Clear Sky Science · en
Organocatalyzed bottom-up formation of protocells
From Simple Chemicals to Cell-Like Droplets
How did lifeless chemistry on the early Earth first give rise to something that looked and behaved like a cell? This study explores a surprisingly simple route: starting from small, readily available molecules, a single reaction network can build soap-like lipids that spontaneously gather into microscopic droplets and, eventually, protocells—cell-like compartments that could have sheltered the first steps toward life.
Why Compartments Matter for Life
Life depends on separating “inside” from “outside.” Modern cells use membranes—thin shells made mostly of fatty molecules—to trap and concentrate useful chemicals while keeping harmful ones out. For decades, origin-of-life researchers have mimicked this with ready-made fatty acids or phospholipids, assembling them into hollow bubbles called vesicles. But this leaves a key question unanswered: could early Earth chemistry have made both the membrane-building molecules and the primitive compartments in one continuous process, without starting from already finished lipids?
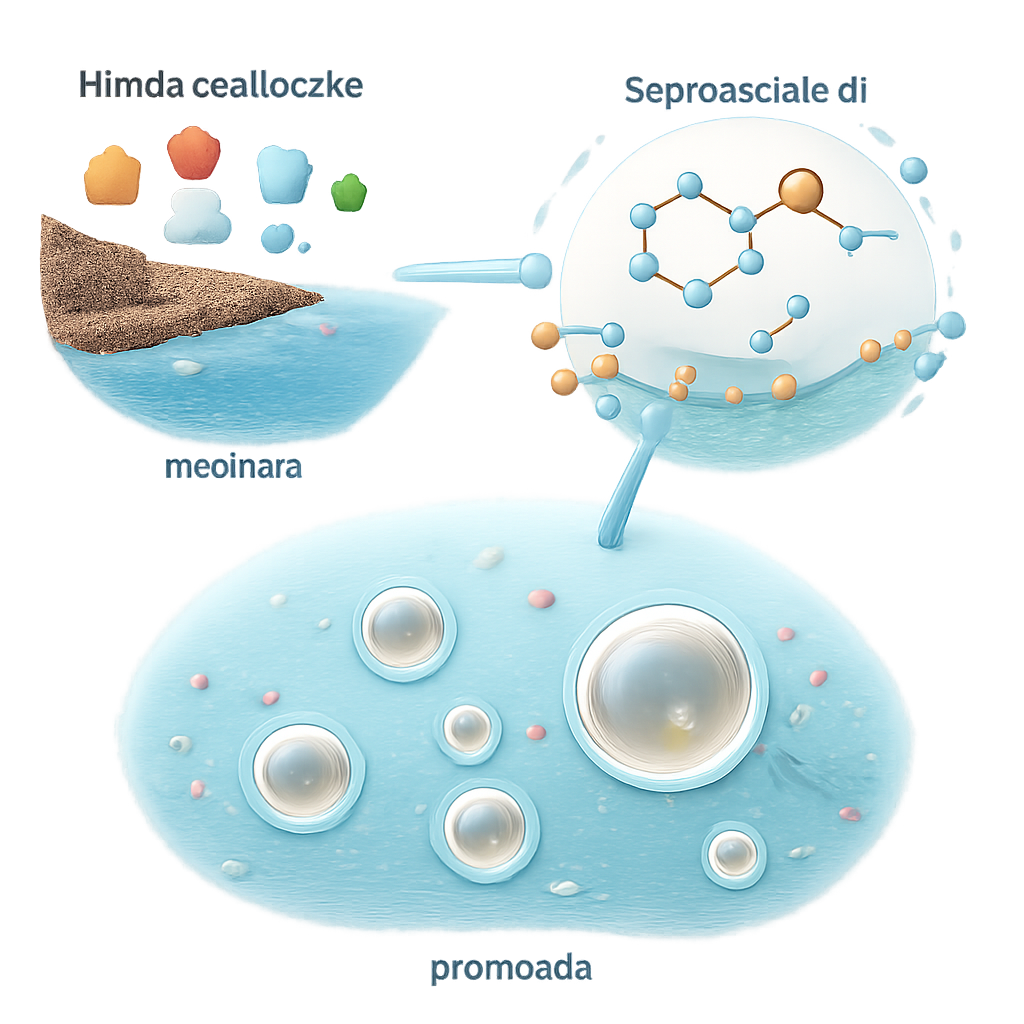
Building Lipids from Scratch
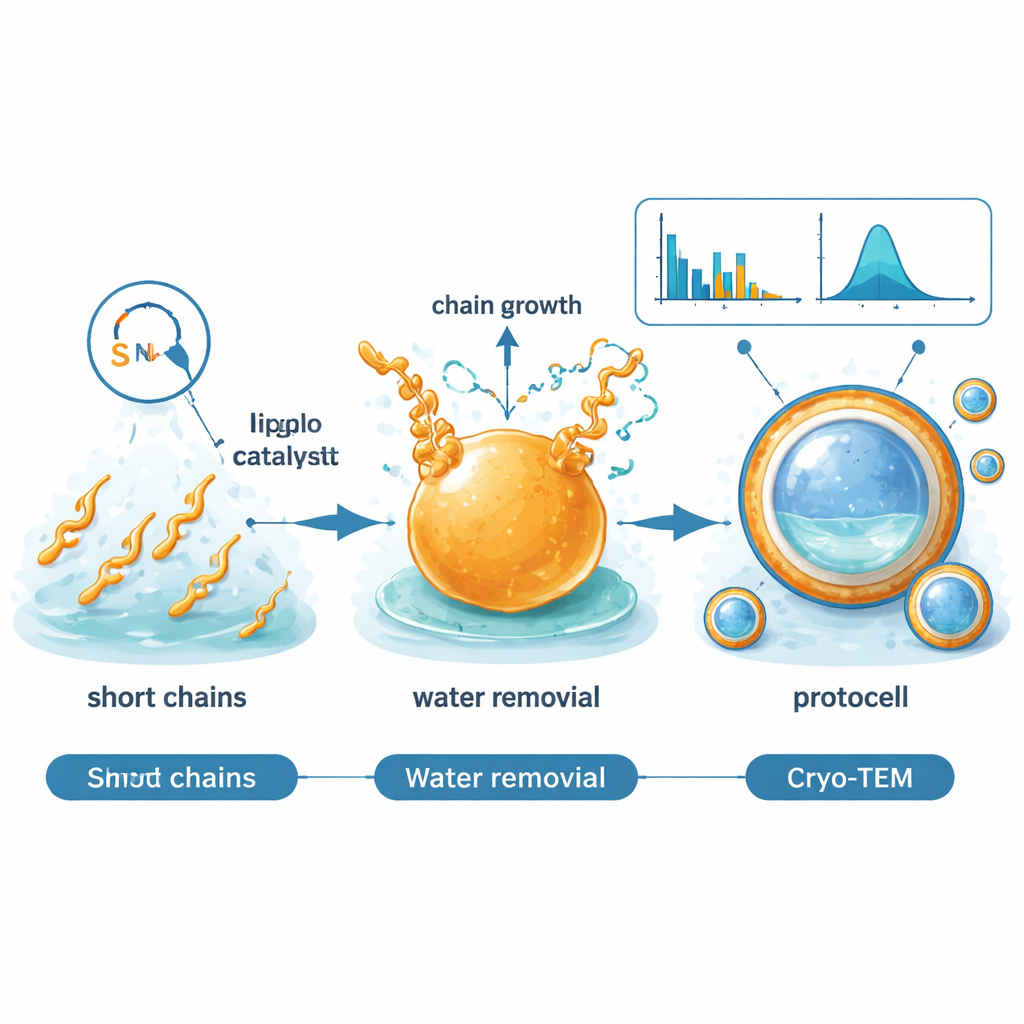
The authors describe a bottom-up route that starts from acetaldehyde, a small, plausible early-Earth molecule that can be produced from carbon dioxide by minerals in volcanic or meteorite material. In mildly acidic water, they add a simple sulfur-containing organic catalyst called an imidazolidine-4-thione. This catalyst links acetaldehyde units together in a repeating pattern, step by step, forming longer carbon chains decorated with a few oxygen atoms. As the reaction progresses, water is eliminated from these chains, turning them into increasingly oily, lipid-like molecules up to 20 carbons long—the same size range favored by modern biological membranes.
Catalysts That Evolve as They Work
A striking twist is that the catalyst itself is not a passive bystander. The new lipid-like aldehydes can chemically attach to, and then reorganize, the catalyst’s structure. In effect, the catalyst swaps its side chains with products it has just made, generating a family of related catalyst molecules with different tails. These modified versions remain active and can further shape which products form next. The system thus behaves a bit like a primitive form of molecular evolution: the reaction network creates a mixture of catalysts, some of which are better tuned to sustain the process under specific conditions of pH, temperature, and salt content that resemble early oceans.
Spontaneous Formation of Protocells
