Clear Sky Science · en
Isolable radical cation and dication of dialumene
Why this aluminum story matters
Chemists are always searching for cheaper, more abundant metals that can do the sophisticated jobs now handled by precious elements like palladium or platinum. This study shows that aluminum – a metal better known from soda cans and aircraft – can be coaxed into unusually reactive forms that behave a bit like tiny electronic switches. Understanding and controlling these exotic aluminum species could eventually help build new catalysts for cleaner chemical processes and materials.
Turning a familiar bond into an electronic switch
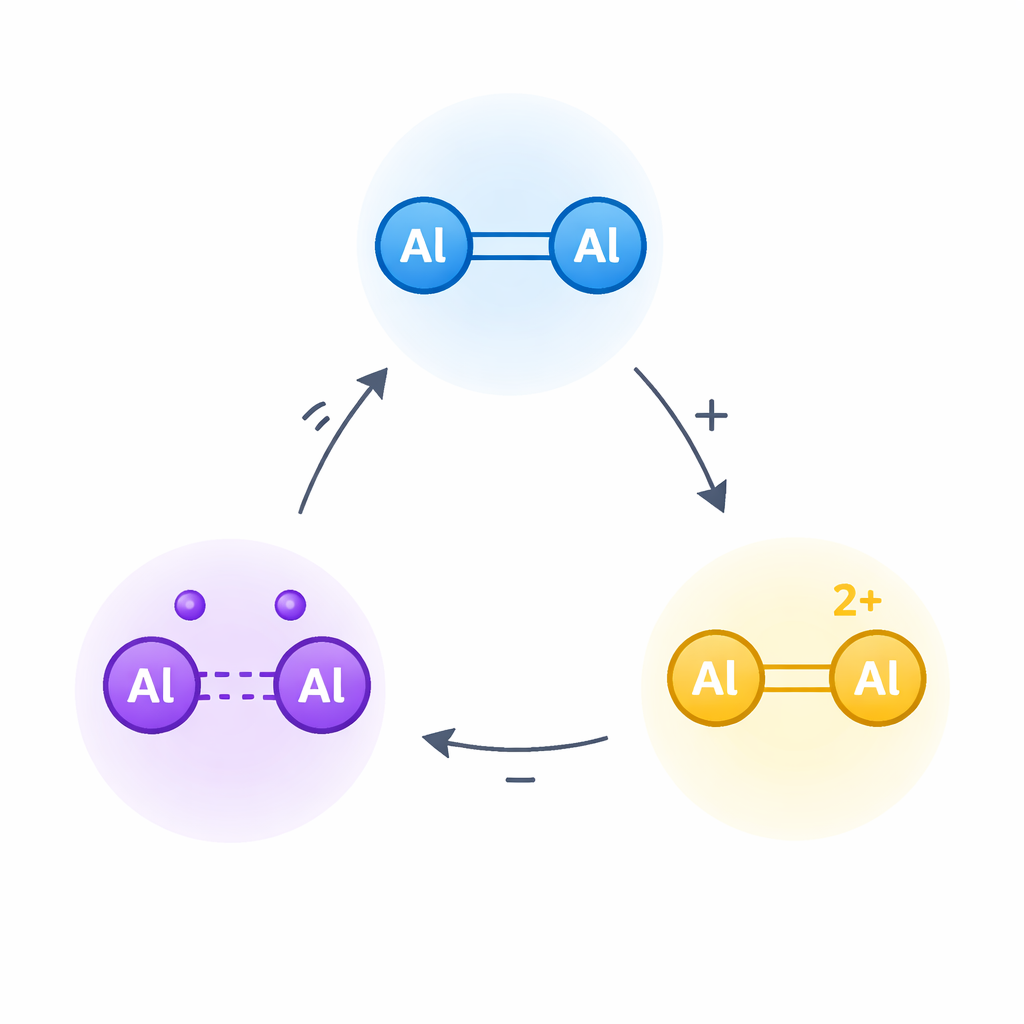
The work centers on dialumene, a molecule in which two aluminum atoms share a double bond, somewhat like the carbon–carbon double bond in common alkenes. In organic molecules, such double bonds can be oxidized stepwise to give radical cations and then dications – highly charged, highly reactive forms that underpin many reactions in electrochemistry and materials science. The authors asked whether the aluminum–aluminum double bond in dialumene could be pushed through a similar two-step oxidation, even though aluminum is strongly electron-hungry and such charged species should be exceptionally unstable.
Designing a protective molecular framework
To tame this reactivity, the team built a dialumene surrounded by bulky silicon-based groups and strong electron-donating “carbene” ligands. These act like padded armor and electronic cushions: they shield the delicate aluminum–aluminum core from unwanted reactions and help supply electron density where it is needed. Starting from this neutral dialumene, labeled 1, they used carefully chosen oxidizing agents to remove first one electron, then a second, generating an aluminum-centered radical cation (2) and then a dication (3). X-ray crystallography provided snapshots of all three states, revealing how the aluminum–aluminum bond gradually lengthens and changes character as electrons are stripped away.
Proving a true three-way redox cycle
Key measurements confirmed that the radical cation really carries an unpaired electron centered between the two aluminum atoms, not on the surrounding ligands. Electron paramagnetic resonance spectroscopy showed a clear signal consistent with a single electron shared by both aluminum centers. Computational studies supported this picture, indicating that the remaining bonding electron occupies a largely aluminum–aluminum orbital. When the second electron is removed to make the dication, the bond weakens toward a single bond and positive charge piles up on the aluminum pair. Importantly, the researchers showed that all of these changes are reversible: adding electrons back with a reductant regenerates first the radical cation and then the original dialumene, and mixing neutral and doubly charged forms gives the radical cation through a comproportionation process. Together, these reactions demonstrate a robust, isolable three-state redox cycle on a simple aluminum–aluminum unit.
