Clear Sky Science · en
Treg-γδ T cell axis determines sexual dimorphism in hepatocarcinogenesis
Why some liver cancers hit men harder
Doctors have long known that liver cancer strikes men roughly twice as often as women, but the reasons go beyond simple differences in sex hormones. This study dives into the immune cells that live inside liver tumors and uncovers a hidden conversation between two types of T cells that helps explain why male livers are more vulnerable—and how this knowledge could point to new, more tailored treatments.
The immune tug-of-war inside liver tumors
Liver cancer, particularly hepatocellular carcinoma, grows within a complex neighborhood of immune cells, blood vessels, and scarred tissue. The authors compared tumors from men and women, in both patients and mouse models, and found a striking imbalance: male tumors were packed with more regulatory T cells, or Tregs, a cell type that normally helps calm down excessive immune reactions. At the same time, the total numbers of other common T cells did not differ much between the sexes. Because high Treg levels were linked with worse survival, the team suspected these cells were not just bystanders, but active accomplices helping tumors escape immune attack. 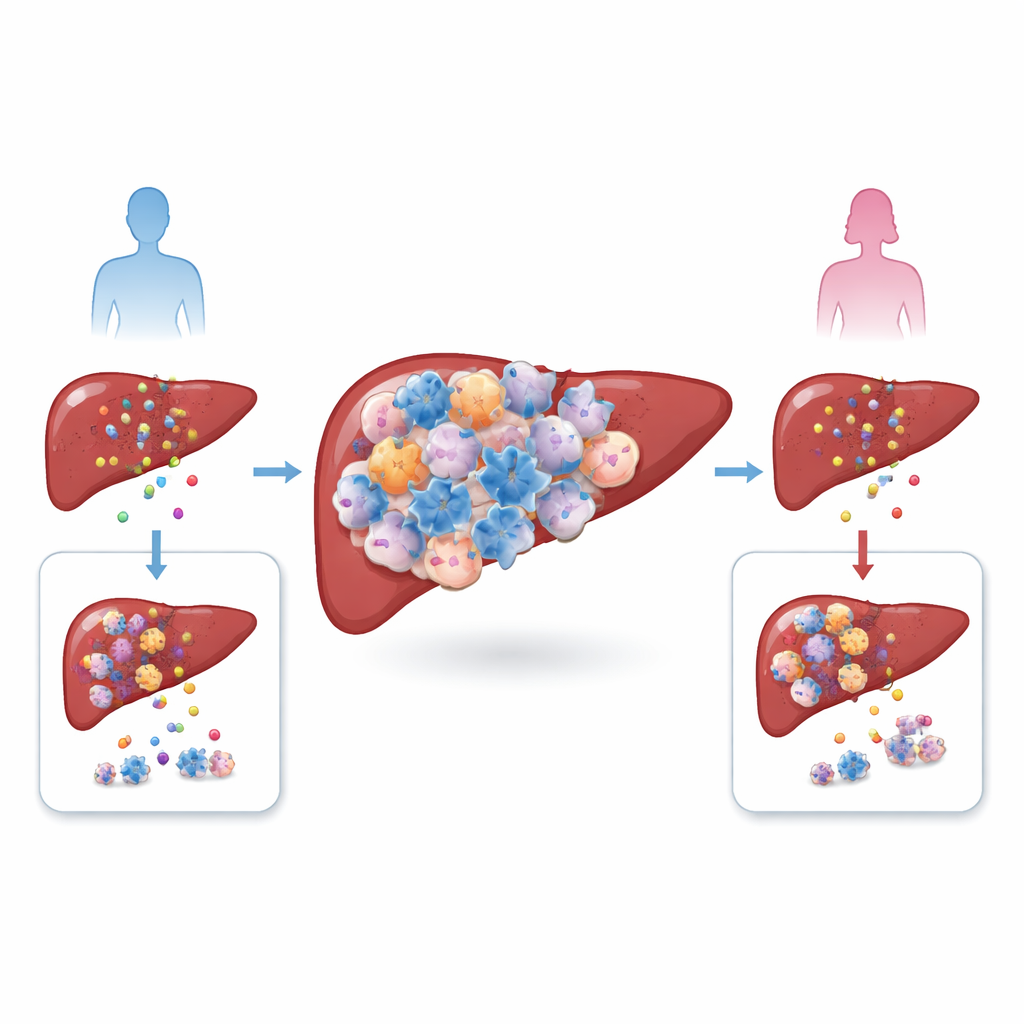
How male hormones call in suppressor cells
To understand why male tumors attract more Tregs, the researchers focused on chemical signals produced by cancer cells. They found that male liver tumors made higher amounts of a molecule called Ccl2, while Tregs carried more of its matching receptor, Ccr2. This pairing acts like a homing beacon, drawing Tregs into the tumor. When the Ccl2 signal or the Ccr2 receptor was removed in mice, tumors shrank and far fewer Tregs entered the liver, especially in males. The team then showed that testosterone, the main male