As more lipid-like molecules accumulate, the reaction mixture turns cloudy. Microscopy, dynamic light scattering, and cryo-electron microscopy show that tiny droplets first appear, then grow and diversify in size from about 10 nanometers to several micrometers. Initially, the droplets behave like oil beads in water, with the catalyst molecules lining the surface so that their water-friendly heads face outward and their oily tails point inward. As the reaction continues, the chemistry steadily removes water from within the oily phase and produces extra water that separates out into small pockets. These internal water droplets merge and sometimes push toward the outside, reshaping the oil droplet into a structure with a thin, lipid-rich boundary enclosing an inner watery compartment—essentially a protocell. The membrane remains permeable enough to let in fluorescent dyes, and by analogy, other small organic molecules, allowing them to concentrate inside.
Robust Under Early-Earth Conditions
The team tested how sturdy these protocells are across different pH values, temperatures, and salt mixtures intended to mimic primitive seas. The organocatalytic system not only tolerates such variation but sometimes benefits from it: certain salts speed up the reaction, and moderately acidic conditions favor both chain-building and water removal. Unlike many modern fatty acid membranes that fall apart in the presence of common metal ions, these protocell structures remain stable with magnesium and calcium. Once formed, they can grow and multiply in number as more lipid-like material is produced, while continuously concentrating organic compounds within their interiors.
What This Means for the Origin of Life
To a non-specialist, the main message is that you can start with very simple chemicals, subject them to gentle conditions, and still end up with tiny, cell-like containers that organize and enrich their internal chemistry. This work suggests a realistic pathway by which Earth’s early environment could have generated both the building blocks of membranes and the first protocells at the same time, using small, plausible catalysts instead of complex enzymes. Such self-assembled, catalytically active protocells offer a natural stage on which more elaborate molecules—such as RNA—could have formed, accumulated, and eventually taken on the roles we now associate with living cells.
Citation: Ebeling, M.S.R., Berninghausen, O., Nguyen, K.H. et al. Organocatalyzed bottom-up formation of protocells. Nat Commun 17, 1983 (2026). https://doi.org/10.1038/s41467-026-69597-5
Keywords: origin of life, protocells, prebiotic chemistry, self-assembly, organocatalysis