Clear Sky Science · en
Concentrating and directing energy flow in plasmonic heterostructures for stable and efficient light-driven methane dry reforming
Turning Greenhouse Gases into Useful Fuel
Methane and carbon dioxide are two of the most powerful greenhouse gases warming our planet, yet they are also rich sources of chemical energy. This study explores a way to turn these problem gases into “syngas,” a building block for cleaner fuels and everyday chemicals, using only light as the driving force. By designing tiny metal structures that act like miniature antennas for light, the researchers show how to run this transformation efficiently while avoiding the soot that usually kills such catalysts.
A Cleaner Route from Waste Gas to Useful Gas
Industry already knows how to combine methane and carbon dioxide into syngas, but today’s methods demand furnace-like temperatures of 700–1000 °C. Those harsh conditions consume large amounts of energy, create extra emissions, and cause the working materials (catalysts) to clog with carbon deposits, or “coke.” The team set out to design a catalyst that could work at far lower temperatures, be powered mainly by light, and resist this carbon buildup. Achieving all three at once would make it much more practical to recycle greenhouse gases into valuable products such as fuels and plastics precursors.
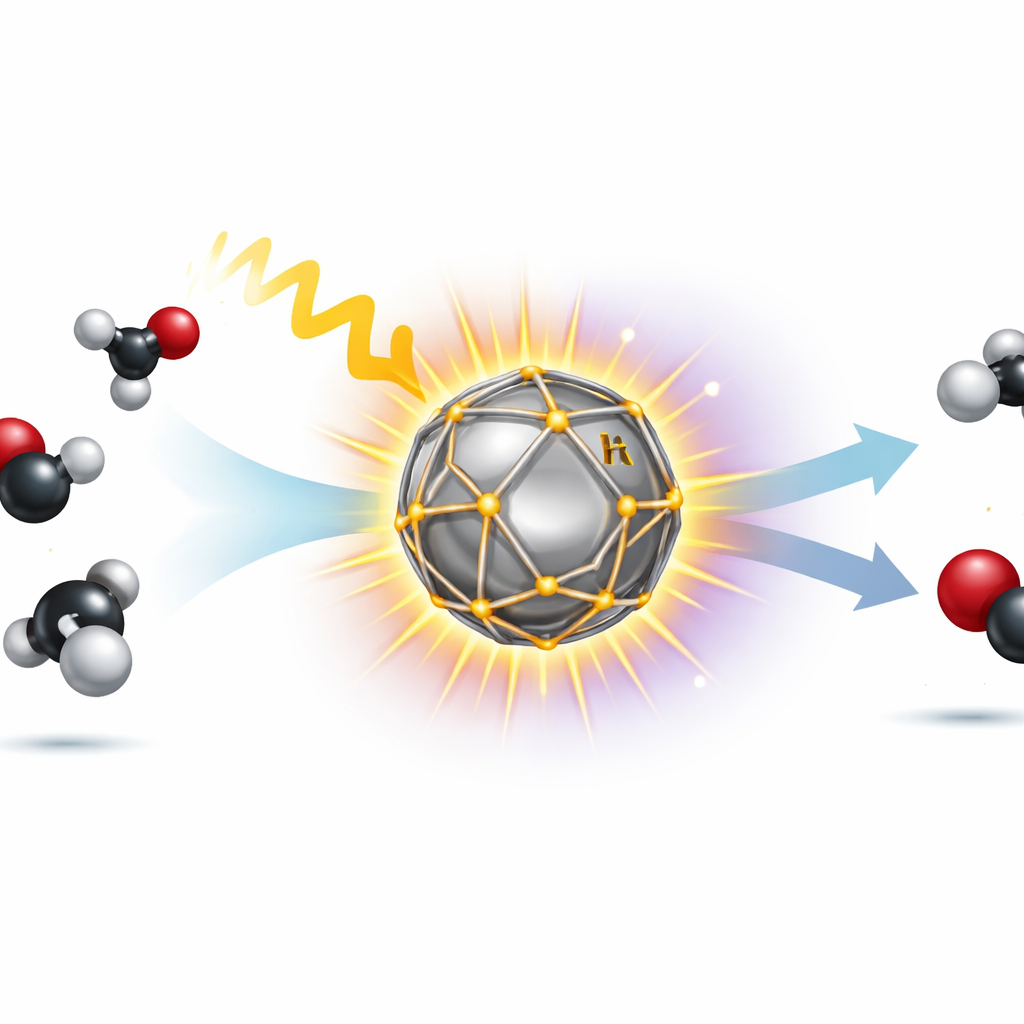
Tiny Metal Cages that Harvest Light
The researchers created nanometer-scale particles with a silver core wrapped in a cage-like shell of iridium. Silver is excellent at concentrating light into intense local fields through an effect known as plasmon resonance, while iridium is highly active for the methane–carbon dioxide reaction. By growing the iridium only on the sharp corners and edges of the silver core, the structure preserves silver’s strong light absorption and directs the concentrated energy precisely where reactions occur. Advanced electron microscopy confirmed that the iridium indeed forms an ultrathin cage at these hot spots rather than a uniform coating that would block light.
