Clear Sky Science · en
Ustilago maydis disrupts carbohydrate signaling networks to induce hypertrophy in host cells
When Crop Diseases Turn Cells into ‘Tumors’
Maize (corn) is one of the world’s most important food and feed crops, and it has an unusual enemy: the smut fungus Ustilago maydis, which causes swollen, tumor‑like growths on leaves and cobs. Beyond the striking appearance, these growths quietly reroute the plant’s sugars away from normal growth and defense and into fungal profit. This study uncovers how a single fungal protein helps flip the plant’s internal energy switches, turning ordinary leaf cells into bloated, starch‑stuffed factories that feed the pathogen.
A Fungus That Rewires Plant Growth
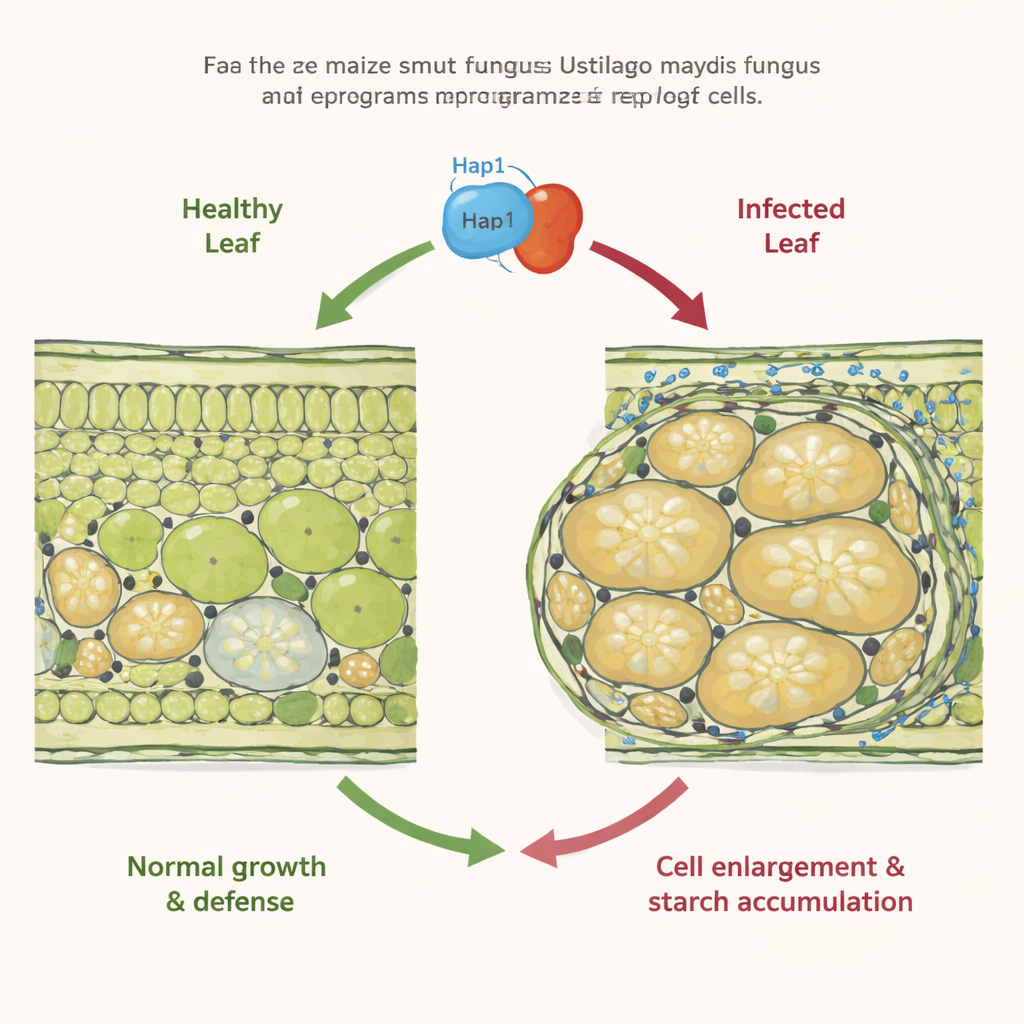
Most smut fungi quietly spread through cereal crops and mainly deform the flowers. Ustilago maydis is different: it can form local tumors on almost any above‑ground part of maize. Earlier work showed that in infected leaves, some cells start dividing too much (hyperplasia), while neighboring mesophyll cells, which normally handle photosynthesis, instead swell dramatically (hypertrophy) and stop dividing. These hypertrophic cells also fill with starch granules in places where C4 plants like maize usually do not store starch. The molecular triggers behind this extreme cell enlargement and sugar hoarding, however, were unknown.
