Clear Sky Science · en
Activating plasmonic catalysis through light-mediated steady-state spin modulation
Turning Light into a Chemical Tuning Knob
Chemists have long dreamed of catalysts that can be switched “on demand” with light, making reactions faster, cleaner, and more selective without adding extra chemicals or applying large magnetic fields. This study shows a way to do just that by using tiny metal structures to let light reshape the internal magnetic state of a catalyst, and then using that state to steer a real-world reaction that turns a common water pollutant, nitrate, into useful ammonia. 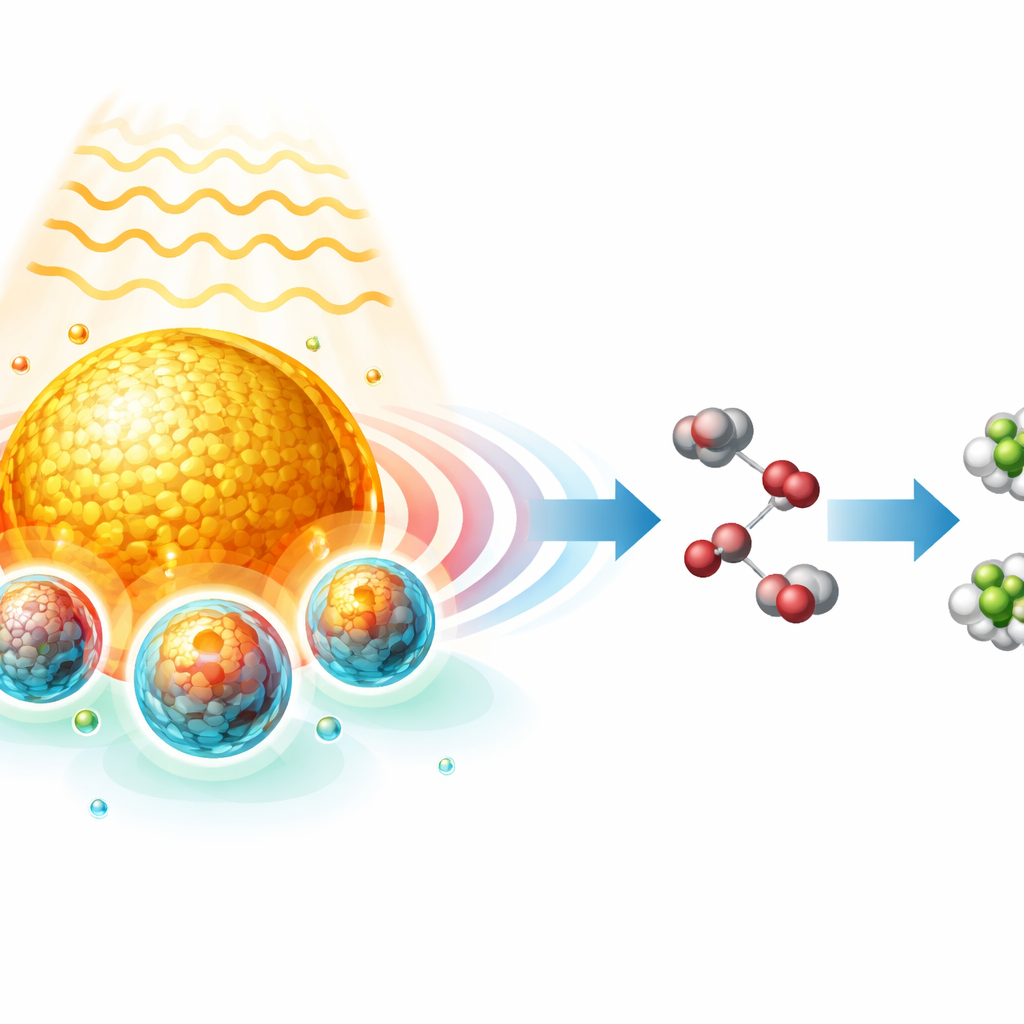
Why Spinning Electrons Matter for Reactions
At the heart of this work is the idea that how electrons spin inside a catalyst atom can change how that surface grabs and transforms molecules. Electrons can sit in a “low-spin” arrangement, where they are more tightly paired, or a “high-spin” arrangement, where more electrons remain unpaired and magnetic. High-spin states can expose more reactive sites and shift how strongly a surface holds on to reacting molecules. The problem is that when light pushes a material into a high-spin state, it usually falls back in a fraction of a billionth of a second—far faster than most chemical steps such as adsorption, diffusion, and bond breaking. That mismatch has made spin control seem more like a fleeting curiosity than a practical tool for catalysis.
