Clear Sky Science · en
Arms races between selfish genetic elements and their host defence in termites
Hidden battles inside termite DNA
Termites are famous for building giant mounds and living in highly organized societies, but inside their cells a very different drama plays out. Their DNA is home to legions of “selfish” genetic elements that copy and move themselves around, threatening to disrupt vital genes. This study shows that termites and these jumping genes are locked in a long-running genetic arms race, revealing how tiny molecular conflicts can shape the evolution, ageing, and even the enormous genomes of these insects. 
Jumping genes as genetic troublemakers
Our genomes are not made only of useful genes. A large fraction consists of transposable elements—DNA segments that can copy and paste or cut and paste themselves to new locations. In termites, roughly half of the genome is made of such elements, far more than in many ants and bees. When these jumping genes land in or near important genes, they can damage normal cell function or cause harmful rearrangements. For that reason, biologists often compare them to parasites: they spread for their own benefit, even if it hurts the organism that carries them.
Termites as a natural testbed
Termites provide a powerful natural experiment for studying this conflict. They have high transposable element content, and earlier work showed that these elements become more active as termites age. Long-lived queens and kings appear better protected against this activity than short-lived workers, hinting that defence against jumping genes might be tied to lifespan. In this study, researchers sequenced the genomes and measured DNA methylation—a chemical mark on DNA that can switch sequences off—from seven termite species spanning about 140 million years of evolution. This allowed them to see not only how many jumping genes each species carries, but also how strongly each type is chemically silenced.
Patterns of attack and defence across evolution
The team found that the kinds and amounts of jumping genes in each species closely mirrored the termite family tree. Closely related species had similar transposable element profiles, suggesting that the elements have been evolving together with their hosts rather than drifting randomly. Strikingly, the pattern of DNA methylation on these elements also followed the termite family relationships, whereas overall methylation on the rest of the genome did not. This means that the silencing of jumping genes is a heritable trait that natural selection is actively shaping, much like immune defences against parasites.
Young invaders, strong shields; old invaders, fading threat
Diving deeper, the researchers grouped families of jumping genes by their evolutionary “age”—whether they were unique to one species or shared across many termite lineages. Younger, species-specific elements were longer, more intact, and more likely to create structural changes in the DNA, especially harmful insertions into protein-coding regions. These young elements also spread more efficiently within genomes. Correspondingly, they were marked by especially high DNA methylation, indicating that the host mounts its strongest defence against the most dangerous invaders. Older element families told the opposite story: over time they shrank into short fragments, spread less, and rarely appeared inside genes. Their methylation levels dropped back toward background levels, implying that once an element’s threat fades, the host relaxes its costly defences. 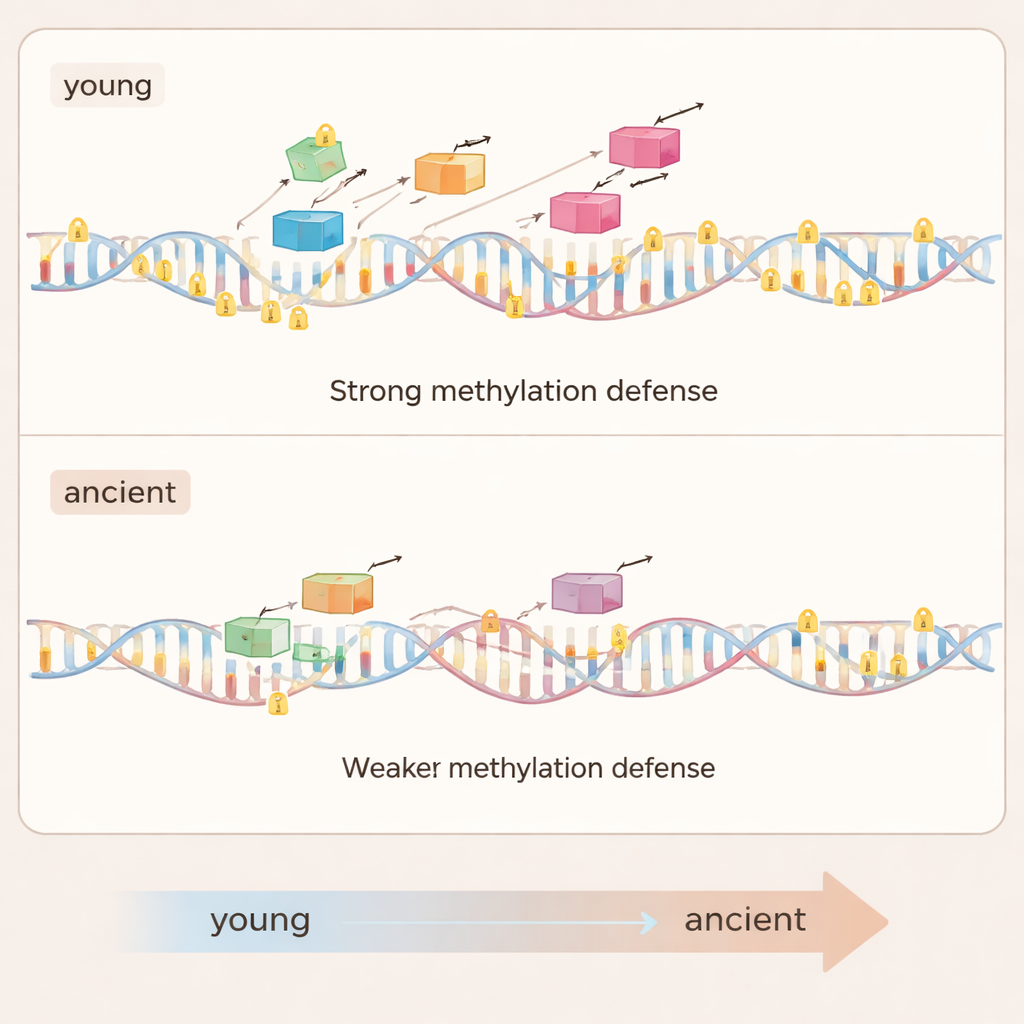
Winners, holdouts, and evolving shields
Not all old elements are harmless relics. Some of the most active jumping gene families in the main study species, the fungus-growing termite Macrotermes bellicosus, turned out to have ancient roots but had recently flared up again, suggesting repeated invasions or escape from control. At the same time, many termite genes involved in silencing transposable elements—particularly those in the piRNA pathway, which helps target troublesome sequences—showed clear signs of positive selection. In other words, these defence genes themselves are evolving rapidly, consistent with continual counter-adaptations to new or resurgent genomic parasites.
What this arms race means for termites and beyond
To a non-specialist, the takeaway is that termite genomes are not static instruction manuals but dynamic ecosystems where parasitic DNA and host defences continually clash. Young jumping genes behave like aggressive invaders, while DNA methylation and related pathways act as adaptive shields that blunt their spread and damage. Over time, many once-dangerous elements decay into mostly harmless fragments, and some may even be repurposed for beneficial functions. By exposing this molecular arms race, the study shows how conflicts at the smallest scales help drive genome size, ageing patterns, and long-term evolutionary innovation.
Citation: Qiu, B., Elsner, D. & Korb, J. Arms races between selfish genetic elements and their host defence in termites. Nat Commun 17, 1702 (2026). https://doi.org/10.1038/s41467-026-69550-6
Keywords: jumping genes, DNA methylation, termite genomes, genetic arms race, ageing