Clear Sky Science · en
Nanobody-based IgG simultaneously inhibit the allergenic and enzymatic activity of the dominant honeybee venom allergen
Why bee sting allergies matter
For most people, a honeybee sting is painful but short‑lived. For others, it can trigger a life‑threatening whole‑body reaction called anaphylaxis. Today’s main preventive treatment, venom immunotherapy, works for many patients but takes years, involves frequent injections, and can itself provoke allergic side effects. This study explores a new, more targeted way to protect people who are dangerously allergic to honeybee venom by disarming one key venom component before it can cause harm.
The main troublemaker in bee venom
Honeybee venom is a cocktail of different molecules, but one protein, called phospholipase A2 (or Api m 1), stands out. It is the dominant allergen for honeybee‑allergic patients and is present in almost all of them. Api m 1 has a double role: it damages cell membranes, contributing to pain and inflammation at the sting site, and it is also a prime target for allergy‑causing antibodies known as IgE. When Api m 1 binds to IgE that is anchored on immune cells such as mast cells and basophils, it can set off a chain reaction leading to the sudden release of inflammatory chemicals and, in severe cases, anaphylaxis.
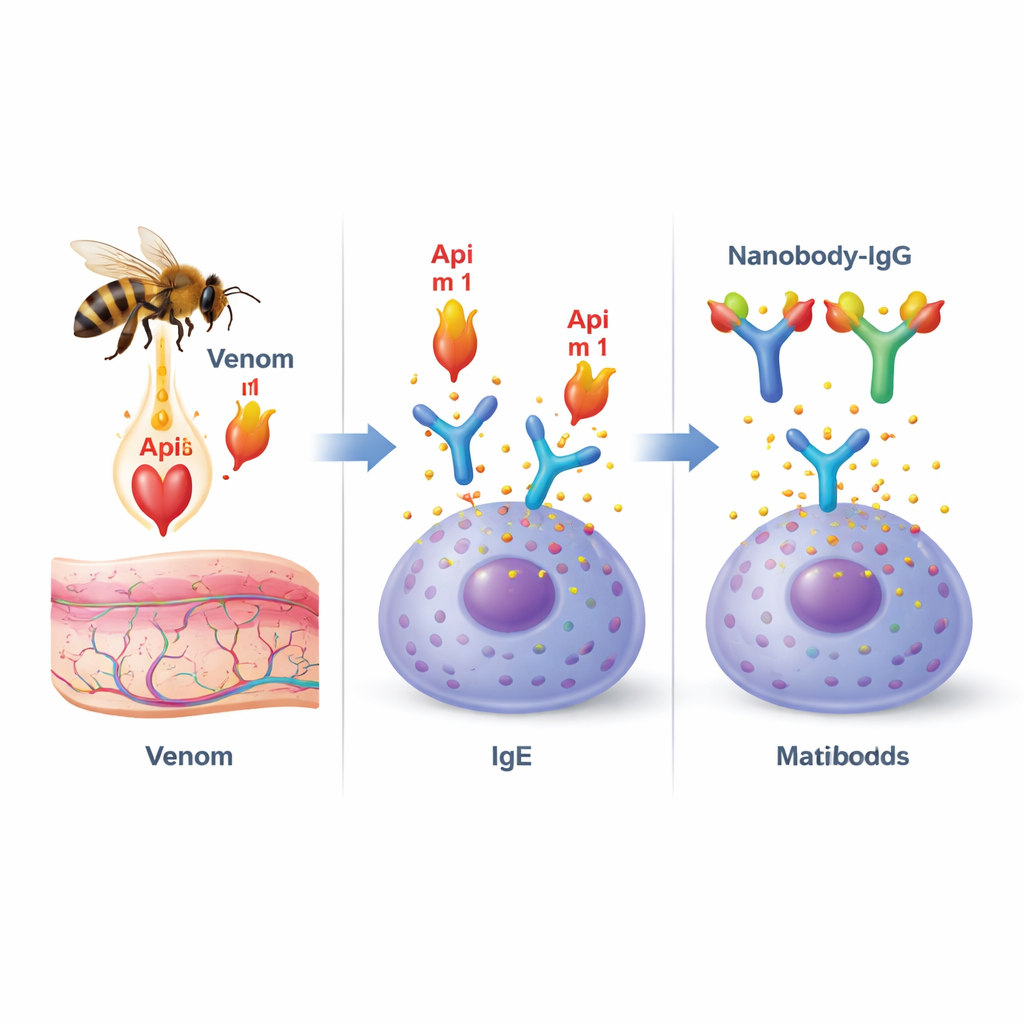
Designing tiny antibodies to block the allergen
The researchers turned to “nanobodies,” tiny, stable antibody fragments originally discovered in camelid species. Because they are small and elongated, nanobodies are particularly good at fitting into grooves and pockets on proteins. The team isolated Api m 1‑specific nanobodies from an immune library and selected two stand‑out candidates, named AM1‑1 and AM1‑4. Detailed structural work using X‑ray crystallography showed that these two nanobodies clamp onto opposite sides of Api m 1 and do not interfere with each other. One nanobody, AM1‑1, sits directly over the enzyme’s active center, suggesting it could block Api m 1’s membrane‑damaging function, while AM1‑4 docks into a separate pocket on the protein surface.
