Clear Sky Science · en
Visible-light-triggered exclusive bistable E/Z photoswitching based on sterically frustrated dicyanostilbene derivatives
Light as a Gentle On–Off Switch
Imagine being able to turn the behavior of a material on and off using only gentle green or blue light, instead of heat or harsh ultraviolet rays. That is the central idea of this research. The authors have designed tiny light‑responsive molecules that can flip between two shapes and then stay put for years, acting like molecular‑scale switches. Because these switches are clean, efficient, and work even in tightly packed solids, they could underpin safer data storage, smart coatings, and light‑controlled devices that waste less energy and create fewer chemical by‑products.
Why Current Light Switches Fall Short
Many natural processes already rely on light‑driven shape changes—our own vision, for instance, depends on a molecule in the eye that twists when it absorbs light. Chemists have tried to mimic this by designing artificial “photoswitches,” small molecules that change shape under illumination. But most existing systems misbehave: instead of following a single tidy route, they can undergo several competing reactions, giving a messy mix of products that are hard to purify. They also often need damaging ultraviolet light, flip back too quickly once the light is turned off, or produce two shapes that are so similar in properties that separating and using them is impractical.
Building a Better Molecular Toggle
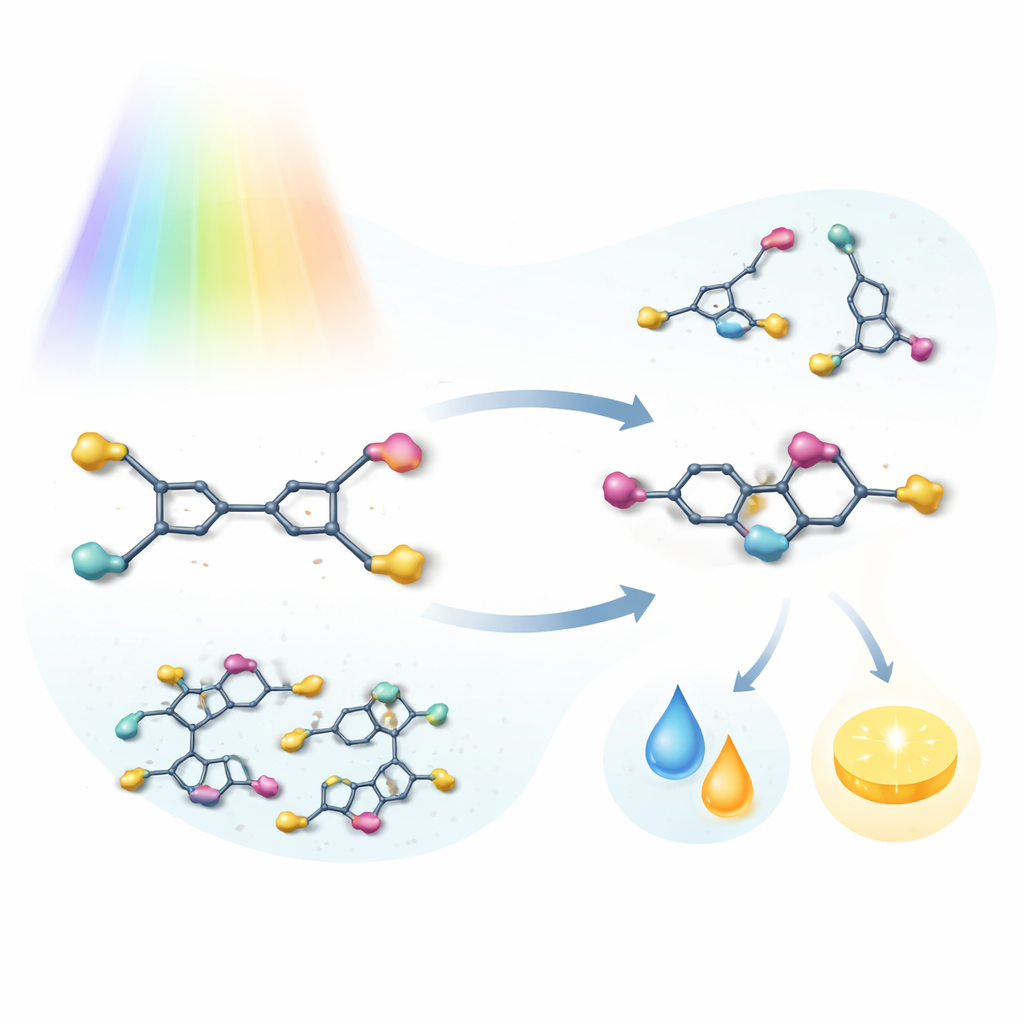
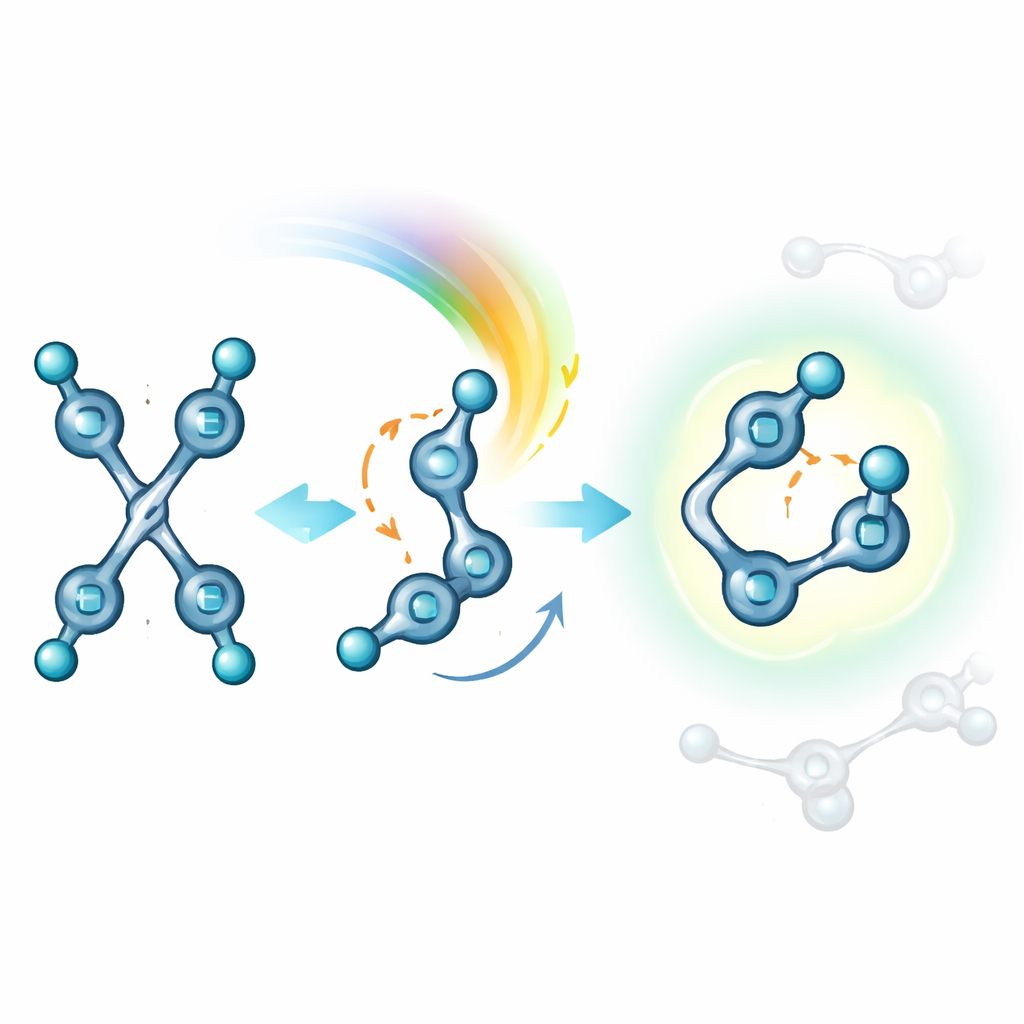
The team focused on a family of molecules related to stilbene, a classic light‑sensitive compound, but modified with strongly pulling cyano groups and bulky end units. These dicyanostilbene switches, named DPA, PTZ, and CBZ, were designed so that their size and shape deliberately crowd the central double bond. Under visible green light, each molecule converts from an extended "E" form to a bent "Z" form; under blue light, it switches back. Careful optical measurements and nuclear magnetic resonance experiments showed that, unlike most relatives, these molecules follow essentially one clean pathway: they only swivel around the double bond, without forming unwanted rings or dimers. In other words, light drives a reversible two‑state toggle instead of a jumble of side reactions.
Extreme Staying Power and Easy Separation
Once in the Z form, these switches barely relax back on their own. By heating samples and tracking their slow return to the E form, the researchers calculated thermal half‑lives at room temperature ranging from about a decade to nearly two millennia—unusually long even by the standards of advanced photoswitches. At the same time, the two shapes differ strikingly in polarity, solubility, and glow. For one compound, the E form is so insoluble that it crystallizes out of solution as it is produced by light, allowing the two shapes to separate themselves. Across the series, the bent and extended forms also emit light with different brightness, so the switching process can be watched directly by eye under ultraviolet illumination as a turn‑on or turn‑off change in fluorescence.