sex hormone, drives this process: it activates a chain of proteins in liver cancer cells that switches on the Ccl2 gene, boosting the chemical call that summons Tregs. Castrated male mice, which produce less testosterone, had lower Ccl2 in their tumors and fewer infiltrating Tregs.
Guard cells that could fight cancer—but are held back
Another T cell type, known as gamma-delta T cells, can act as rapid-response “guards” that recognize and kill tumor cells without the usual antigen display machinery. In healthy liver, these cells are relatively scarce, but in liver cancer they can be potent tumor fighters. The study found that female tumors contained more of these gamma-delta cells and that they were more active and better armed with toxic molecules. Removing male sex hormones in mice increased the number and vigor of gamma-delta cells in tumors, while leaving classic killer CD8 T cells largely unchanged. In lab dishes, gamma-delta cells readily damaged liver cancer cells, but this effect was weakened when Tregs were added, suggesting that Tregs specifically restrain this promising anti-tumor population.
Hypoxia, secret packages, and silenced defenders
Digging deeper, the researchers asked how Tregs manage to turn down gamma-delta T cells. Inside tumors, oxygen is often scarce, a state called hypoxia. Under these low-oxygen conditions, Tregs in male tumors began to produce large amounts of a protein called S100a4. Instead of acting directly as a free molecule, S100a4 was packaged into tiny membrane-bound particles known as extracellular vesicles. These vesicles budded off from Tregs and were taken up by neighboring gamma-delta T cells. Once inside, S100a4 rewired the cells’ genetic control systems: it reduced the openness of DNA regions that control key signaling and killing molecules and lowered their activity. The gamma-delta cells became less able to respond through their T cell receptors and less capable of releasing toxic proteins and cytokines, effectively dulling their anti-cancer blade. 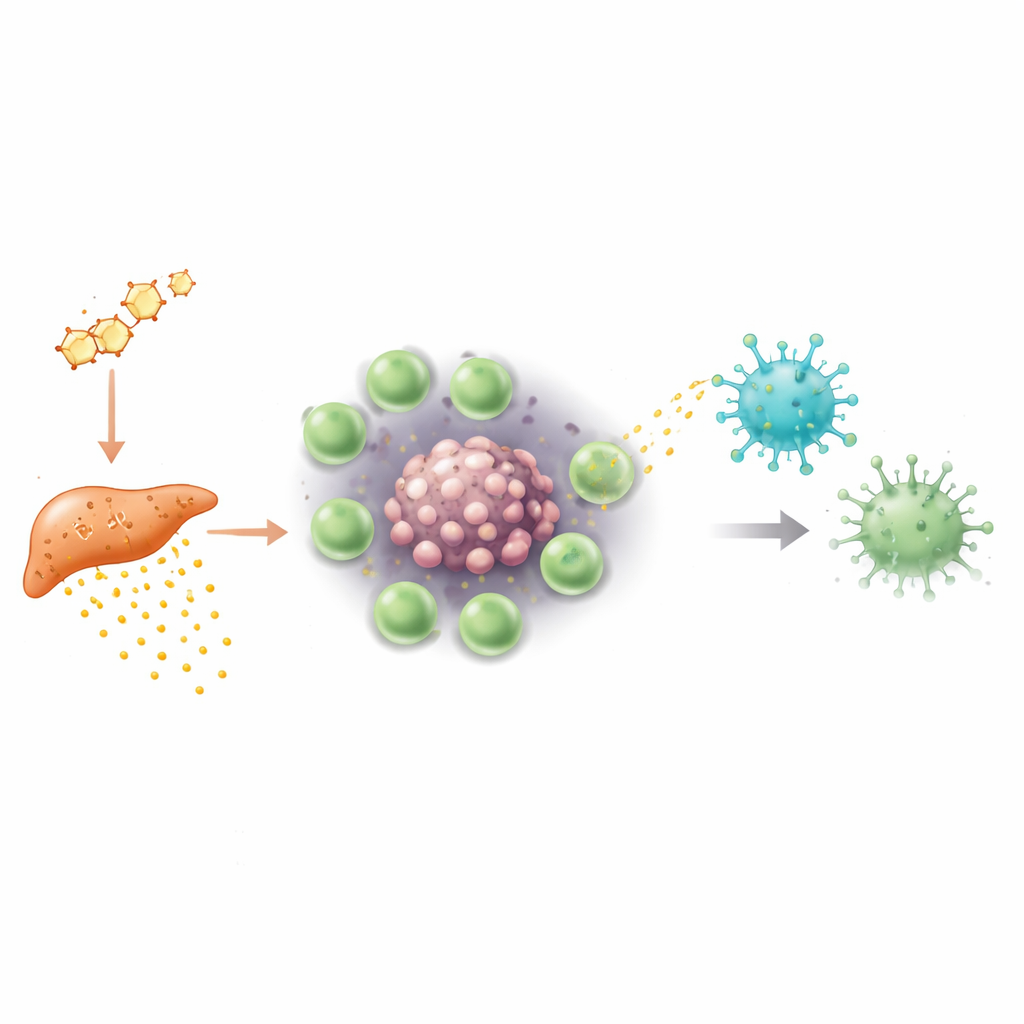
