Clear Sky Science · en
Light tunes long-term threat avoidance behavior in male mice
How Light Helps Brains Remember Danger
Imagine walking through a park where you were once nearly hit by a falling branch. Even if the danger is gone, you might still skirt that spot days later. This study asks a similar question in mice: how does light, through the eyes, help the brain remember where a threat once appeared and guide future avoidance? The answer reveals an unexpected role for a special class of light-sensing cells in the eye that quietly tune long‑term decisions about risk and safety.
A Subtle Threat That Leaves a Lasting Mark
The researchers designed a simple yet powerful test they call long-term threat avoidance (LTTA). Male mice explored a square arena with a video screen overhead. At first, the screen showed only a neutral gray background while the animals freely roamed both the safe edges and the center “threat zone.” Then, just once, the screen briefly displayed a dark expanding disk—a looming shadow that mimics an approaching predator. After this single event, the mice went back to their home cages. Two days later, they returned to the same arena, now with no threat at all. Remarkably, even when the original looming shadow had been so faint that it did not trigger obvious fear behavior at the time, the animals now strongly avoided the central threat zone. This showed that the brain can form a durable memory of a mild visual danger and use it later to steer where the animal dares to walk.
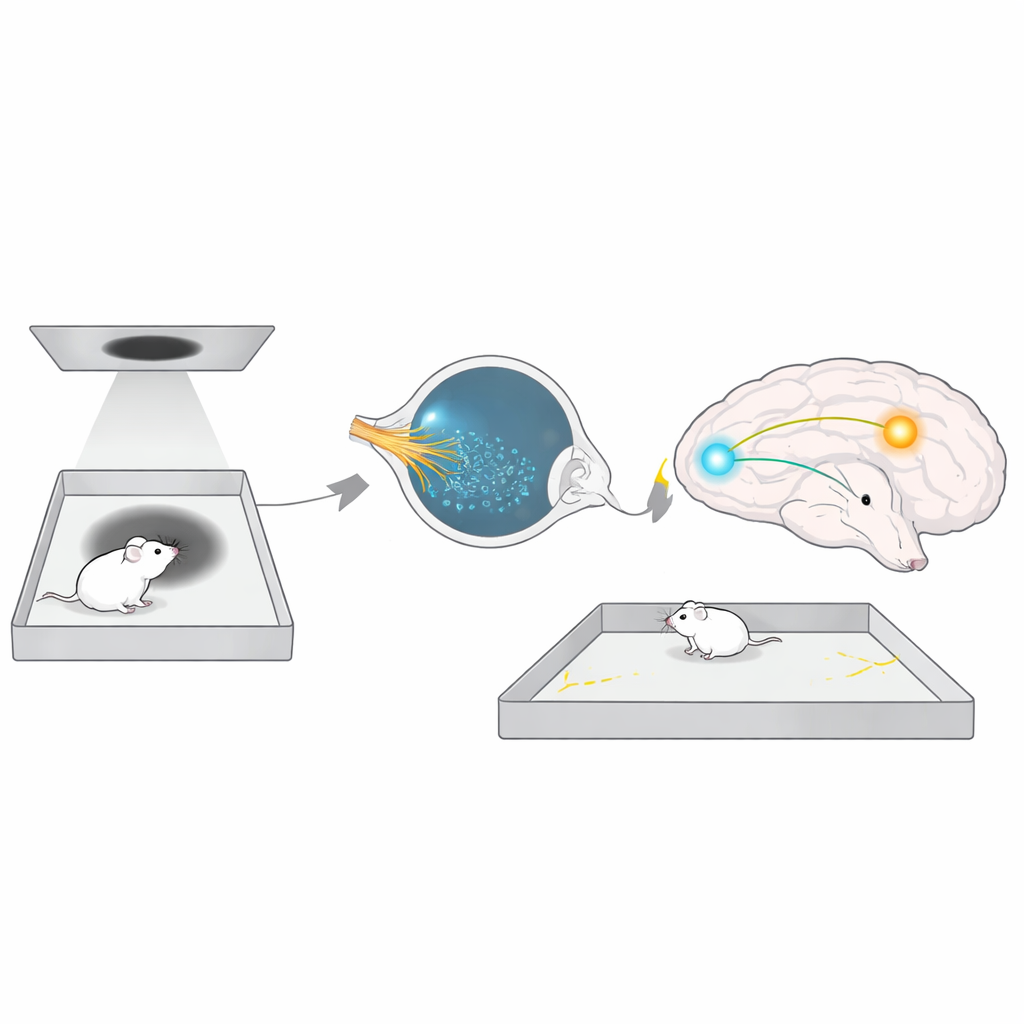
Light Is Required, But Not Just Any Light Sensor
The team next asked whether ordinary vision was enough to explain this cautious behavior, or whether other light‑sensing systems were involved. When mice were tested in complete darkness two days after the looming event, their avoidance vanished—they ventured into the center as if nothing had happened. In low or normal room light, however, avoidance returned. This pointed to a light‑dependent circuit that is active during the recall of the threat, even though no threat is present. Focusing on a known class of retinal cells called intrinsically photosensitive retinal ganglion cells (ipRGCs), which detect overall brightness rather than fine visual detail, the researchers studied mice lacking their key pigment, melanopsin. These animals detected the looming stimulus just as well as normal mice, but later failed to avoid the threat zone. Turning melanopsin off only in adulthood, or blocking the main chemical signal (glutamate) these cells send to the brain, produced the same deficit. This showed that ipRGCs and their melanopsin‑based light sensing are specifically needed to tune long‑term avoidance, not to see the threat in the first place.
