Clear Sky Science · en
Phosphoglycerate dehydrogenase-mediated serine reprogramming aggravates macrophage hyperinflammation in murine Pseudomonas aeruginosa pneumonia
When the Body’s Defenders Go Too Far
Pneumonia caused by the bacterium Pseudomonas aeruginosa is notoriously hard to treat, especially in people with weakened lungs. Antibiotics target the microbe, but this illness is often deadly not only because of the bacteria themselves, but because the body’s own immune response spirals out of control. This study uncovers how a single metabolic “switch” inside immune cells can push them from helpful to harmful—and suggests that tweaking diet or cell metabolism could calm life‑threatening lung inflammation.
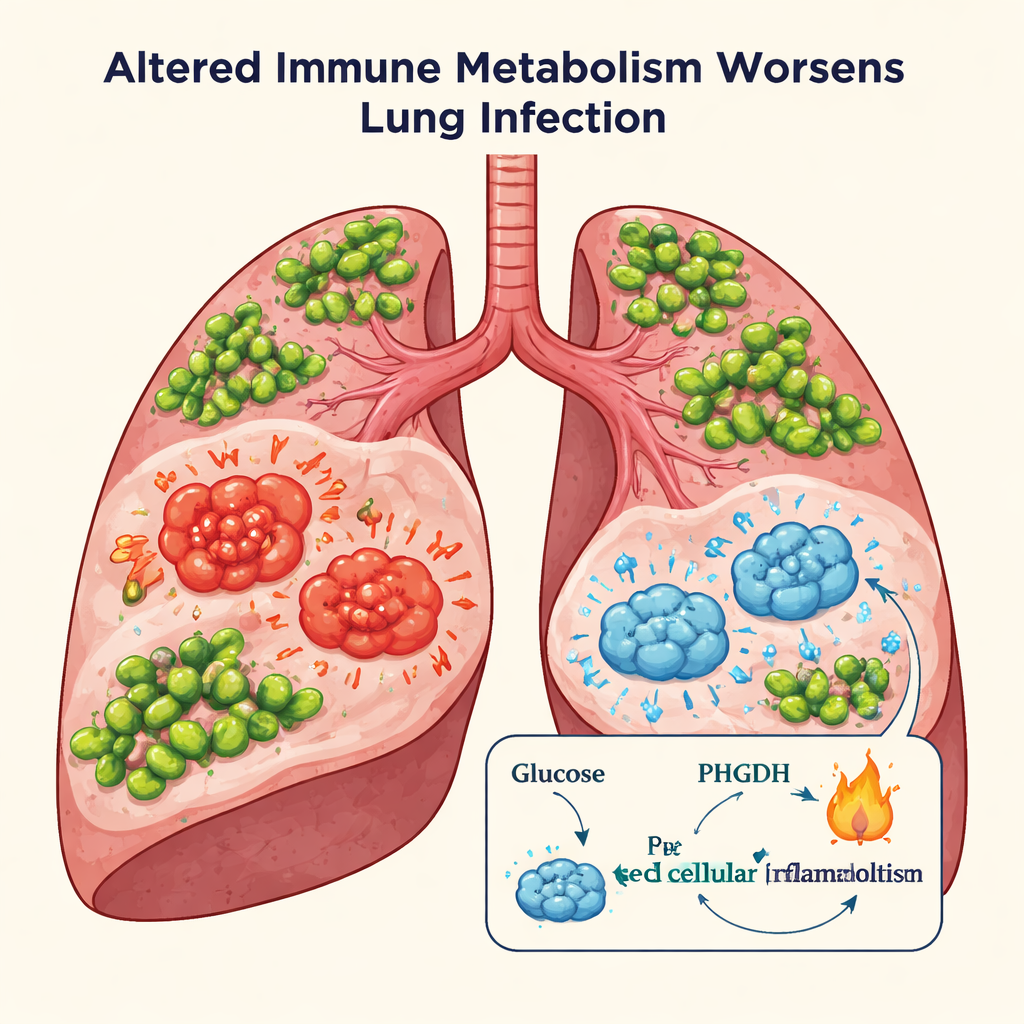
A Dangerous Dance Between Germ and Immune System
Pseudomonas aeruginosa is a common Gram‑negative bacterium that can invade the lower airways and trigger severe pneumonia. Normally, lung‑resident immune cells called macrophages patrol the air sacs, gobbling up invaders and sounding the alarm with chemical signals. But when the bacterial load is high, these cells recruit large numbers of backup macrophages from the bloodstream. The result can be a storm of inflammatory molecules that damage fragile lung tissue, impair repair processes, and, in severe cases, lead to respiratory failure and death. The authors asked whether the internal metabolism of these macrophages might be driving this harmful overreaction.
A Metabolic Enzyme in the Hot Seat
The team focused on an enzyme called phosphoglycerate dehydrogenase (PHGDH), a key entry point into the pathway cells use to make the amino acid serine from glucose. In mouse models of Pseudomonas lung infection, PHGDH levels and activity rose sharply in the lungs, especially in macrophages. When mice were treated with a small‑molecule PHGDH inhibitor, or when the gene for PHGDH was deleted specifically in myeloid cells (which include macrophages), the animals fared better: they survived longer, had less lung injury, carried fewer bacteria, and produced lower amounts of potent inflammatory cytokines such as interleukin‑6 and interleukin‑1β. At the cellular level, blocking PHGDH shifted macrophages away from a strongly pro‑inflammatory state without boosting the opposite, wound‑healing phenotype.
