Clear Sky Science · en
Cooperative clamp-mediated promoter recognition by poxviral RNA polymerase and its TBP/TFIIB-like partner
How a Virus Rewires Gene Control Inside Our Cells
Poxviruses, which include the virus historically responsible for smallpox, carry out all of their gene expression in the cytoplasm of our cells, far from the cell’s own DNA control center in the nucleus. To pull this off, they bring along their own miniature "genetic factory". This study uncovers, at atomic detail, how one such virus, Vaccinia, switches on a specific wave of its genes mid‑infection, using an unexpected clamp‑like protein that works together with the viral copying machine.
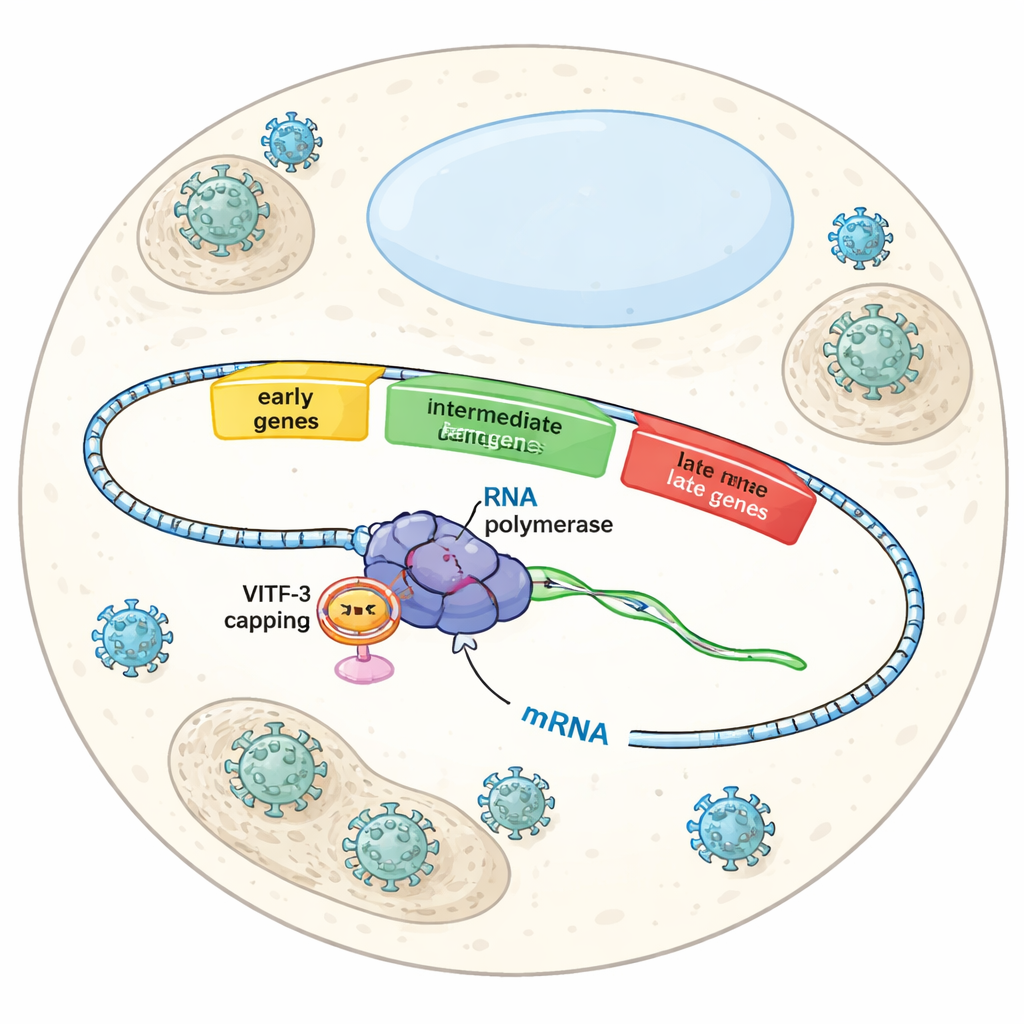
Three Waves in a Viral Takeover
When Vaccinia infects a cell, it does not turn on all of its genes at once. Instead, it follows a carefully timed script with early, intermediate, and late genes. Early genes prepare the ground for viral replication, intermediate genes help build up the machinery for making new virus particles, and late genes finish assembly and packaging. Each wave depends on the same viral RNA polymerase—the enzyme that reads DNA to make RNA—but different helper factors tell the polymerase which group of genes to activate at each stage. Until now, scientists understood early gene control fairly well, but how the virus specifically recognized and activated intermediate genes remained a mystery.
A Ring-Shaped Helper That Needs a Partner
The researchers focused on a two‑part protein called VITF‑3, known to be required for turning on intermediate genes. Many organisms, including humans, use a pair of proteins called TBP and TFIIB to guide their RNA polymerase to the right starting point on DNA. The team showed that VITF‑3 is a highly modified version of this same ancient pair. However, unlike its cellular cousins, VITF‑3 on its own does not grab onto DNA. Instead, its two subunits interlock to form a closed ring that is actually inert toward DNA unless the viral RNA polymerase is present. This was unexpected, because in most systems the TBP‑like component binds the promoter DNA first and then recruits the polymerase; here, the virus appears to have rewired the order of events.
