Clear Sky Science · en
FOXF2 regulates pericyte–endothelial signaling required for vascular homeostasis after neonatal hyperoxic lung injury
Why newborn lungs need careful protection
For babies born too early, breathing can be a struggle. Many require extra oxygen to survive, but that lifesaving therapy can also injure their fragile lungs and blood vessels, contributing to a chronic condition called bronchopulmonary dysplasia. This study explores a little-known group of support cells wrapped around tiny lung blood vessels and uncovers a key genetic switch that helps those cells protect and repair the newborn lung after oxygen injury.
The hidden helpers around lung blood vessels
The air sacs in the lung, where oxygen enters the blood, are lined with a dense web of capillaries. Wrapped around many of these capillaries are pericytes, support cells that help keep vessels stable, guide new vessel growth, and maintain a tight barrier so fluid does not leak into the air spaces. The authors focused on a gene called FOXF2, a transcription factor that controls which other genes are turned on or off inside pericytes. By analyzing single-cell RNA data from developing mouse lungs, they found that Foxf2 activity in pericytes rises after birth in step with genes linked to pericyte maturity, blood vessel development, and cell migration. In contrast, its activity is much lower in other nearby cell types, suggesting a specialized role in these vascular support cells.
When the brake on pericytes is removed
To test what FOXF2 actually does, the team created mice in which Foxf2 could be deleted specifically in PDGFRβ-positive pericytes right after birth. Under normal oxygen conditions, overall lung structure and blood oxygen levels looked largely normal, but the fine details told a different story. Pericytes multiplied excessively and packed densely around capillaries, while markers of mature, well-functioning pericytes fell. These pericytes were more likely to be in the DNA-synthesis phase of the cell cycle, and lab experiments confirmed that blocking Foxf2 directly in cultured pericytes boosted their proliferation and reduced their migration. At the same time, genes that normally help vessels grow and remain stable, such as Angptl4 and Angpt2, were turned down, while genes driving cell division and energy production were turned up. Together, this paints a picture of pericytes that are numerous but less competent.
Oxygen stress exposes a critical weakness