Guiding Energy Instead of Wasting Heat
Optical measurements and computer simulations showed that when illuminated, the silver core generates energetic charge carriers—“hot” electrons—that can quickly move into the iridium cage. Compared with pure silver particles, the core–cage design channels more of the absorbed light into these hot carriers instead of simply heating up. Ultrafast laser experiments revealed that the lifetime of these carriers roughly doubles in the Ag–Ir structures, giving them more time to drive chemical steps on the surface. Simulations of the electromagnetic field confirmed that the strongest energy concentration appears at the iridium-decorated corners and edges, just where the reacting molecules land.
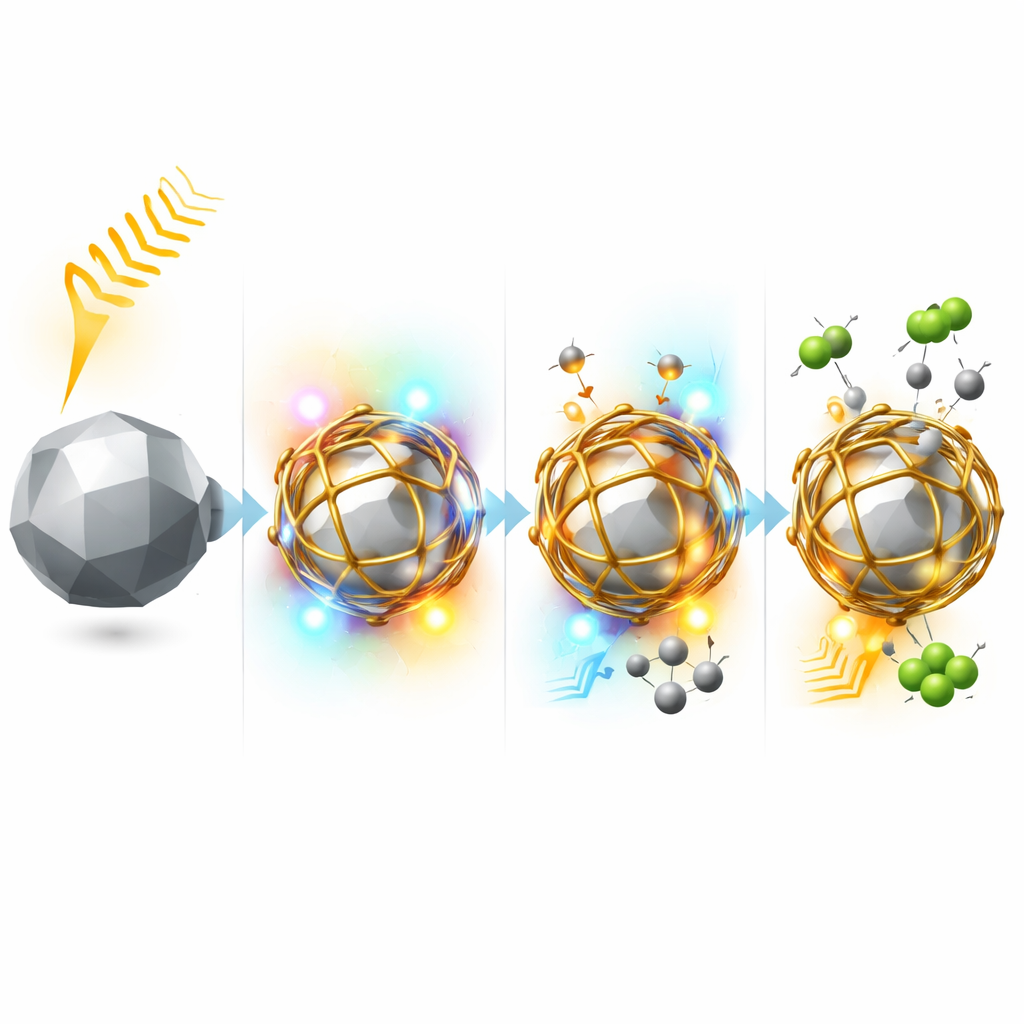
Stable, Light-Driven Conversion Without Soot
When tested under bright lamp illumination with no external heating, the silver–iridium cages produced hydrogen and carbon monoxide at high rates, with product selectivity above 97%, and remained active for more than 300 hours. In contrast, iridium-only cages quickly lost activity and accumulated carbon deposits, while silver-only particles barely reacted at all. Temperature and light-intensity studies showed that the reaction is governed mainly by light-generated charge carriers, with some modest heating needed just to get things started. Infrared spectroscopy and theoretical calculations further revealed that on the Ag–Ir surface, methane tends to be converted into oxygen-containing fragments that can be fully oxidized to carbon monoxide, instead of leaving behind solid carbon. This altered pathway is key to avoiding coke.
Why This Matters for Future Energy
In everyday terms, the study demonstrates a tiny, highly engineered “light funnel” that gathers energy from visible light and delivers it directly to the spots where hard-to-activate molecules sit. By steering this energy into useful chemical steps and away from destructive side reactions, the silver–iridium cages turn methane and carbon dioxide into valuable syngas efficiently and for long periods without fouling. The approach offers a blueprint for designing next-generation light-powered catalysts that could reclaim waste gases and help close the carbon loop in future chemical and fuel production.
Citation: Yin, T., Yuan, H., Wang, Q. et al. Concentrating and directing energy flow in plasmonic heterostructures for stable and efficient light-driven methane dry reforming. Nat Commun 17, 2672 (2026). https://doi.org/10.1038/s41467-026-69581-z
Keywords: methane dry reforming, plasmonic photocatalysis, greenhouse gas conversion, nanostructured catalysts, syngas production