Breaking the harmful circuit
Finally, the team tested what happens if this Treg–S100a4–gamma-delta axis is interrupted. In mice engineered so that Tregs cannot make S100a4, male liver tumors shrank, the overall S100a4 level in tumors fell, and gamma-delta T cells became more abundant and more active. Notably, Treg numbers in the tumor did not drop—only their suppressive impact on gamma-delta cells was lifted. This change nearly erased the usual gap in tumor severity between male and female mice, underscoring how central this pathway is to sex-based differences in liver cancer.
What this means for future treatments
In accessible terms, the study shows that male hormones coax liver cancer cells to send out stronger “welcome signals” for immune-suppressive Tregs. Once inside the tumor—and under the stress of low oxygen—these Tregs ship out S100a4-filled vesicles that quietly disarm nearby gamma-delta T cells, which might otherwise keep the cancer in check. By targeting either the Ccl2 signal that brings Tregs in, or the S100a4 messages that silence gamma-delta cells, future therapies could restore the body’s own defenses, especially in men. This work suggests that liver cancer treatment may benefit from being sex-aware and from harnessing, rather than ignoring, the unique strengths of gamma-delta T cells.
Citation: Liang, Q., Zhang, Q., Zhang, W. et al. Treg-γδ T cell axis determines sexual dimorphism in hepatocarcinogenesis. Nat Commun 17, 2640 (2026). https://doi.org/10.1038/s41467-026-69603-w
Keywords: hepatocellular carcinoma, regulatory T cells, gamma-delta T cells, sex differences, tumor immunology