Clear Sky Science · en
Catalytic hybrid solvent regeneration in membrane vacuum processes for direct air capture
Capturing Carbon from Everyday Air
Pulling carbon dioxide directly out of the air is one of the tools scientists hope to use to slow climate change, but today it costs a lot of energy. This study explores how to make a particular kind of direct air capture system much less energy-hungry by rethinking both the liquid that catches CO2 and the way that liquid is cleaned and reused. The result is a system that can regenerate its CO2‑laden solvent at lower temperatures and with far less heat, bringing direct air capture a step closer to large-scale, climate‑relevant deployment.
Why Cleaning the Capture Liquid Is So Hard
Most existing plants that scrub CO2 from gases rely on liquids that chemically bind the gas. The challenge is that once those liquids are full, they must be heated to high temperatures so the CO2 lets go, after which the liquid can be reused. For air, where CO2 is extremely dilute, this energy bill becomes especially painful. Traditional solvents also need temperatures of around 120–140 °C to be refreshed, which strains equipment and can shorten the lifetime of the fluid. The team behind this work set out to redesign this “clean‑up” step so it could run at much lower temperatures while still releasing large amounts of CO2.
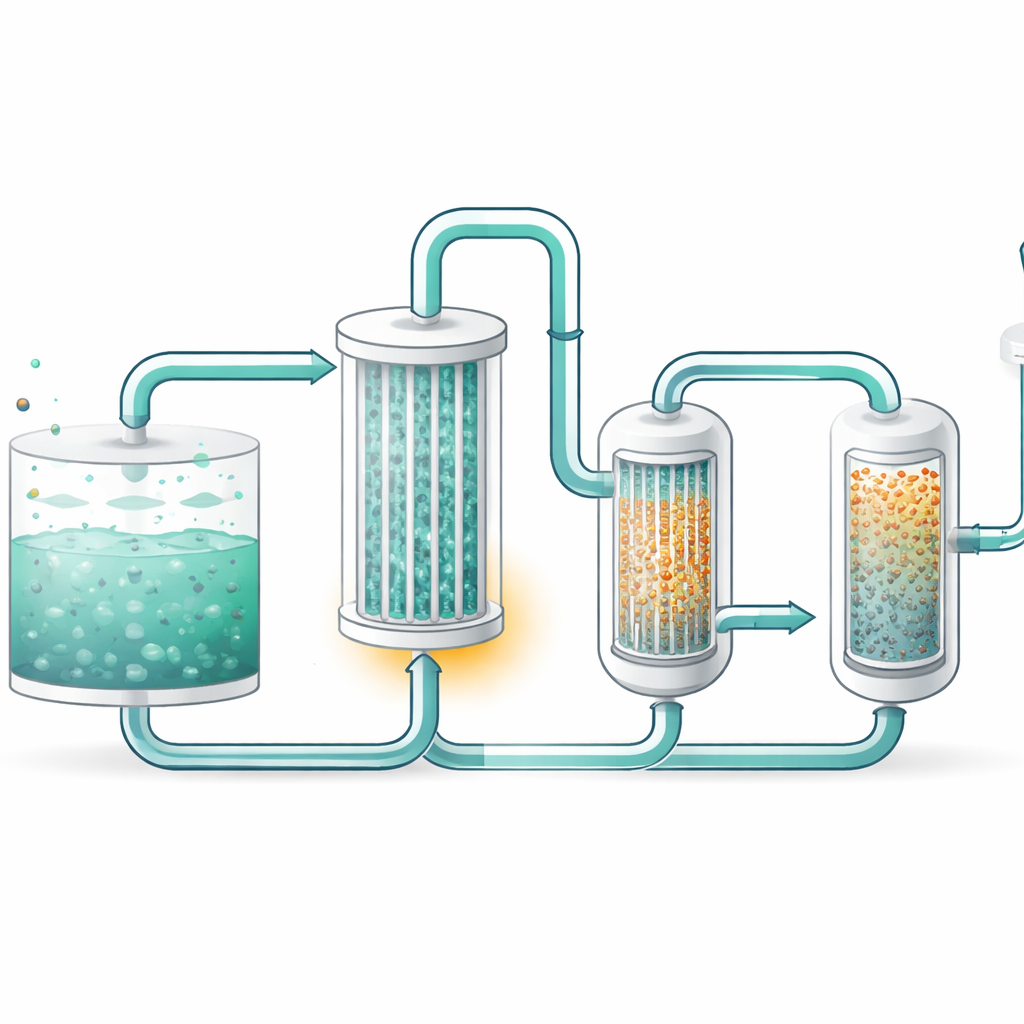
A Gentler Way to Regenerate the Liquid
The researchers focused on a technology called membrane vacuum regeneration. Here, warm solvent flows past a bundle of tiny hollow fibers. CO2 and some water vapor slip through the fiber walls into a low‑pressure side, leaving the cleaned solvent behind. By carefully choosing and testing three different membrane modules, they identified a configuration that allowed strong CO2 removal while limiting water loss: a hollow fiber module with a very thin protective coating. This design balances how easily CO2 moves with how strongly the membrane resists being flooded by liquid, an issue that can otherwise reduce performance over time.
