Clear Sky Science · en
A synthetic cell with integrated DNA self-replication and lipid biosynthesis
Building Life from Scratch
What makes a living cell more than just a bag of molecules? One answer lies in its ability to copy its genetic instructions and build its own protective membrane. This study takes a major step toward recreating that trick in the lab. The authors design tiny bubbles of fat, known as liposomes, that can read a piece of DNA, copy that DNA, and make new membrane material—all inside the same miniature compartment. Their work edges us closer to artificial cells that can grow, adapt, and perhaps one day evolve on their own.
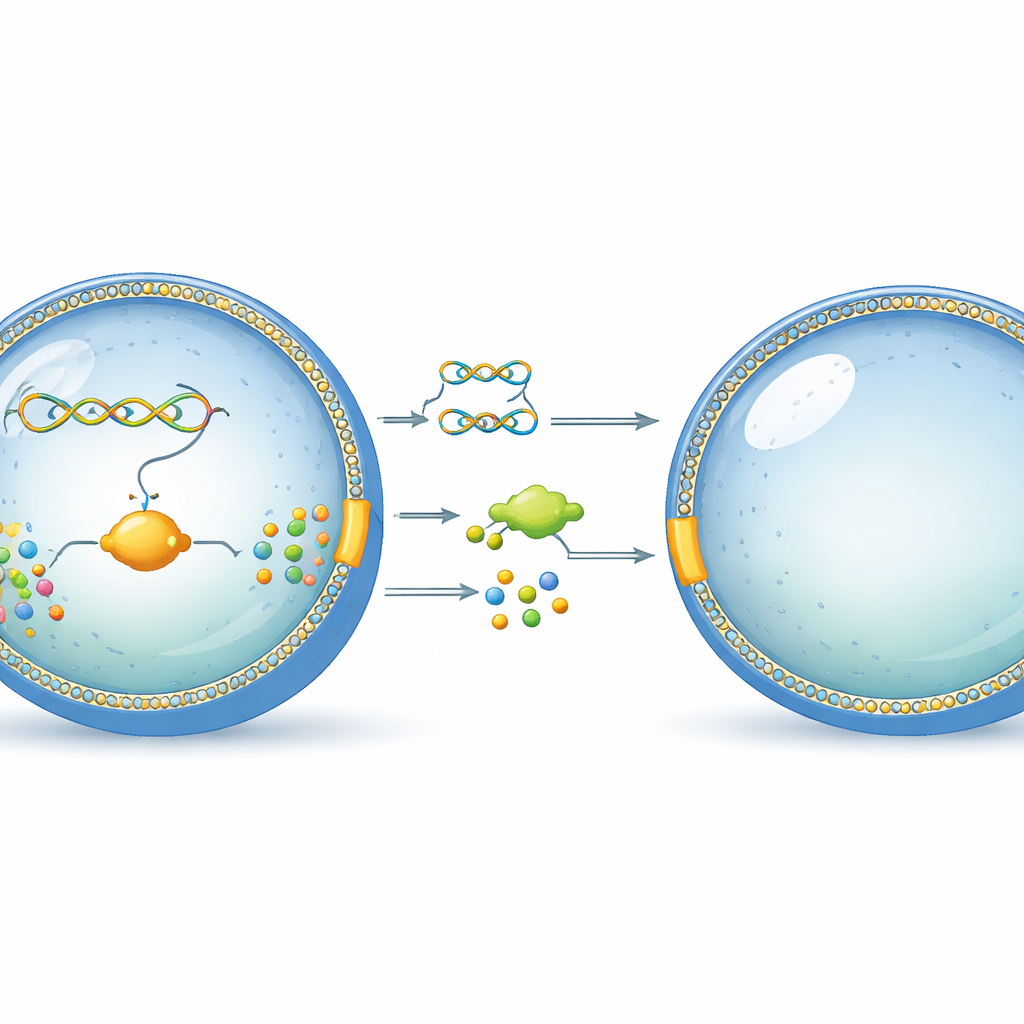
A Tiny Bubble That Acts Like a Cell
The researchers start with simple, cell-sized bubbles made of phospholipids, the same type of molecules that form the outer shell of real cells. Into these bubbles they pack a carefully designed DNA strand and a set of purified cellular machines that can read DNA and make proteins. This setup, called a cell-free expression system, acts like the stripped-down core of a living cell, able to turn genetic information into working molecules without any living organism present. The key idea is to place everything inside the liposome so that the genetic program and its products stay together, much like in a natural cell.
