Clear Sky Science · en
Laser interstitial thermal therapy and adjuvant pembrolizumab in recurrent high-grade astrocytoma: a Phase 1/randomized Phase 2b trial
Why this brain cancer study matters
Recurrent high-grade astrocytoma, including glioblastoma, is one of the most lethal brain cancers, and current treatments rarely keep it in check for long. This study tests a two-step strategy that uses heat from a laser to destroy tumor tissue and open the brain’s defenses, followed by an immune drug that helps the body’s own T cells attack any remaining cancer. For patients and families facing this diagnosis, the work explores whether a minimally invasive procedure can turn an almost unresponsive cancer into one that finally reacts to immunotherapy.
A new way to tackle stubborn brain tumors
Patients in this trial all had high-grade astrocytomas that had come back after standard therapy, which usually includes surgery, radiation, and chemotherapy. The researchers focused on two tools. The first, laser interstitial thermal therapy (LITT), is a keyhole procedure in which a thin probe is guided by MRI into the tumor and used to heat and kill cancerous tissue while sparing much of the surrounding brain. The second tool is pembrolizumab, a widely used immunotherapy drug that blocks PD-1, a molecular “brake” that often keeps T cells from attacking tumors. Earlier attempts to use such drugs alone in these brain cancers had been disappointing. The team therefore asked whether using the laser first could prime the tumor and the immune system to respond better to pembrolizumab. 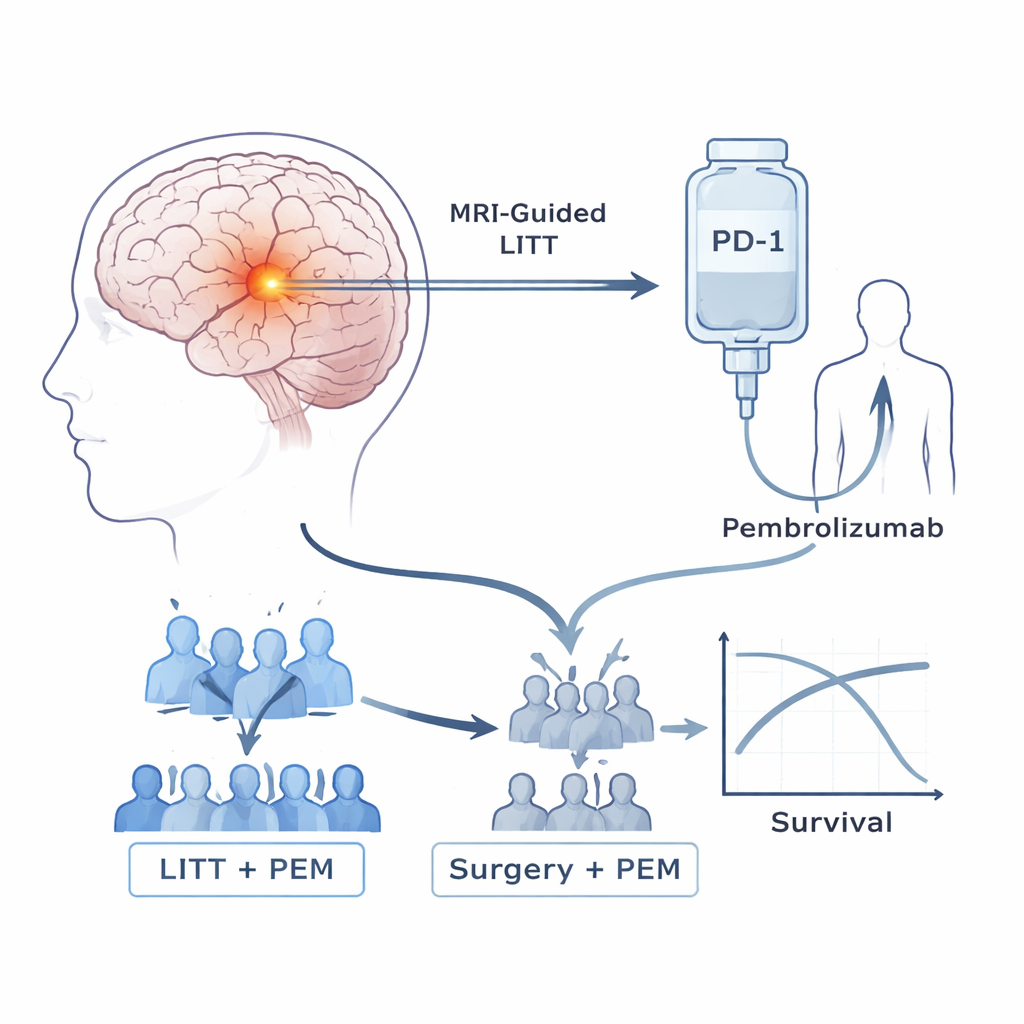
How the clinical trial was run
The study began with a small safety phase, showing that patients could safely receive pembrolizumab every three weeks after LITT, with no dose-limiting side effects. It then moved into a Phase 2b trial. Initially, patients were randomly assigned to one of two groups: LITT followed by pembrolizumab, or conventional repeat brain surgery without laser, followed by pembrolizumab. As outside evidence grew that surgery plus immunotherapy offered little benefit on its own, an independent committee reviewed early results from this trial. Seeing signs that the non-laser group was doing poorly while the LITT group appeared to live longer, they stopped further randomization, and all later patients received the laser-plus-drug combination.