Finding the Fungal Trigger for Cell Swelling
The researchers focused on fungal “effector” proteins—molecules secreted into plant tissue that reshape host biology. From a list of effectors active specifically in hypertrophic tumor cells, they used CRISPR‑Cas9 to knock out several candidate genes in U. maydis. One gene, UMAG_02473, stood out: mutants lacking it caused far fewer tumors and showed plant cells with smaller nuclei and much less starch in leaf mesophyll. Because enlarged nuclei are a hallmark of endoreduplication—cells copying their DNA without dividing—the team concluded that this effector, which they named Hap1 (hypertrophy‑associated protein 1), is crucial for pushing mesophyll cells into a growth‑without‑division program and for driving starch build‑up.
Hijacking the Plant’s Energy Master Switch
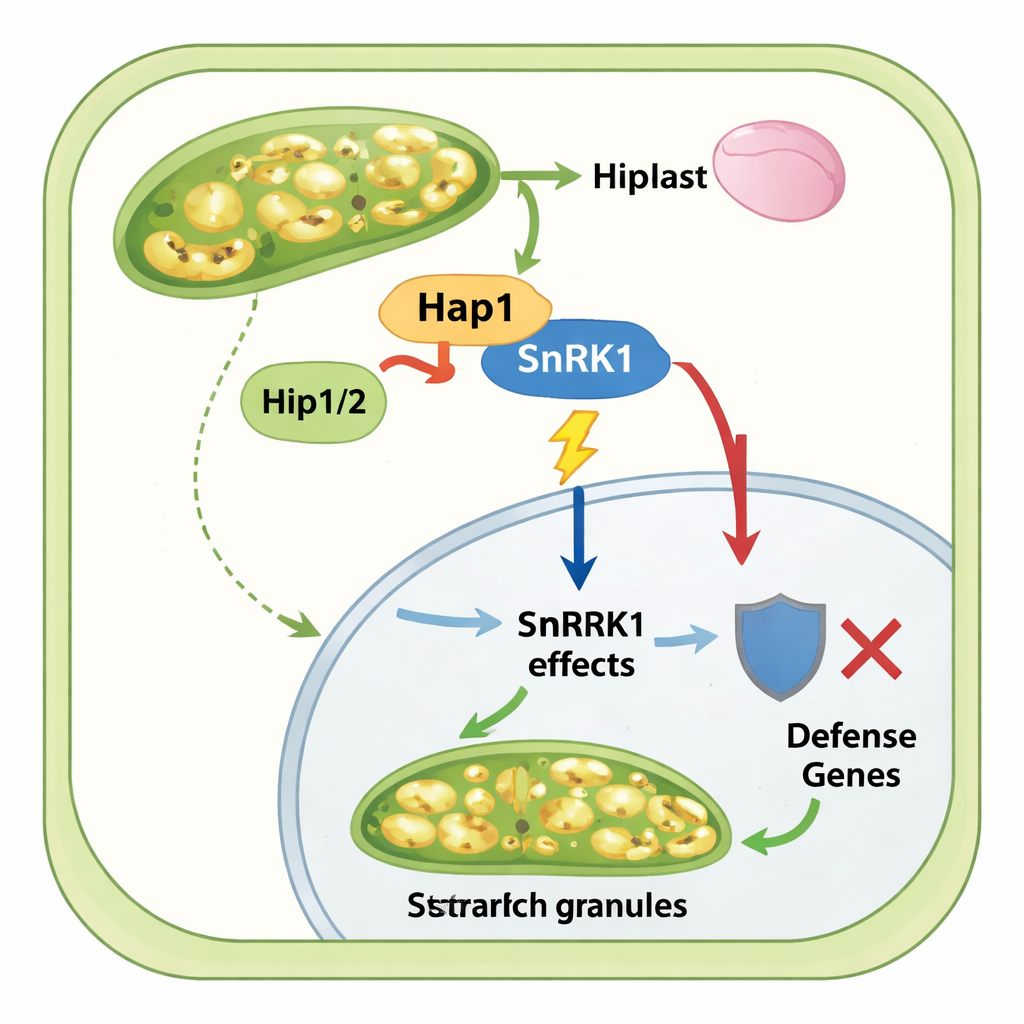
To understand how Hap1 reshapes maize metabolism, the scientists looked for its plant targets. They showed that Hap1 is delivered into host cells and accumulates in both cytoplasm and nucleus. Using protein pull‑downs and mass spectrometry, they found that Hap1 binds to the maize protein SnRK1, a central energy sensor that normally responds to stress and low energy by turning on sugar‑releasing pathways, dialing down energy‑expensive synthesis (like starch formation), and boosting defense responses. Further tests confirmed that Hap1 physically interacts with the catalytic subunits of SnRK1. When Hap1 was present, phosphorylation patterns on SnRK1 components and known SnRK1 target proteins changed, and a direct kinase assay showed that Hap1 dampens SnRK1’s ability to phosphorylate a standard test substrate. In short, Hap1 interferes with the plant’s energy “thermostat,” weakening its usual response to stress and nutrient shortage.
Helper Effectors and a Sugar‑Rich Tumor Niche