Fueling the Fire: Serine and an Overactive Signal Circuit
Digging deeper, the researchers showed that infection reprograms macrophages to channel more glucose into new serine production, and that this serine in turn worsens inflammation. Extra serine made macrophages secrete more inflammatory cytokines and express more surface markers of activation, while serine‑restricted diets protected infected mice and lowered bacterial counts. Mechanistically, serine fed into a “one‑carbon” metabolic cycle that helps supply the cell’s methyl groups, chemical tags used in epigenetic regulation. This boosted a specific histone mark, H3K27 trimethylation, near the gene for a braking protein called DUSP4, which normally helps turn off the ERK1/2 signaling pathway. When DUSP4 was suppressed, ERK1/2 stayed switched on, amplifying inflammatory signals. Inhibiting PHGDH or lowering serine reversed this chain, restored DUSP4, and reduced ERK1/2 phosphorylation.
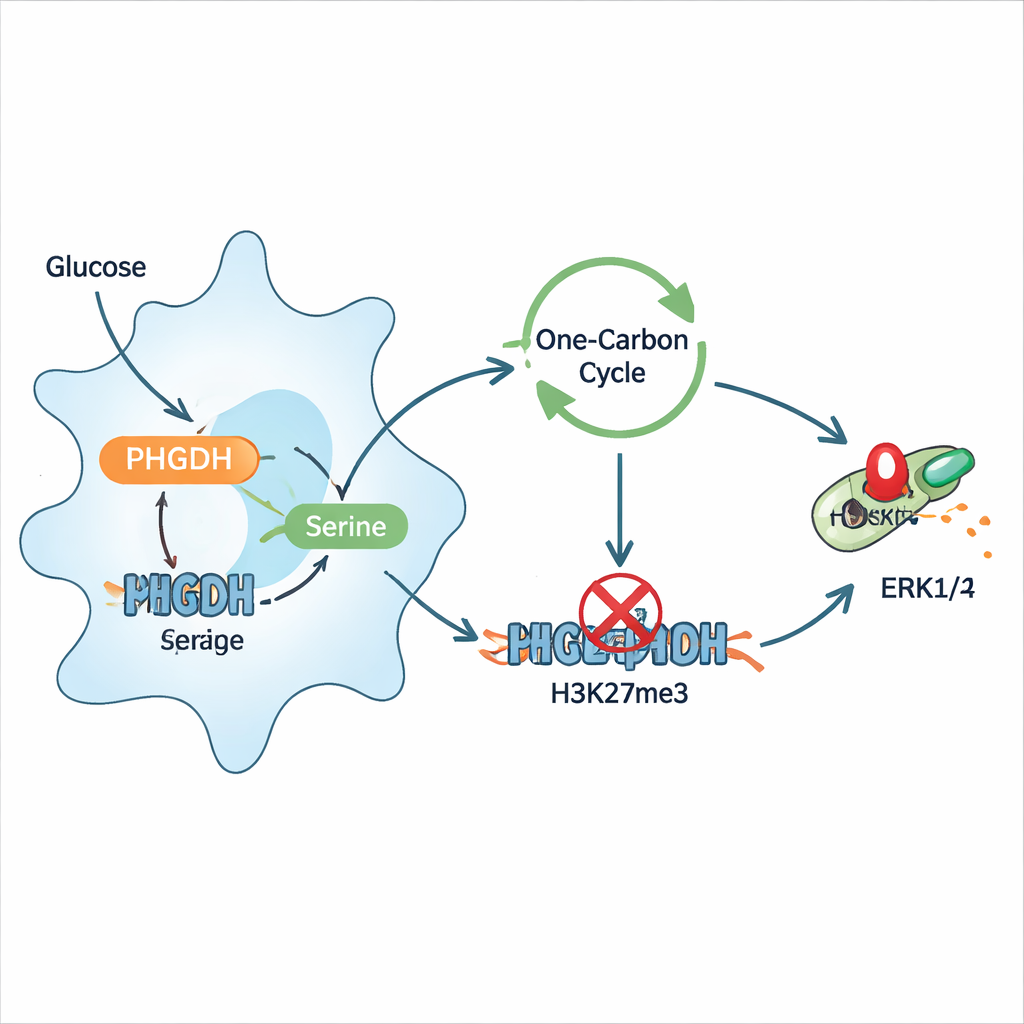
Rebalancing Macrophages Without Blunting Defense
Importantly, dialing down PHGDH did not simply paralyze the immune system. In fact, macrophages lacking PHGDH were better at engulfing and killing Pseudomonas early after infection, even while they produced fewer damaging cytokines. Transferring these PHGDH‑deficient macrophages into normal mice improved outcomes after bacterial challenge, underscoring that the benefits came from reprogramming the host response rather than directly targeting the microbe. The work also hints that serine‑rich diets may unintentionally favor hyperinflammation during severe bacterial pneumonia, whereas controlled serine restriction might offer a new way to tame excessive lung damage.
What This Means for Treating Hard‑to‑Cure Pneumonia
In accessible terms, this study reveals that some macrophages in Pseudomonas pneumonia burn sugar in a way that makes extra serine, and that this metabolic detour locks their inflammatory machinery in overdrive. By blocking the enzyme PHGDH or limiting serine, the researchers were able to calm this overreaction, protect lung tissue, and still allow immune cells to clear bacteria. While the work is in mice and far from clinical use, it points to a promising idea: pairing traditional antibiotics with drugs or dietary approaches that subtly adjust immune‑cell metabolism could turn a destructive immune fire into a controlled, bacteria‑fighting flame.
Citation: Chen, R., Zeng, R., Shi, M. et al. Phosphoglycerate dehydrogenase-mediated serine reprogramming aggravates macrophage hyperinflammation in murine Pseudomonas aeruginosa pneumonia. Nat Commun 17, 1944 (2026). https://doi.org/10.1038/s41467-026-69539-1
Keywords: Pseudomonas pneumonia, macrophage metabolism, serine biosynthesis, lung inflammation, PHGDH