Clear Sky Science · en
Contrast-free identification of glioma blood-brain barrier status via generative diffusion AI and non-contrast MRI
Why this matters for brain tumor patients
People with brain tumors routinely undergo MRI scans, and many also receive injected contrast dye so doctors can see whether the brain’s protective blood–brain barrier has been damaged. That information guides surgery, radiation, and prognosis—but the dye can carry risks, adds time and cost, and is not always available. This study presents an artificial intelligence (AI) system that reads routine, dye‑free MRI scans and virtually recreates the missing contrast images, allowing doctors to infer blood–brain barrier status without injecting any contrast agent.
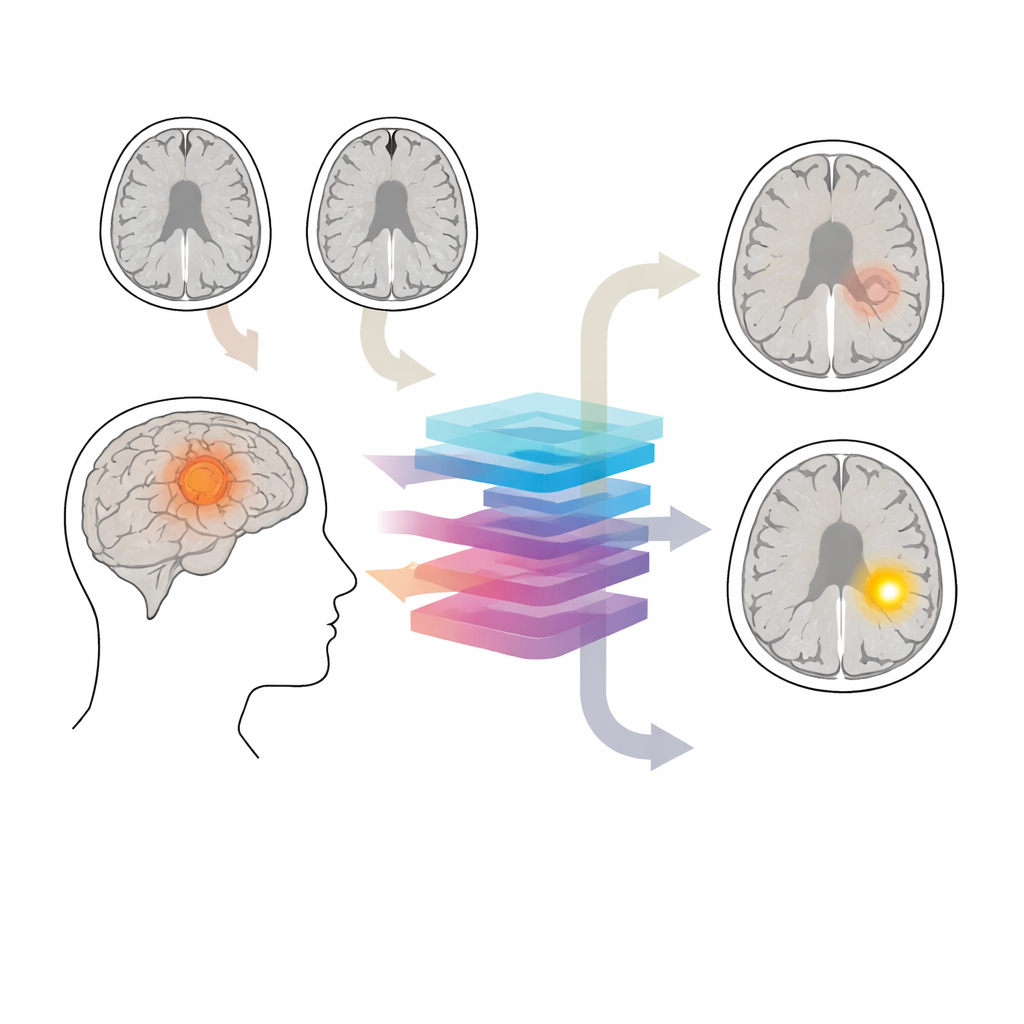
The brain’s protective gatekeeper
The blood–brain barrier acts like a microscopic filter around blood vessels in the brain, letting only tiny molecules pass through. In many glioma tumors, this barrier becomes leaky. On standard contrast MRI, leaky areas light up after injection, while intact regions stay dark. These patterns influence how aggressively a tumor is treated and help define what to remove surgically or target with radiation. Yet contrast scans are sometimes skipped—for example in early work‑ups or long‑term follow‑up of slow‑growing tumors—and some patients should avoid contrast entirely because of kidney disease, allergic reactions, or concerns about gadolinium deposition in the brain.
Turning ordinary scans into virtual contrast
The authors asked whether non‑contrast MRI scans, which are already widely obtained, contain enough subtle information for AI to infer what a contrast scan would have shown. They focused on two common sequences: T1 and T2‑FLAIR. While these do not directly show blood–brain barrier leakage, they capture tumor structure, surrounding swelling, and tissue damage that often accompany barrier breakdown. The team built a system called CBSI that first generates synthetic contrast‑enhanced images from these non‑contrast scans and then uses them, together with the original images, to decide whether the barrier is intact or disrupted in each glioma.