A Custom DNA Program with Two Jobs
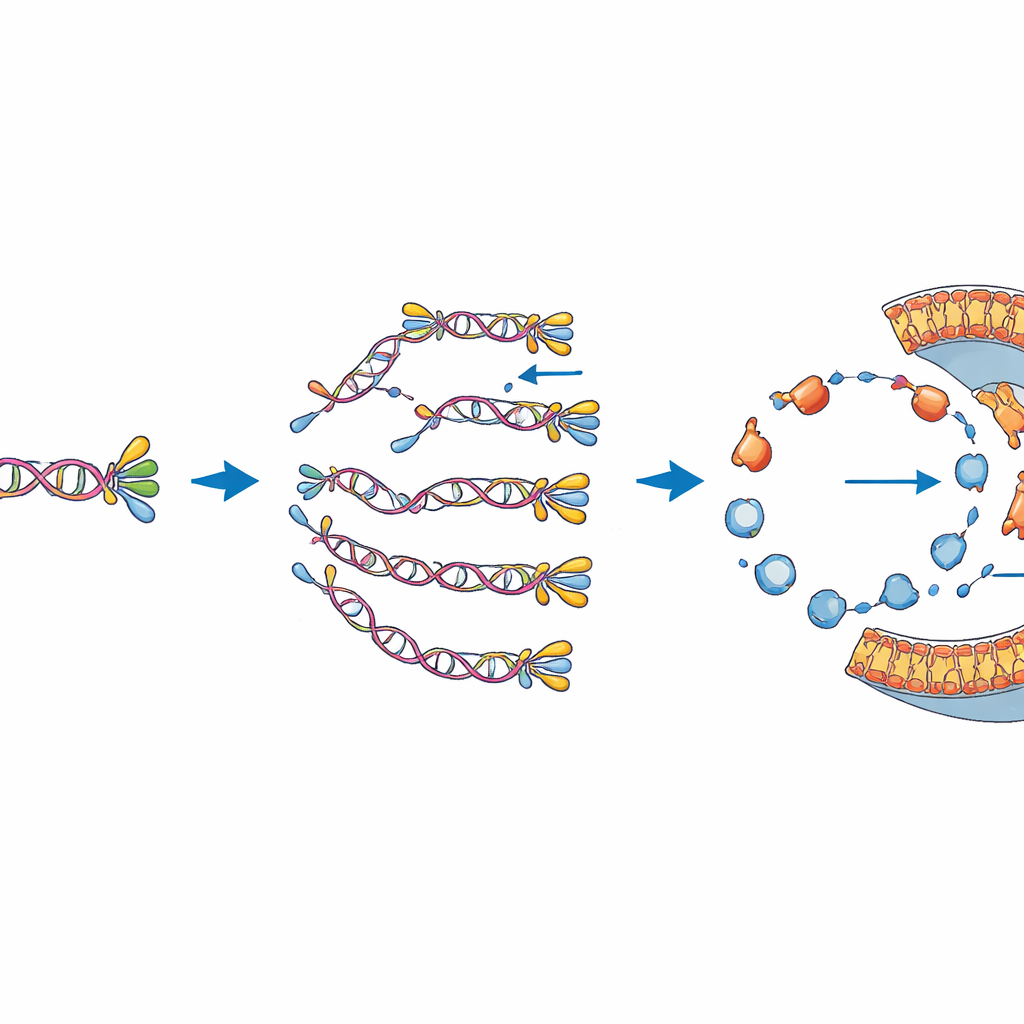
At the heart of their synthetic cell is a custom-built DNA molecule the authors call DNArep-PLsyn. This DNA carries instructions for six proteins. Two of them come from a virus that infects bacteria and together are able to copy the DNA itself, providing a built-in self-replication module. The other four come from the gut bacterium E. coli and form a chain of reactions that turns simple starting materials into a specific phospholipid used in membranes. To assemble this unusual genome, the team had to stitch together pieces of DNA in test tubes and in yeast cells, then convert the result into a linear DNA strand that the viral copying machinery can recognize and replicate.
Making and Testing Synthetic Activity
Once the DNA and protein-making machinery are sealed inside the liposomes, the bubbles are warmed to different temperatures and left to run. The team then checks what happened using fluorescent markers: one dye lights up when it binds DNA, revealing how much DNA is present, and another attaches to the new phospholipid if it is produced and inserted into the membrane. Using flow cytometry and high-resolution microscopy, they can analyze tens of thousands of individual vesicles. They find that many bubbles successfully copy the genome, many others make new membrane building blocks, and a smaller but significant fraction manage to do both at once. Additional tests using DNA quantification and mass spectrometry confirm that the full-length genome is amplified and that new phospholipid molecules are genuinely synthesized, though in modest amounts.
Balancing Two Essential Tasks
The authors then probe how these two functions influence one another. By turning the DNA-copying or membrane-making chemistry on and off through their required ingredients, they show that each process can operate largely without disturbing the other. However, when both modules are encoded on the same DNA, the membrane-synthesis side proves more fragile: fewer vesicles show this activity compared with vesicles carrying only the lipid-related genes. Similarly, the combined genome does not copy as efficiently as a smaller version that contains only the DNA-replication genes. This suggests that even in this stripped-down system, there is competition for shared resources and physical space on the DNA, echoing trade-offs seen in real cells.
Preparing for Evolution in the Test Tube
To move beyond a one-off demonstration, the team designs their system so it can, in principle, be improved through evolution. They generate cleaner, more reliable DNA versions using plasmids grown in yeast and bacteria, which boosts the fraction of fully functional synthetic cells. They also show that it is possible to encapsulate, select, and recover genomes from vesicles that perform both DNA copying and membrane synthesis. This sets the stage for future cycles in which slightly different genomes compete, and those that perform better are enriched and copied.
Why This Matters for Understanding Life
In everyday terms, the researchers have built a microscopic bubble that can read its own recipe, make more copies of that recipe, and use it to patch and extend its outer skin. Although these synthetic cells cannot yet grow large or divide like living organisms, the work demonstrates that core features of life—information storage, self-copying, and basic self-construction—can be combined in a simple, controllable package. This lays important groundwork for exploring how life might have started from nonliving chemistry and for engineering artificial cells that carry out useful tasks, from smart drug delivery to self-renewing tiny factories.
Citation: Restrepo Sierra, A.M., Ramirez Gomez, F., van Tongeren, M. et al. A synthetic cell with integrated DNA self-replication and lipid biosynthesis. Nat Commun 17, 2727 (2026). https://doi.org/10.1038/s41467-026-69531-9
Keywords: synthetic cells, DNA self-replication, lipid biosynthesis, artificial life, bottom-up biology