Boosting Performance with Smart Solvents and Catalysts
The second innovation lies in both the liquid recipe and the solid helper particles it passes through. Instead of relying on a single ingredient, the team blended two amino acid–based salts, taurinate and sarcosinate, which are attractive because they are low‑volatility, resistant to degradation, and relatively benign. By tuning their mixture, they discovered that a blend containing three parts potassium taurinate and one part potassium sarcosinate could absorb more CO2 from air and then give it up more readily during regeneration. On top of that, they added a finely engineered solid catalyst made from iron‑doped sulfated zirconia dispersed on porous silica. As the warm solvent passes through a fixed bed of these particles before reaching the membrane, chemical sites on the solid speed up the unbinding of CO2 from the liquid, raising the CO2 flow and allowing more gas to be stripped out in the same time.
Finding the Sweet Spot for Energy Savings
Through dozens of experiments, the authors adjusted how the catalyst was built and how much of it was used. Silica proved to be a better support than alumina, and a one‑to‑one ratio of active material to silica particles gave the best performance: too little and there were not enough active sites, too much and pores became clogged. They also found that loading about nine percent catalyst by weight into the fixed bed gave nearly the maximum benefit before further additions stopped helping much. With the optimized hybrid solvent and catalyst together inside the low‑temperature membrane system running at just 90 °C, the amount of heat required to regenerate the liquid dropped dramatically compared with a common benchmark solvent, potassium glycinate.
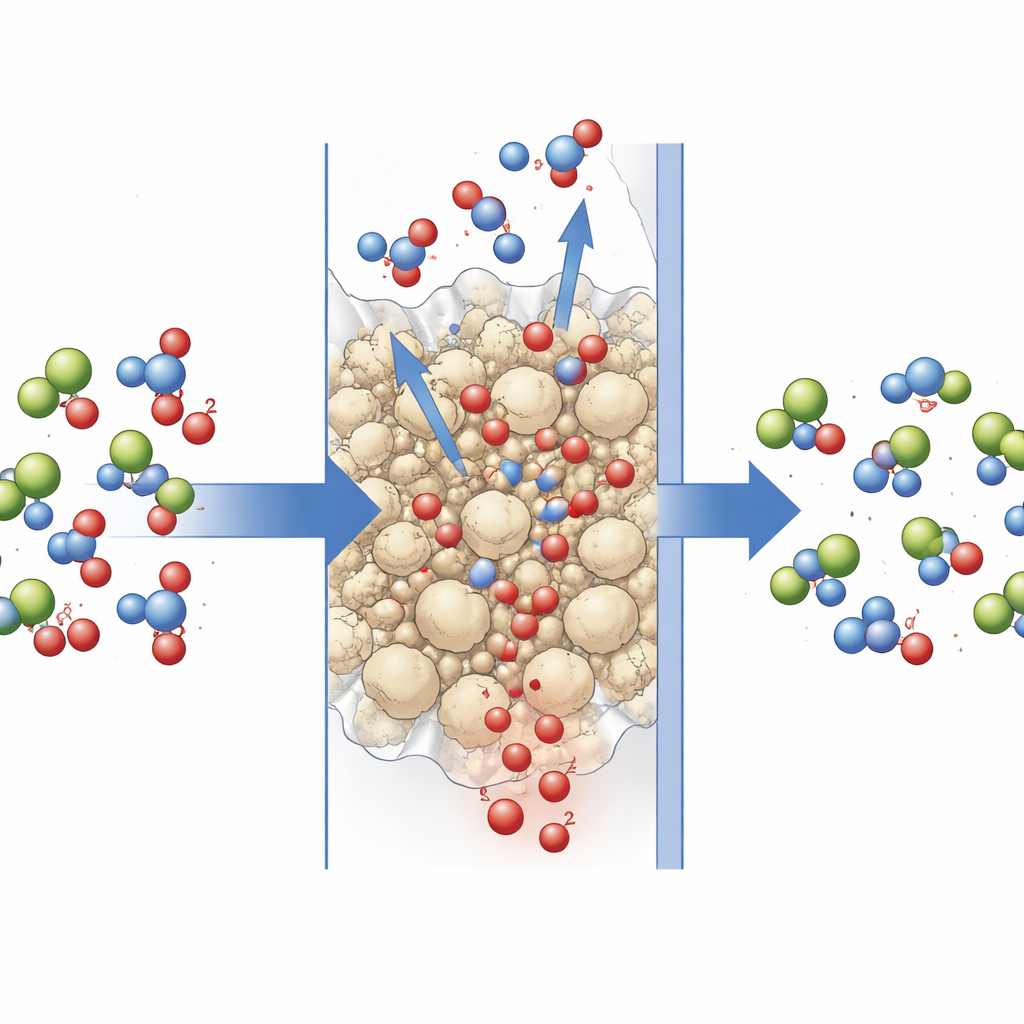
A Leaner Path to Pulling CO2 from Air
When all the pieces were combined—the tuned hollow‑fiber module, the hybrid amino acid solvent, and the carefully engineered solid catalyst—the system cut its thermal energy use for the regeneration step by about two‑thirds. In practical terms, the heat demand fell to around 2.6 gigajoules per ton of CO2 for the sensible heat portion, and to an estimated total of 6.5 gigajoules per ton when other contributions are included, comparable to well‑known direct air capture designs. For non‑experts, the key message is that by co‑optimizing the liquid, the solid helper, and the membrane layout, the authors show a credible route to making direct air capture less energy‑intensive and more compatible with low‑temperature, renewable heat sources, improving its prospects as a long‑term climate tool.
Citation: Momeni, A., Anisi, H., McQuillan, R.V. et al. Catalytic hybrid solvent regeneration in membrane vacuum processes for direct air capture. Nat Commun 17, 2247 (2026). https://doi.org/10.1038/s41467-026-69542-6
Keywords: direct air capture, carbon removal, membrane separation, catalytic regeneration, hybrid solvents