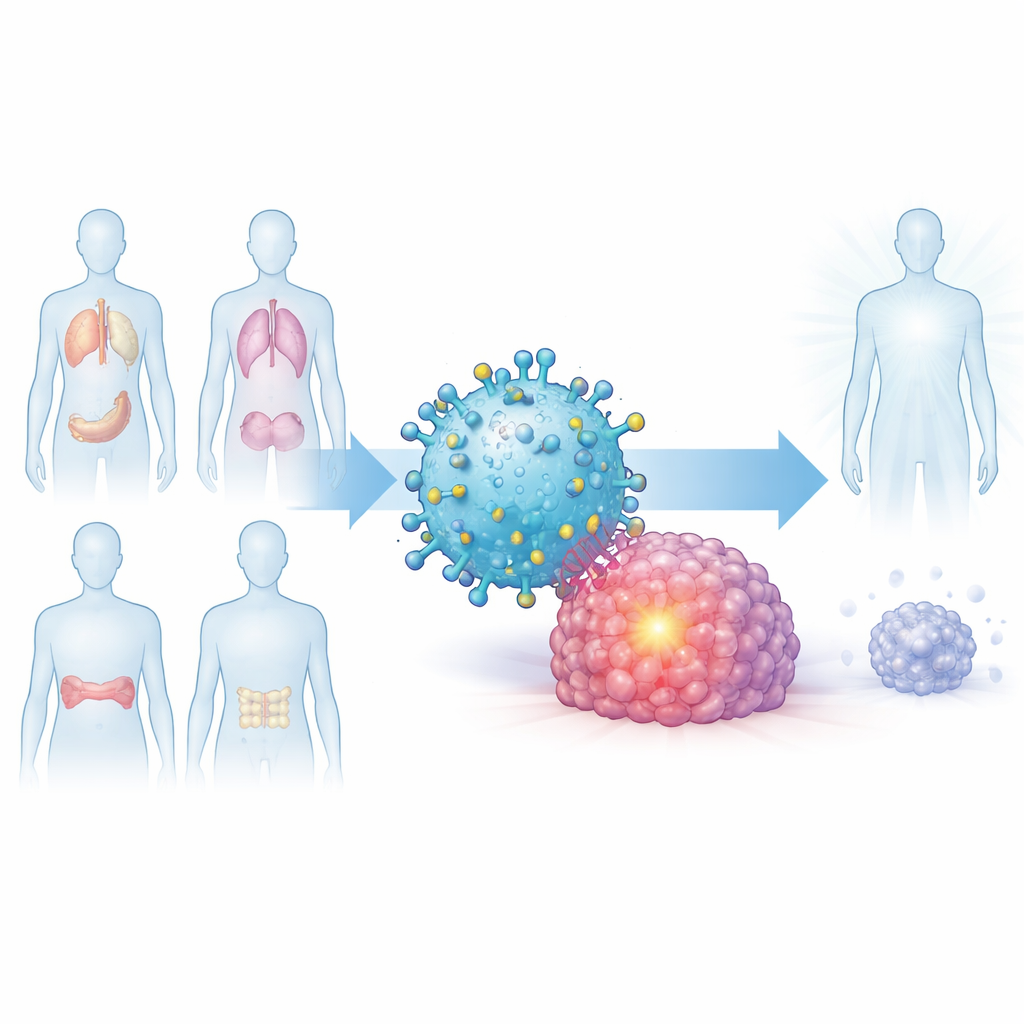
A Common Signal Shared by Many Tumors
The researchers began by asking whether there is a shared molecular feature that appears across different cancers but rarely in healthy organs. They focused on a protein called Dickkopf-1, or DKK1, which helps regulate cell growth pathways and is found at high levels in multiple myeloma, pancreatic cancer, lung cancer, and aggressive triple-negative breast cancer. By mining large public gene databases and examining tumor samples, they showed that DKK1 is much more abundant in many tumors than in matching normal tissues, and that patients whose tumors make more DKK1 often have worse survival.
Turning a Tumor Flag into a Precise Target
Because DKK1 normally floats outside cells, it is not an obvious handle for T cells, which recognize short protein snippets presented on cell surfaces by molecules called HLA. Earlier work had identified a short DKK1 fragment that is displayed by a very common HLA type known as HLA-A2. The team had already created an antibody, called C2, that recognizes this DKK1 fragment when it is held by HLA-A2 on the tumor cell surface, but not on most healthy cells. In the current study, they built a chimeric antigen receptor (CAR) using the recognition part of this antibody, fusing it to internal T cell signaling components so that ordinary T cells can be reprogrammed to recognize the DKK1–HLA-A2 combination, referred to as the DKK1-A2 complex.
Engineered T Cells That Seek and Destroy Tumors
The scientists then tested these DKK1-A2 CAR-T cells against a broad panel of human cancer cell lines in the lab, including multiple myeloma, pancreatic, lung, and triple-negative breast cancer cells. Whenever tumor cells displayed both DKK1 and HLA-A2, the engineered T cells multiplied vigorously, released potent immune molecules, and efficiently killed the cancer cells. When DKK1 was removed from the tumor cells, or when cells lacked HLA-A2, the CAR-T cells no longer attacked, demonstrating that their activity is highly specific to the intended target.