Using Tiny Gold Antennas to Hold Spin in Place
The researchers solve this timing problem by combining two components into a single “antenna–reactor” particle. A gold nanoparticle acts like a miniature antenna that responds strongly to certain colors of light through a phenomenon called localized surface plasmon resonance, in which the metal’s electrons slosh back and forth collectively and create an intense, rapidly oscillating electromagnetic near field. Wrapped around this antenna, but separated by a thin, transparent silica shell, sit nanocrystals of cobalt ferrite (CoFe₂O₄), a spin-active oxide whose iron atoms can switch between low- and high-spin states. When illuminated at the right wavelength, the near field from the gold concentrates energy directly into the nearby cobalt ferrite, driving its iron sites into a high-spin state and, crucially, maintaining that state for tens of microseconds—long enough to overlap with the slower chemical events taking place on the surface.
Proving That Light Really Rewrites the Catalyst’s State
To confirm that they were not just heating their material but genuinely changing its spin and structure, the team used a suite of sensitive probes. X-ray emission and absorption measurements showed clear shifts in iron spectral lines toward lower binding energies under light, as expected when more unpaired electrons appear in high-spin states. Raman spectroscopy revealed new vibrational peaks that only emerge under resonant illumination, again pointing to a spin change rather than simple warming. Transient absorption experiments captured a long-lived excited species with a lifetime of about 60 microseconds, matching the picture of a stabilized high-spin population. Calculations using quantum mechanical models supported these findings, indicating that high-spin cobalt ferrite has longer metal–oxygen bonds, greater magnetic moment, and an electronic landscape that favors stronger and more flexible bonding to incoming reactants. 
Steering Nitrate into Ammonia with Light-Boosted Spins
The authors then tested whether this spin-tuned catalyst actually performs better in a demanding reaction: the electrochemical reduction of nitrate to ammonia in alkaline water. Under sunlight-like illumination, the gold–cobalt ferrite particles produced much larger currents and far higher ammonia yields than in the dark, or than control samples lacking the plasmonic antenna. The light-modded catalyst not only accelerated the overall reaction but also shifted the pathway to favor ammonia over unwanted side products such as nitrogen gas or hydrogen. In situ Raman measurements detected key nitrogen-containing intermediates forming and disappearing over time, while theoretical energy diagrams showed that the high-spin state lowers the energy barriers for crucial steps and makes it easier for the final ammonia molecules to let go of the surface and escape into solution.
Broad Promise for Cleaner, Smarter Chemistry
In simple terms, this study shows that carefully designed nanoparticles can use light not just as fuel but as a fine control knob, locking a catalyst into a more reactive magnetic state long enough to matter for real chemical reactions. By stabilizing high-spin iron in cobalt ferrite through the near field of a gold antenna, the team greatly improves the sunlight-driven conversion of nitrate—a widespread pollutant—into valuable ammonia with high efficiency and selectivity. Because the strategy does not rely on bulky external magnets or permanent structural changes, it could be adapted to many other combinations of plasmonic metals and spin-active catalysts, offering a general route toward smart, light-programmable materials for catalysis, sensing, and energy conversion.
Citation: Hu, X., Liu, J., Zhu, Z. et al. Activating plasmonic catalysis through light-mediated steady-state spin modulation. Nat Commun 17, 2849 (2026). https://doi.org/10.1038/s41467-026-69577-9
Keywords: plasmonic catalysis, spin modulation, nitrate reduction, nanoparticles, photocatalysis