Working in Crowded, Solid‑Like Environments
Many light‑responsive molecules work only when well dispersed; in crowded solids their neighbors sit so close that they tend to lock together or react in pairs. Here, single‑crystal X‑ray studies showed how the bulky end groups keep these dicyanostilbenes apart in the solid or aggregated state. The central double bonds of neighboring molecules are simply too far from each other to fuse, and the overall packing is loose enough to leave “free volume” for the internal twisting motion. At the same time, the bent Z form is internally stabilized by weak attractions between its own rings, making it both hard to over‑react and slow to fall apart. The net result is that the same clean E↔Z switching seen in solution also operates in densely packed aggregates, with similar or even higher efficiencies.
From Invisible Signals to Visible Messages
Because light switching strongly alters fluorescence, the authors demonstrated simple information‑processing functions. By shining green and blue light on mixtures of two different switches in different orders, they could produce distinct patterns of brightening and dimming, which they mapped onto letter codes to spell short words. They also embedded the Z forms into transparent polymer films that change both color and glow permanently when heated enough to force them into the E form. Such films could serve as built‑in over‑temperature indicators or anti‑counterfeiting tags that reveal their history through a visible change.
What This Means in Everyday Terms
In practical terms, the study shows that it is possible to design tiny molecular components that respond cleanly and reliably to safe, visible light, then stay in their chosen state for years unless deliberately reset. By using clever three‑dimensional crowding to block all but one reaction pathway, the researchers turned a finicky chemical scaffold into a robust, bistable light switch that also carries its own color and brightness readout. This combination of precise control, durability, and easy observability makes these dicyanostilbene switches promising building blocks for greener chemical processes, smart materials that signal damage or overheating, and future optical data storage or encryption schemes written and read with nothing more than beams of light.
Citation: Bi, H., Zhao, Y., Deng, S. et al. Visible-light-triggered exclusive bistable E/Z photoswitching based on sterically frustrated dicyanostilbene derivatives. Nat Commun 17, 2666 (2026). https://doi.org/10.1038/s41467-026-69516-8
Keywords: photoswitching, visible light, molecular switches, smart materials, photochromism