Clear Sky Science · en
Dual recruitment of two CCM2 molecules to KRIT1 suppresses KLF4 expression
Why the Brain’s Blood Vessels Need Careful Control
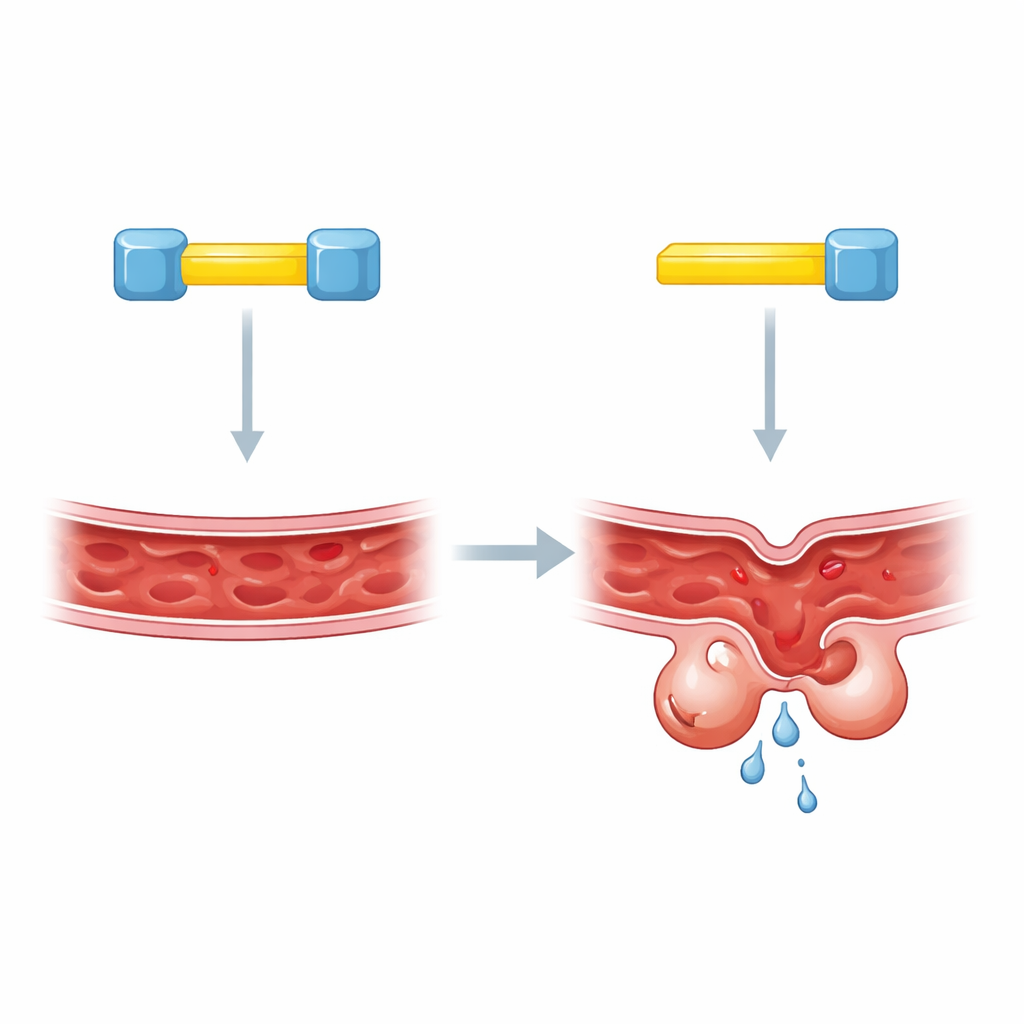
Blood vessels in the brain must walk a tightrope: they have to be sturdy enough to avoid leaks yet flexible enough to respond to the body’s needs. When this balance fails, people can develop cerebral cavernous malformations—clusters of fragile, bubble-like blood vessels that may bleed and cause seizures or strokes. This study uncovers a previously unknown molecular handshake between three proteins that helps keep brain vessels stable, and shows how breaking that handshake can drive disease.
The Gatekeepers of Vessel Health
Cells lining blood vessels rely on a set of master switches, called KLF2 and KLF4, to control genes that maintain vessel strength and identity. If these switches are turned up too high, vessels become abnormal and prone to damage. Two other proteins, KRIT1 and CCM2, are known to guard this system. People who inherit faulty versions of either protein are at high risk for cerebral cavernous malformations. Until now, however, scientists did not understand exactly how KRIT1 and CCM2 worked together to keep KLF4 in check.
A Surprising Team of Three
The researchers used human endothelial-like cells grown in the lab and selectively reduced the amount of CCM2 or KRIT1 using genetic tools. When either protein was depleted, KLF4 levels shot up, echoing what is seen in animal models and patients. Restoring normal CCM2 or KRIT1 brought KLF4 back down—but only if the restored proteins could still stick to each other. By engineering precise changes in the regions where CCM2 and KRIT1 interact, the team showed that this physical contact is essential for keeping KLF4 from rising. These experiments firmly place the KRIT1–CCM2 partnership at the heart of the control system for this powerful transcription factor.