What happened to survival and tumor control
Among 39 patients who received treatment as planned, those given LITT plus pembrolizumab lived notably longer than those who had surgery plus pembrolizumab. Median overall survival was about 11.8 months with the combined LITT approach, versus 5.2 months with surgery alone, and none of the surgery patients were alive at 18 months compared with 42% of those in the LITT group. The time before the tumor started growing again also improved, from 1.6 months in the surgery group to 4.5 months with LITT. About 27% of LITT-treated patients had a measurable shrinkage of their tumors and 70% achieved at least temporary disease control, while all patients who had non-laser surgery plus pembrolizumab experienced progression. Side effects from the combined treatment were mostly mild and manageable, with some cases of diarrhea, muscle weakness, and liver enzyme increases, but no treatment-related deaths.
What the immune system was doing
To understand why the combination might work, the scientists closely examined blood cells over time using single-cell RNA sequencing and T-cell receptor tracking. After LITT, they saw a specific group of innate immune cells, called non-classical monocytes, shift into a more active state linked to migration, inflammation, and interaction with T cells—changes not seen after standard surgery. When pembrolizumab was then started, patients who survived longer showed strong waves of CD8 T-cell activation and clonal expansion, meaning many identical T cells targeting the same antigens were multiplying. These T cells tended to move into memory and effector states that can recognize and kill tumor cells, rather than becoming exhausted and ineffective. Long-term survivors also showed a drop in IDO-1, a molecule associated with immune suppression, hinting that the combination may relieve multiple layers of immune braking. 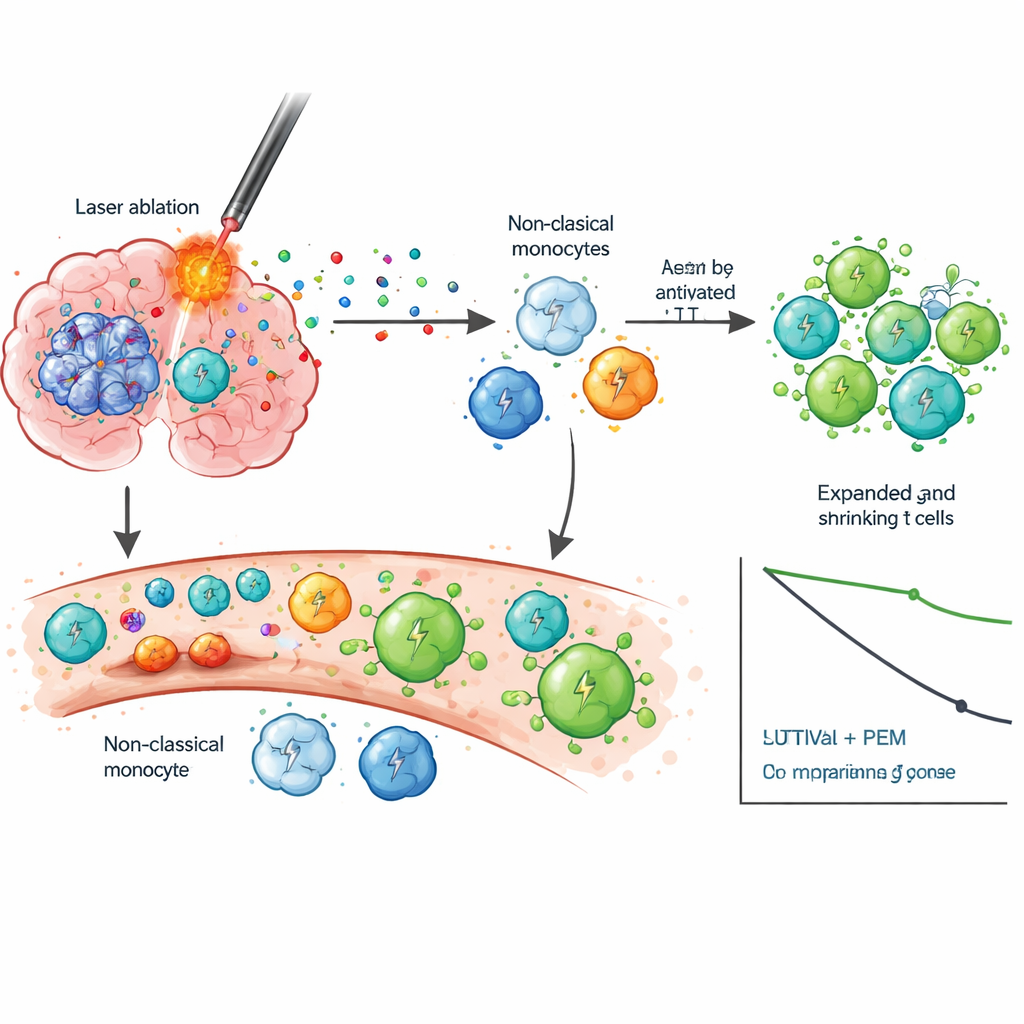
What this could mean for patients
For people living with recurrent high-grade astrocytoma, this work suggests that using MRI-guided laser ablation to debulk the tumor, briefly loosen the blood–brain barrier, and spill tumor fragments into circulation can set the stage for immunotherapy to work where it usually fails. By lighting a “signal flare” for the immune system and then removing the PD-1 brake with pembrolizumab, the combination appears to generate more durable T-cell responses and extend survival in a cancer with very few options. Because the study was relatively small and randomization ended early, the authors stress that larger, fully controlled trials are still needed. Even so, the results point toward a promising new strategy: pairing precisely targeted physical tumor destruction with immune drugs to turn a cold, resistant brain tumor into one that the body can finally recognize and fight.
Citation: Campian, J.L., Le, S.B., Ghiaseddin, A. et al. Laser interstitial thermal therapy and adjuvant pembrolizumab in recurrent high-grade astrocytoma: a Phase 1/randomized Phase 2b trial. Nat Commun 17, 1763 (2026). https://doi.org/10.1038/s41467-026-69522-w
Keywords: glioblastoma, laser ablation, pembrolizumab, immunotherapy, brain tumor