How the new AI system works
At the heart of CBSI is a diffusion model—a kind of AI that starts from noisy images and iteratively “denoises” them into realistic pictures. Here, it learns to turn non‑contrast input into two alternative virtual contrast images: one assuming an intact barrier and one assuming a leaky barrier. A guidance mechanism tells the model which pattern of enhancement to aim for, and an auxiliary segmentation step helps it pay extra attention to the tumor region. In a second stage, a separate network compares the two synthetic outcomes for each patient and judges which one is more self‑consistent with the original MRI data. That decision is then translated into the predicted blood–brain barrier status.
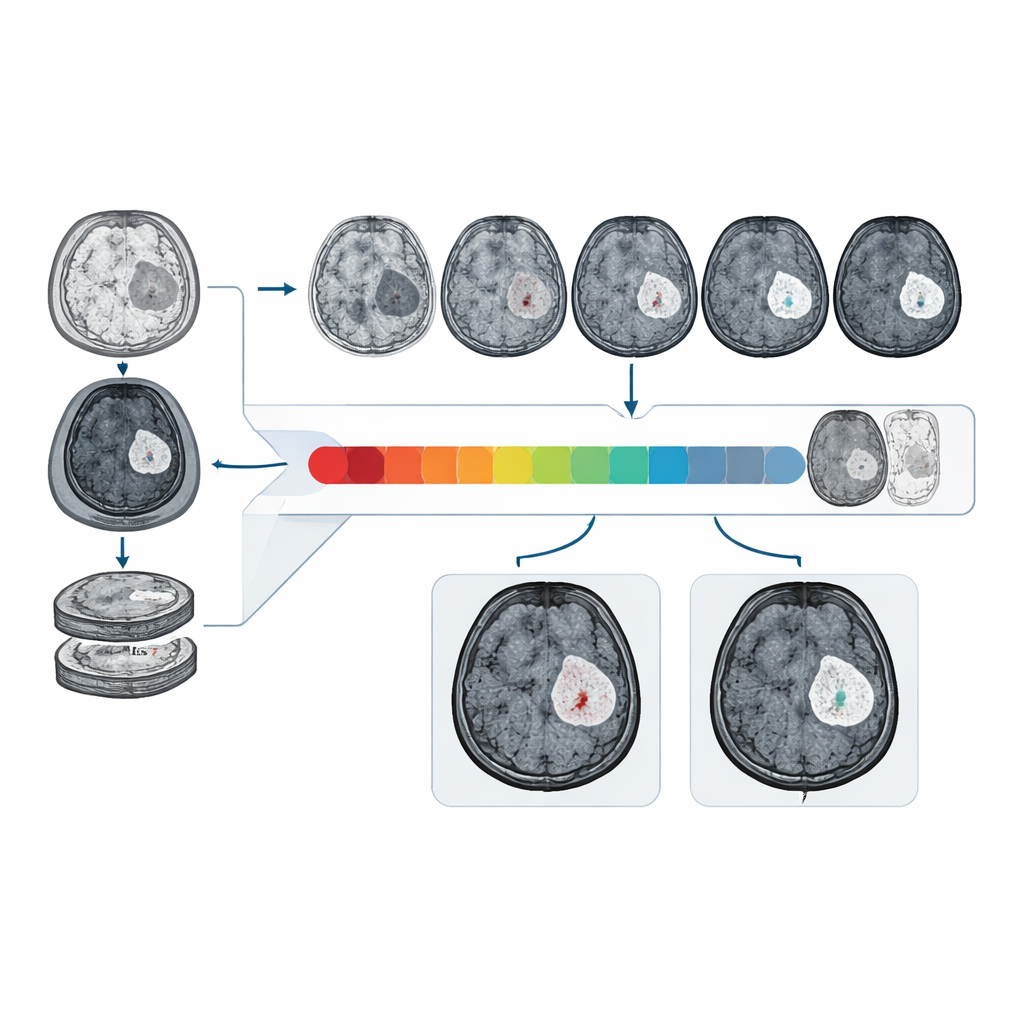
How well it performs in real and diverse data
The researchers trained and tested CBSI on MRI data from 1,535 glioma patients collected at multiple hospitals and from international public datasets. On an external clinical test set, CBSI distinguished intact from disrupted barriers with an area under the curve of about 81%, clearly better than models that relied only on non‑contrast images and approaching the performance of models that had access to real contrast scans. Radiologists, when shown randomized mixes of real and synthetic contrast images from the system, judged over 90% of the synthetic images to be of high diagnostic quality, and their ability to identify contrast enhancement on synthetic images was similar to their performance on real scans.
Benefits for treatment planning and future care
Because the synthetic contrast images produced by CBSI capture tumor detail and enhancement patterns, they also improved downstream tasks such as outlining tumor boundaries and estimating tumor grade—both of which depend heavily on how the blood–brain barrier behaves. The method generalized well to different scanners, imaging protocols, and patient populations, including data from Sub‑Saharan Africa and larger international cohorts. While the authors acknowledge that any deep‑learning system remains a statistical tool rather than a direct window into biology, their results suggest that virtual contrast imaging could reduce reliance on injected agents, especially for vulnerable patients or in settings where contrast scans are difficult to obtain. In practical terms, this AI approach could allow doctors to extract key treatment‑shaping information about the blood–brain barrier from the scans patients are already getting—without an extra needle stick.
Citation: Zheng, K., Zhang, Y., Shu, H. et al. Contrast-free identification of glioma blood-brain barrier status via generative diffusion AI and non-contrast MRI. Nat Commun 17, 2162 (2026). https://doi.org/10.1038/s41467-026-69578-8
Keywords: glioma, blood-brain barrier, brain MRI, medical imaging AI, contrast-free imaging