Two Locks on One Key
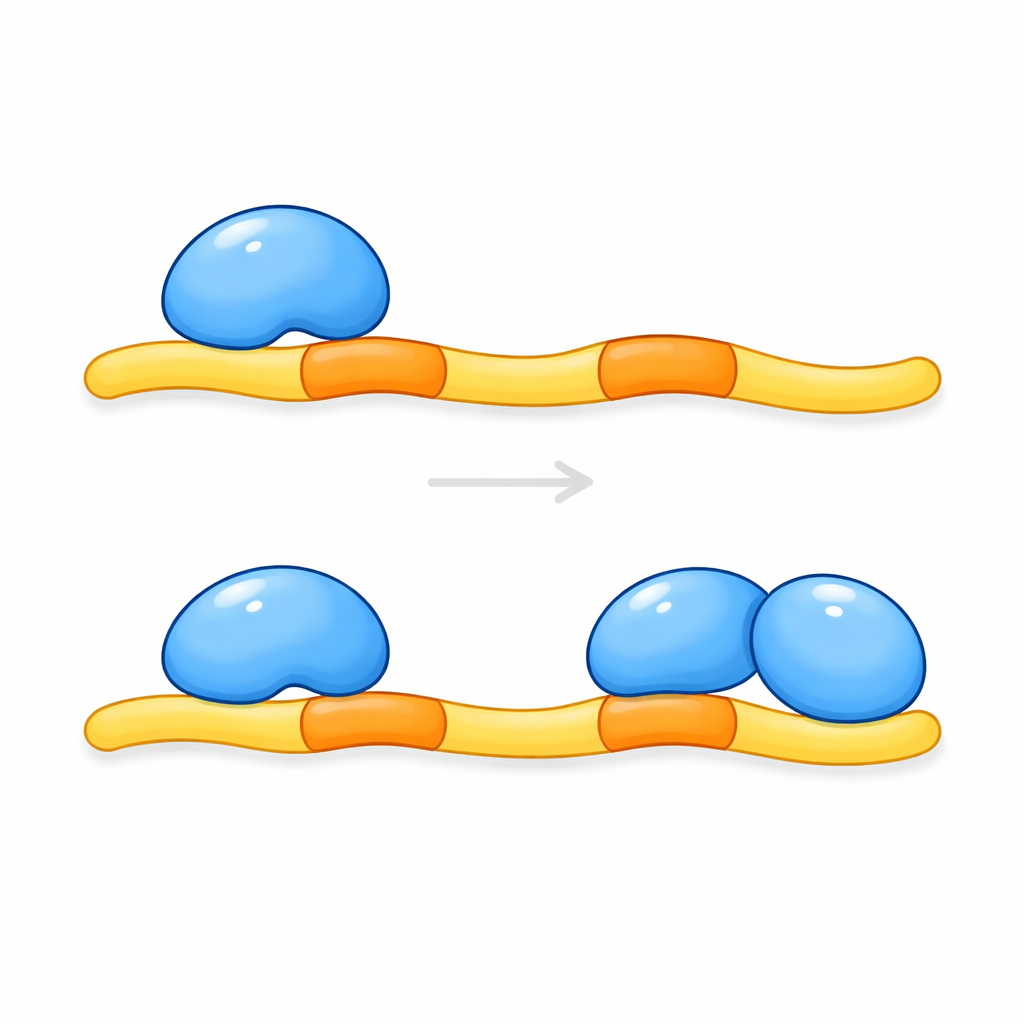
To dig deeper, the authors focused on short segments within KRIT1 that act as molecular docking sites. Earlier work suggested that CCM2 grabs onto KRIT1 at one of these sites, but different methods had produced conflicting answers about which one. The new study reveals that the confusion arose because the true picture is more complex: KRIT1 carries two nearby docking segments, and both are needed. When either segment was disrupted, KLF4 levels remained abnormally high, just as if KRIT1 were missing altogether. Biochemical tests in engineered cells showed that a single KRIT1 protein can bridge two separate CCM2 molecules at once, hinting at an unexpected three-part assembly.
Seeing the Molecular Clamp in Detail
The team then purified the relevant pieces of CCM2 and KRIT1 and examined how they come together in solution and in crystals. Measurements of the complex’s mass indicated that two copies of a key CCM2 region bind to a single strip of KRIT1 carrying the pair of docking sites. High-resolution structural snapshots confirmed this 2:1 arrangement: one extended KRIT1 peptide threads between two CCM2 domains, with each docking site nestling into a similar pocket on a different CCM2 molecule. A short helical segment between the docking sites helps position the two CCM2 domains side by side, creating a snug, clamp-like structure. This type of dual engagement—two binding domains latching onto tandem sites along one partner—had not been seen before for this family of protein modules.
Implications for Brain Disease and Beyond
These findings show that KRIT1 does not simply bind one CCM2 molecule; instead, it acts as a scaffold that recruits a pair of CCM2 proteins. Only when both docking sites on KRIT1 are occupied can the complex properly restrain KLF4 production and, by extension, help prevent fragile vessel growth. Mutations in KRIT1 that interfere with either docking site are therefore expected to weaken this protective clamp and may explain some inherited forms of cerebral cavernous malformations. Because many other human proteins carry similar twin docking motifs, this study also hints at a broader principle: dual binding by pairs of related domains may be a common way that cells fine-tune signaling networks. Understanding this tri-partner interaction could ultimately guide strategies to stabilize blood vessels in people at risk of brain bleeds.
Citation: Huet-Calderwood, C., Fisher, O.S., Das, S. et al. Dual recruitment of two CCM2 molecules to KRIT1 suppresses KLF4 expression. Nat Commun 17, 2719 (2026). https://doi.org/10.1038/s41467-026-69595-7
Keywords: cerebral cavernous malformations, endothelial cells, KRIT1, CCM2, KLF4