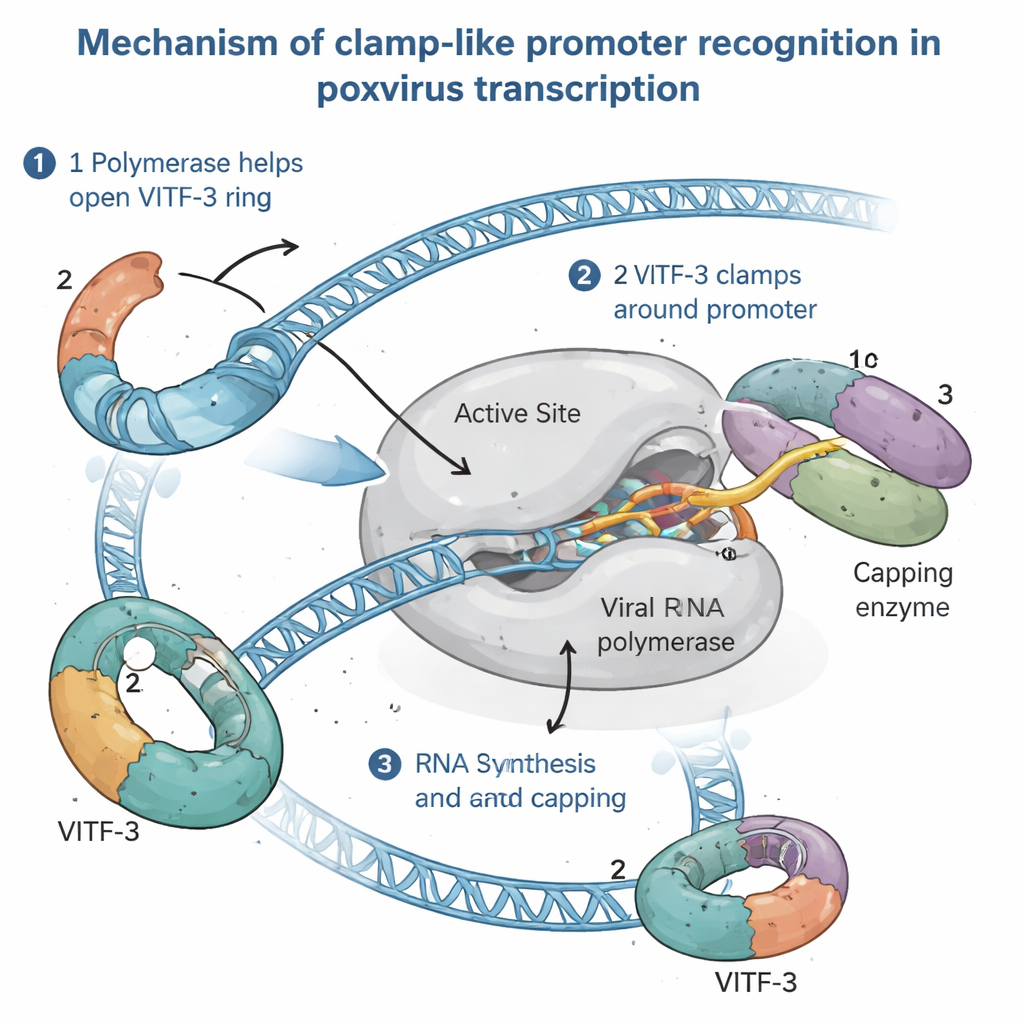
Seeing the Intermediate Transcription Machine in Action
To understand how this unusual setup works, the authors isolated the full intermediate pre‑initiation complex—the assembly poised just before RNA synthesis begins—from infected human cells. Using high‑resolution cryo‑electron microscopy, they visualized the complex at about 2.4 Ångström detail, fine enough to see side chains of amino acids and individual DNA bases. The images revealed viral RNA polymerase gripping the promoter DNA, with VITF‑3 forming a tight ring around an AT‑rich stretch of DNA just upstream of the start site. This ring sharply bends the DNA by about 90 degrees and feeds it into a cleft in the polymerase. At the same time, a viral capping enzyme—responsible for adding the protective cap to the 5′ end of viral RNA—is docked on the polymerase, ready to modify new transcripts as soon as they emerge.
A Clamp-Loader Mechanism Borrowed in Spirit
By combining structural snapshots with biochemical tests, the study proposes that the viral RNA polymerase actively loads VITF‑3 onto DNA, acting like a "clamp loader." First, the polymerase binds to an intermediate promoter and helps pry open the VITF‑3 ring. Then, together with the capping enzyme, it closes the ring around the AT‑rich region, clamping the complex in place. The DNA is melted near the start site, and the polymerase directly reads a short four‑letter signal (a TAAA motif) that marks exactly where RNA synthesis should begin. As RNA grows, it follows the same path occupied by part of VITF‑3 in the initial complex, so the emerging RNA likely pushes VITF‑3 out of the way, allowing the polymerase to leave the promoter and continue along the gene while VITF‑3 may remain behind to speed re‑use of the same promoter.
What This Means for Understanding and Targeting Poxviruses
In plain terms, this work shows that Vaccinia virus has repurposed a universal gene‑control module into a clamp that only grips DNA when its own RNA polymerase tells it to. That clever twist lets the virus use one core enzyme to run three very different transcription programs, simply by swapping stage‑specific helpers. Because the same kind of factors are conserved across many poxviruses, including human pathogens, the newly revealed clamp‑loader mechanism and the unique VITF‑3 ring offer concrete structural targets for antiviral strategies aimed at disrupting the virus’s ability to time and control its genes during infection.
Citation: Jungwirth, S., Bartuli, J., Lamer, S. et al. Cooperative clamp-mediated promoter recognition by poxviral RNA polymerase and its TBP/TFIIB-like partner. Nat Commun 17, 1648 (2026). https://doi.org/10.1038/s41467-026-69571-1
Keywords: poxvirus transcription, Vaccinia virus, RNA polymerase, promoter recognition, transcription factors