The consequences became far more serious when the newborn mice were exposed to high oxygen levels, a standard model of preterm lung injury. In both mouse lungs and human samples from infants with bronchopulmonary dysplasia, FOXF2 in pericytes was markedly reduced after such injury. When Foxf2-deficient mice experienced hyperoxia, their survival fell sharply, their blood oxygen saturation dropped, and their air sacs became enlarged and simplified, indicating poor alveolar development. At the same time, capillary coverage in the alveoli shrank, endothelial cells divided less, and more fluorescent tracer leaked out of blood vessels into the lung tissue—a sign of a failing vascular barrier. Flow cytometry confirmed that, under injury, endothelial cell numbers declined while pericytes further accumulated and remained highly proliferative, highlighting a breakdown in the normal partnership between these two cell types.
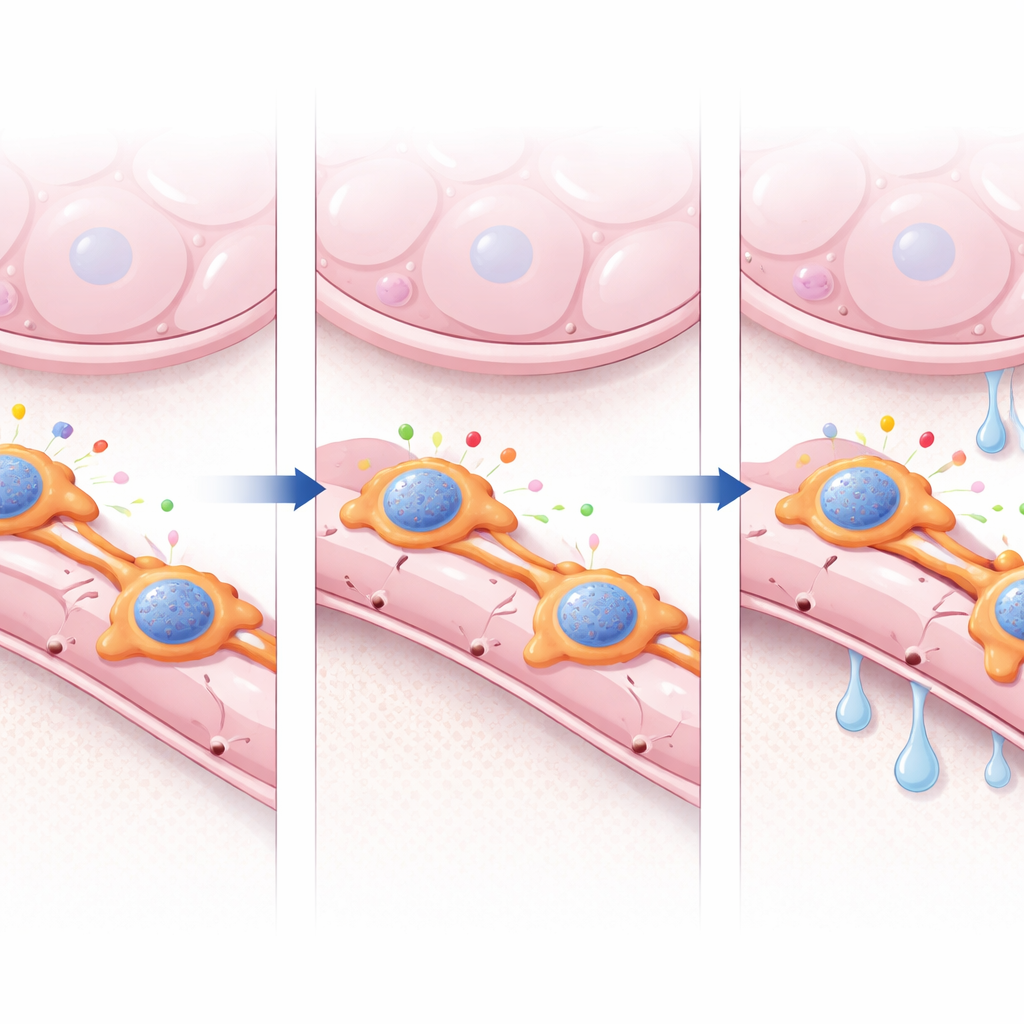
How pericyte signals shape vessel repair
Diving deeper into the molecular crosstalk, the researchers used computational tools to map signaling routes between pericytes and endothelial cells. In healthy lungs, pathways promoting vessel growth and stability were prominent, including those involving ANGPTL4, a secreted protein with pro-angiogenic and barrier-modulating roles. In Foxf2-deficient lungs, these helpful signals weakened, while stress and inflammation pathways strengthened. Conditioned media from Foxf2-lacking pericytes supported less tube formation by human lung endothelial cells in culture, but adding back recombinant ANGPTL4 partly rescued this defect, pointing to a FOXF2–ANGPTL4 axis as a key driver of repair. Chromatin profiling further revealed that FOXF2 binds near many pro-angiogenic genes and helps keep their surrounding DNA regions accessible. When FOXF2 was lost, accessibility dropped at thousands of these sites, including at the Angptl4 locus, suggesting that FOXF2 acts as a chromatin organizer that enables other factors to maintain a healthy pericyte program.
What this means for fragile newborn lungs
Taken together, the findings show that FOXF2 acts as a master regulator that keeps neonatal lung pericytes mature, restrained in their growth, and able to send the right signals to neighboring endothelial cells. When FOXF2 levels fall after oxygen injury, pericytes become overabundant but dysfunctional: they send fewer nurturing cues like ANGPTL4, capillaries fail to regrow properly, vessel walls become leaky, and the architecture of the air sacs deteriorates. For families and clinicians caring for preterm infants, this work highlights pericytes—and the FOXF2-driven gene networks within them—as promising targets for future therapies aimed at preserving tiny blood vessels, improving gas exchange, and reducing long-term lung damage after early-life oxygen exposure.
Citation: Sun, F., Zhao, Y., Do, J. et al. FOXF2 regulates pericyte–endothelial signaling required for vascular homeostasis after neonatal hyperoxic lung injury. Nat Commun 17, 2691 (2026). https://doi.org/10.1038/s41467-026-69525-7
Keywords: bronchopulmonary dysplasia, lung development, pericytes, angiogenesis, neonatal hyperoxia