A Hidden Hub Between Eye and Motivation Circuits
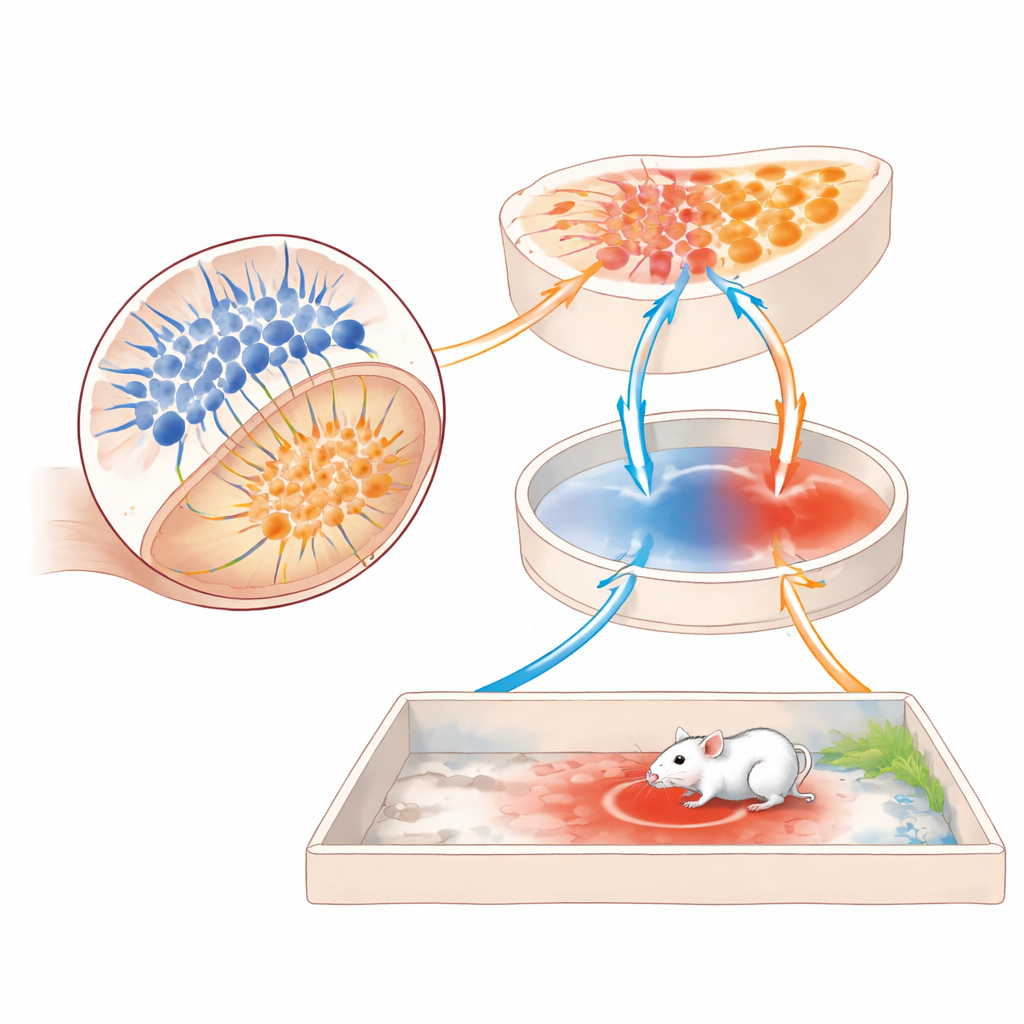
To track where in the brain these signals go, the authors searched ipRGC target regions for activity that rose only when mice showed strong threat avoidance. One small structure, the perihabenular nucleus (PHb) deep in the thalamus, stood out. In normal mice that avoided the threat zone, PHb neurons were strongly activated; in melanopsin‑deficient mice that did not avoid it, PHb activity stayed low. Silencing a specific group of inhibitory PHb cells erased avoidance, while exciting nearby excitatory PHb cells also disrupted it, suggesting that a delicate balance of inhibition and excitation in this hub is crucial. Using in‑brain calcium recordings, the team found that PHb activity rises during the later test session and dips sharply when control mice dare to enter the threat zone—an internal warning signal that is blunted when melanopsin is missing.
From Light to Action Through a Reward Center
The story does not end in the thalamus. The PHb sends signals forward to several brain regions involved in motivation and decision‑making. By selectively boosting or suppressing PHb connections, the authors discovered that projections to the nucleus accumbens—a key reward and action‑selection center—are essential for LTTA. Artificially exciting this PHb‑to‑accumbens pathway restored normal avoidance in melanopsin‑deficient mice, while blocking PHb terminals in the accumbens of healthy mice made them lose their caution and wander back into the threat zone. Notably, many classic fear and anxiety regions, such as the amygdala and midbrain escape centers, were not required in this paradigm, underscoring that this light‑guided avoidance circuit is distinct from the better‑known fear pathways.
Why This Matters for Everyday Choices
Taken together, the work outlines a new chain of influence: special light‑sensing cells in the eye feed into a thalamic hub, which in turn shapes activity in a reward‑related center so that mice remember and avoid a risky place days after a mild visual scare. This circuit operates under ordinary lighting and without pain or shock, making it a close parallel to how real‑world experiences shape our sense of where it is safe to go. By revealing that light and melanopsin‑based signals help calibrate long‑term risk‑taking, the study opens the possibility that similar pathways in humans may link everyday lighting, mood, and decisions about danger—and suggests new ways light could be used to gently nudge behavior toward safety.
Citation: Aranda, M.L., Min, E., Liu, L.T. et al. Light tunes long-term threat avoidance behavior in male mice. Nat Commun 17, 2728 (2026). https://doi.org/10.1038/s41467-026-69564-0
Keywords: threat avoidance, melanopsin, retinal ganglion cells, perihabenular nucleus, nucleus accumbens