Making aluminum act like a transition metal
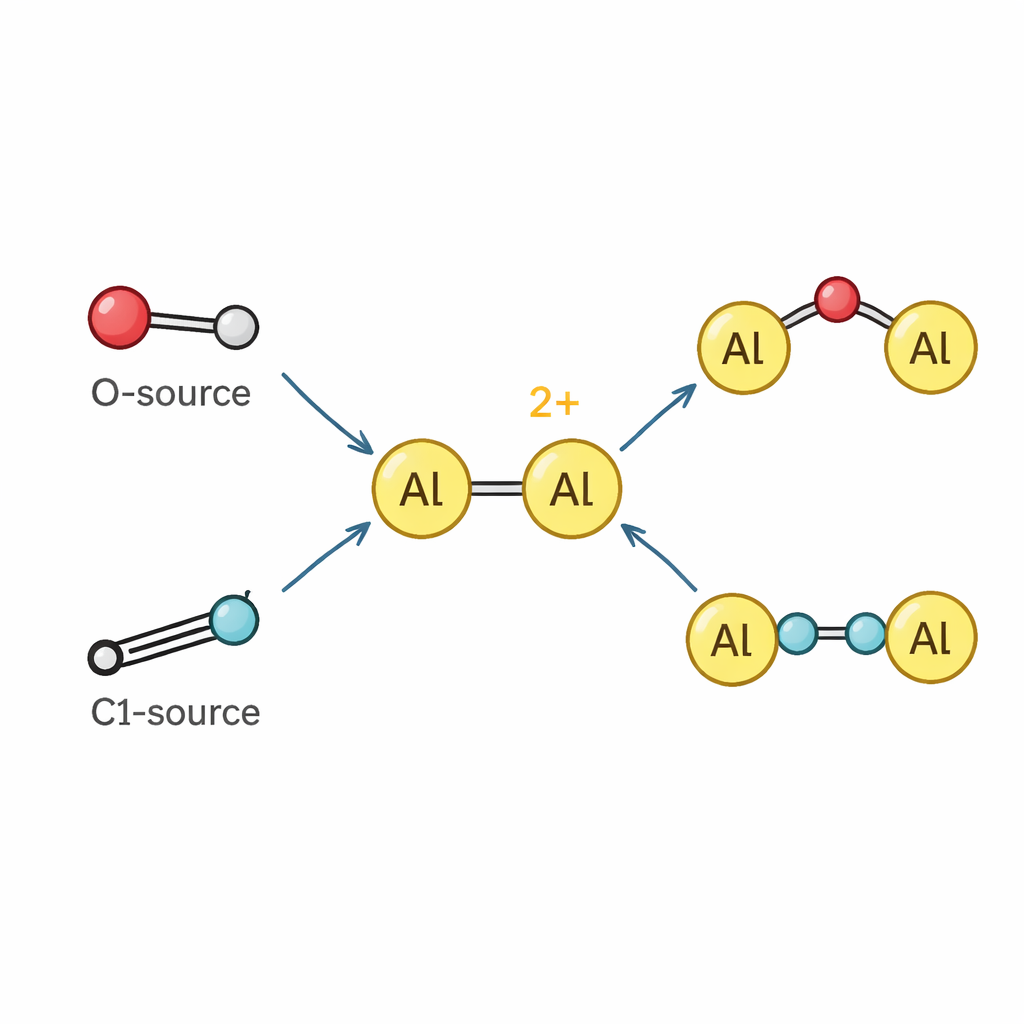
With the dication in hand, the team explored how it reacts with other molecules. Because its aluminum atoms are strongly electron-poor, the dication behaves as a powerful Lewis acid, readily binding basic partners such as pyridine and related molecules to give new dialuminum complexes. Beyond simple binding, the dication can split and insert into chemical bonds. It pulls oxygen atoms out of nitrous oxide and pyridine N-oxide, forming stable species where an oxygen atom bridges the two aluminum centers or is incorporated into a short aluminum–oxygen–silicon chain reminiscent of a tiny fragment of a zeolite framework. It also engages with isocyanides – small carbon–nitrogen units used as one-carbon building blocks – to stitch them together into longer N–C–C–N fragments that span between two aluminum atoms, demonstrating a rare example of isocyanide “homologation” driven by a main-group cation.
What this means going forward
In everyday terms, the researchers have turned an aluminum–aluminum bond into a controllable three-position electronic switch that can be toggled between neutral, singly charged, and doubly charged states, each with distinct shapes and reactivities. The doubly charged version is especially versatile, grabbing electrons and small fragments from other molecules in ways usually associated with more expensive transition metals. By showing that such behavior is possible with aluminum and that the different states can be reliably interconverted, this work opens the door to designing new, sustainable catalysts and functional materials built from one of Earth’s most abundant metals.
Citation: Liu, X., Kostenko, A., Körber, E. et al. Isolable radical cation and dication of dialumene. Nat Commun 17, 1937 (2026). https://doi.org/10.1038/s41467-026-69607-6
Keywords: aluminum catalysis, redox-switchable molecules, main-group chemistry, radical cations, small-molecule activation