Proving Effectiveness in Living Models
To see whether this approach could shrink established cancers in living organisms, the team used several mouse models carrying human tumors. DKK1-A2 CAR-T cells controlled or eliminated multiple myeloma growing in the bone marrow, pancreatic tumors implanted in the pancreas, lung cancer that had spread through the bloodstream, and aggressive breast cancers, whereas standard CD19-targeted CAR-T cells used as a control had little effect. The therapy also worked against tumors grown from real patient pancreatic cancers transplanted into mice, more closely mimicking human disease. A humanized version of the CAR, designed to be suitable for clinical use, showed even better long-term control, partly because these T cells adopted a memory-like state that allowed them to persist and respond again when tumors reappeared.
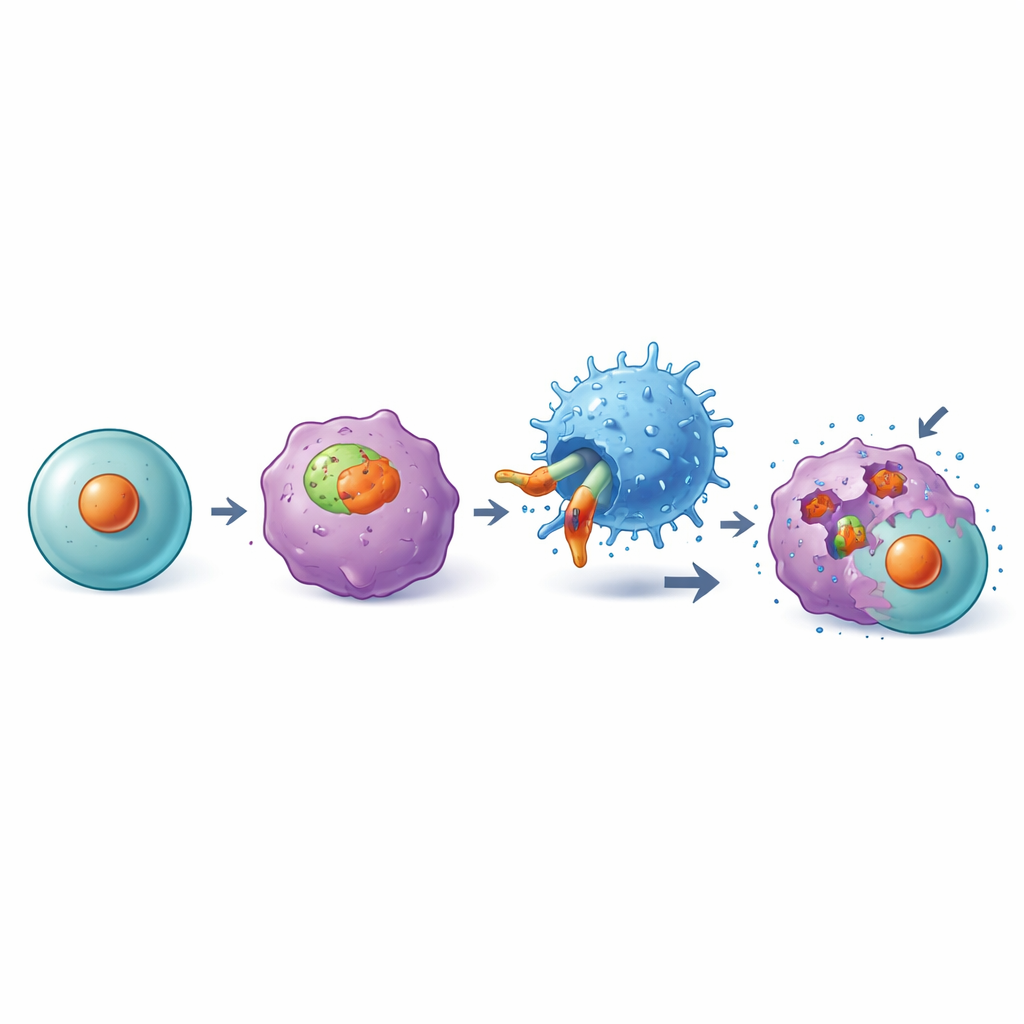
Safety Checks on Healthy Blood and Organs
Any targeted therapy must spare healthy tissues. The authors therefore stained dozens of types of normal human tissues and found that, aside from tonsils, the DKK1-A2 complex was essentially absent. In laboratory tests, the DKK1-A2 CAR-T cells did not damage blood or bone marrow cells from healthy HLA-A2–positive donors, in contrast to CD19 CAR-T cells, which predictably destroyed normal B cells. In mouse models engineered to express human DKK1 and HLA-A2, the new CAR-T cells did not deplete key blood cell populations or platelets and caused fewer signs of dangerous cytokine release than some existing CAR designs, supporting a favorable safety profile.
What This Could Mean for Patients
Taken together, the findings suggest that the DKK1-A2 complex is a promising “bull’s-eye” that appears on many hard-to-treat cancers but largely stays off normal tissues. T cells armed with the DKK1-A2 CAR were able to recognize and destroy diverse tumors in preclinical models while showing limited toxicity. If these results translate to humans, this approach could offer a new, shared immunotherapy for patients with HLA-A2–positive multiple myeloma, pancreatic, lung, and certain breast cancers, expanding the reach of CAR-T treatment beyond blood cancers into the realm of solid tumors.
Citation: Zhang, Y., Xiong, W., Qian, J. et al. T cells engineered against Dickkopf-1-A2 complex can be used to treat HLA-A2+ solid and hematologic cancers. Nat Commun 17, 2818 (2026). https://doi.org/10.1038/s41467-026-69621-8
Keywords: CAR-T cell therapy, DKK1, pancreatic cancer, multiple myeloma, solid tumor immunotherapy