Turning nanobodies into powerful blockers
To turn these small binders into long‑lasting drugs, the scientists fused each nanobody to the tailpiece (Fc region) of human IgG1, creating larger antibody‑like molecules with better stability in the bloodstream. They also engineered a “bispecific” version that carries both AM1‑1 and AM1‑4 in a single IgG‑like molecule. Laboratory tests showed that these nanobody‑IgG fusions attach to Api m 1 with extremely high affinity and, when present, can strongly reduce the binding of IgE from honeybee‑allergic patients’ blood to Api m 1. In cell‑based assays, they diminished activation of basophils, the immune cells that normally respond vigorously when exposed to Api m 1 in allergic individuals.
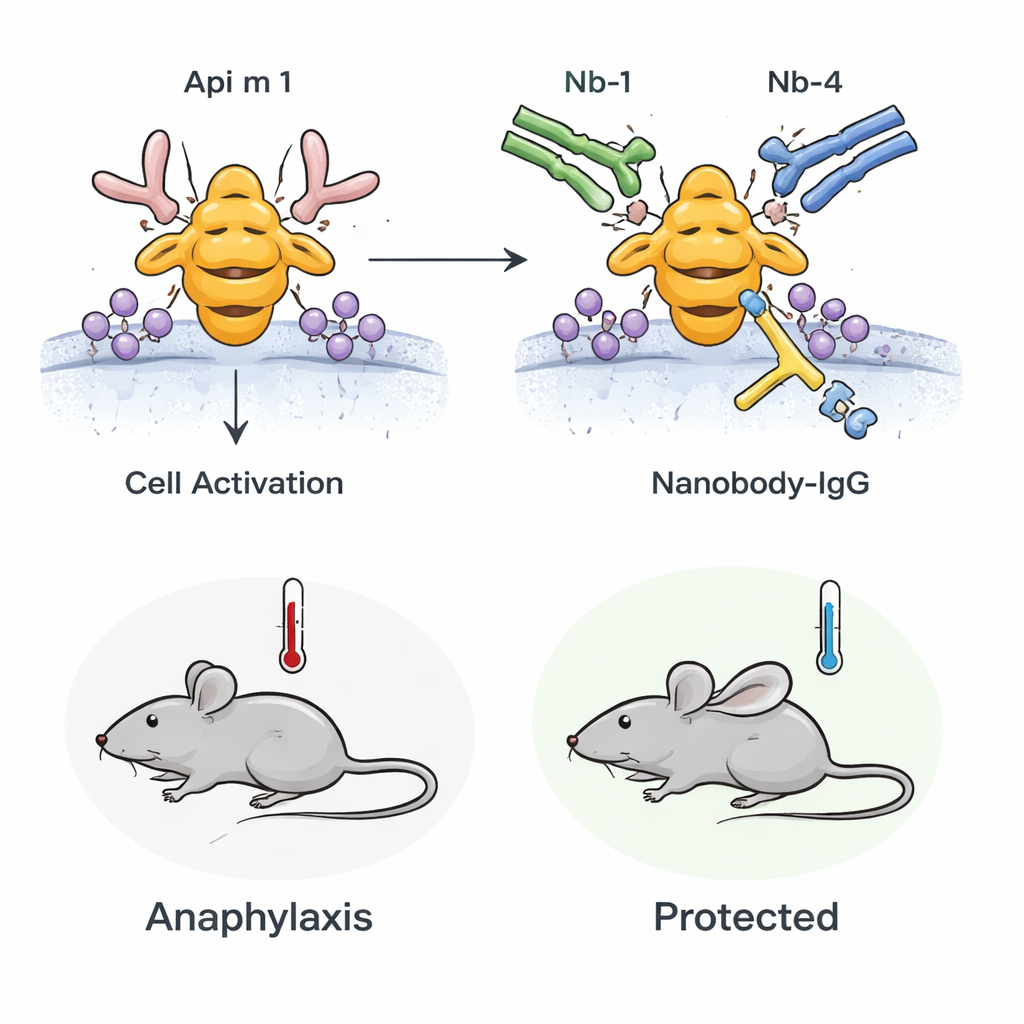
From test tube to living animals
The team next asked whether this blocking effect would hold up in a living organism. They used mice sensitized to honeybee venom so that their immune systems reacted to Api m 1 in a way that mimics human allergy. When these mice were challenged with Api m 1, they developed signs of anaphylaxis, including a drop in body temperature and markers of mast cell activation in the blood. However, if the mice were pre‑treated with the bispecific nanobody‑IgG, both the temperature drop and mast cell activation were significantly reduced. This demonstrates that pre‑existing, high‑affinity blocking antibodies can blunt the systemic allergic reaction to Api m 1 in vivo.
What this could mean for people with bee venom allergy
Taken together, the study shows that carefully designed nanobody‑based IgG molecules can simultaneously block the allergic and enzymatic actions of Api m 1, the main culprit in honeybee venom allergy. For patients, this raises the possibility of a seasonal, passive immunization: a few injections of such antibodies during the months when bee stings are most likely could provide temporary protection against severe reactions, without the long commitment and risks of traditional venom immunotherapy. While more work is needed to broaden the approach to other venom components and to test safety and effectiveness in humans, this nanobody strategy offers a precise new way to shield vulnerable individuals from dangerous bee stings.
Citation: Aagaard, J.B., Gandini, R., Ballegaard, AS.R. et al. Nanobody-based IgG simultaneously inhibit the allergenic and enzymatic activity of the dominant honeybee venom allergen. Nat Commun 17, 1814 (2026). https://doi.org/10.1038/s41467-026-69572-0
Keywords: bee venom allergy, nanobodies, passive immunotherapy, phospholipase A2, anaphylaxis prevention