The story does not end with Hap1 alone. The team discovered two additional fungal effectors, Hip1 and Hip2, that specifically interact with Hap1 and are also expressed in hypertrophic tumor cells. Although deleting these helpers had limited impact on overall disease severity, biochemical experiments showed that Hip1 and Hip2 bind both Hap1 and the SnRK1 complex, help maintain Hap1 protein levels, and are required for Hap1 to efficiently pull down SnRK1 from infected maize tissue. Phosphoproteomics and gene‑expression data together showed that when Hap1 is present, maize tumors upregulate starch‑biosynthesis genes and enzymes, while genes tied to plant defense and normal protein‑making machinery are subdued. Without Hap1, starch accumulation in mesophyll cells drops sharply, and stress‑ and defense‑related signaling pathways become more active.
What This Means for Maize and Its Defenses
From a lay perspective, this work shows that Ustilago maydis succeeds not just by damaging tissues, but by skillfully tampering with the plant’s internal energy controls. The Hap1 effector, bolstered by its partners Hip1 and Hip2, appears to press “mute” on SnRK1, a key guardian that usually conserves energy and supports immune defenses. With this safeguard weakened, maize mesophyll cells stop behaving like normal green factories and instead swell, copy their DNA without dividing, and hoard starch. These tumor‑like cells become nutrient‑rich islands that nourish the fungus. Understanding this fungal strategy opens the door to breeding or engineering maize varieties in which SnRK1 or related pathways are less vulnerable, helping crops resist tumor‑forming infections without sacrificing growth.
Citation: Lee, Y.J., Zhang, D., Stolze, S.C. et al. Ustilago maydis disrupts carbohydrate signaling networks to induce hypertrophy in host cells. Nat Commun 17, 1990 (2026). https://doi.org/10.1038/s41467-026-69532-8
Keywords: maize smut, plant immunity, fungal effectors, starch metabolism, SnRK1 signaling