Clear Sky Science · en
Strategically significant synthesis of conjugated porous organic polymers via retro diazotization chemistry
Turning Light into Chemical Control
Chemists are constantly searching for cleaner ways to make useful materials and run industrial reactions using only abundant elements, air, and visible light. This study introduces a new family of sponge-like plastics—called conjugated porous organic polymers—that are built under surprisingly mild conditions and then double as powerful light-driven catalysts. The work matters because it points toward metal-free routes to make complex chemicals and to manage reactive oxygen species, which are central to green chemistry, environmental cleanup, and even medicine.
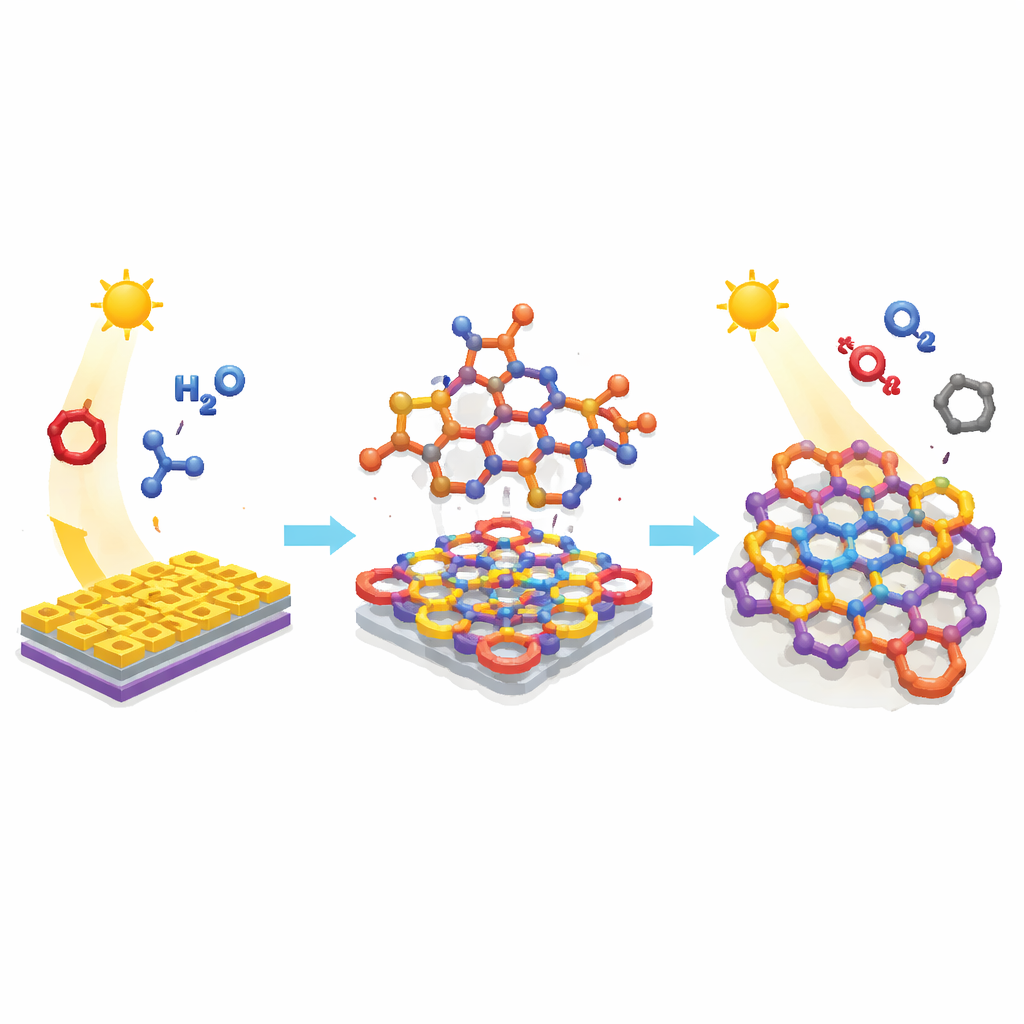
Building a Better Porous Plastic
The authors focus on a special class of rigid, carbon-based networks that conduct charges along their backbones and contain tiny pores throughout their structure. Such materials can absorb light, move electrons, and host gas molecules, making them attractive for solar cells, batteries, and catalysis. However, the usual ways of making these polymers rely on expensive metal catalysts and pre-decorated starting blocks, and they struggle to place heavy halogen atoms like bromine and iodine in precise positions. Those atoms are important because they can tune how the material absorbs light and separates charges, but current methods often require harsh conditions and leave behind metal residues.
Letting a Two-Dimensional Metal Sheet Do the Work
To sidestep these limitations, the team turned to “photoredox” chemistry—reactions powered by visible light that shuttle single electrons between molecules. Their key player is bismuthene, an ultrathin sheet of the metal bismuth that behaves like a tiny semiconductor. Under blue light, bismuthene can activate simple amine-containing building blocks in the reaction mixture, temporarily converting them into highly reactive species that link together with aromatic rings to form long, connected chains. Crucially, this happens through a stepwise, single-electron pathway rather than the traditional metal-catalyzed coupling that demands pre-installed halogens or boron groups on every starter molecule.
Designing Smart Building Blocks
Using this strategy, the researchers assembled several families of polymers with record-high chain lengths—up to about 322,000 grams per mole—while maintaining relatively narrow size distributions, a sign of controlled growth. They mixed electron-rich aromatic “cores” with electron-poor “linkers” to create so-called donor–acceptor architectures that naturally favor charge separation when illuminated. By choosing linkers that carry sulfone groups and by incorporating bromine or iodine atoms into the cores, they were able to adjust how strongly the polymers absorb from the visible into the near-infrared range, as well as how efficiently they move charges and withstand heat. Microscopy and spectroscopy confirmed that the resulting materials form layered or network-like particles with defined pores, robust carbon frameworks, and halogens embedded directly into the backbone rather than tacked on after the fact.
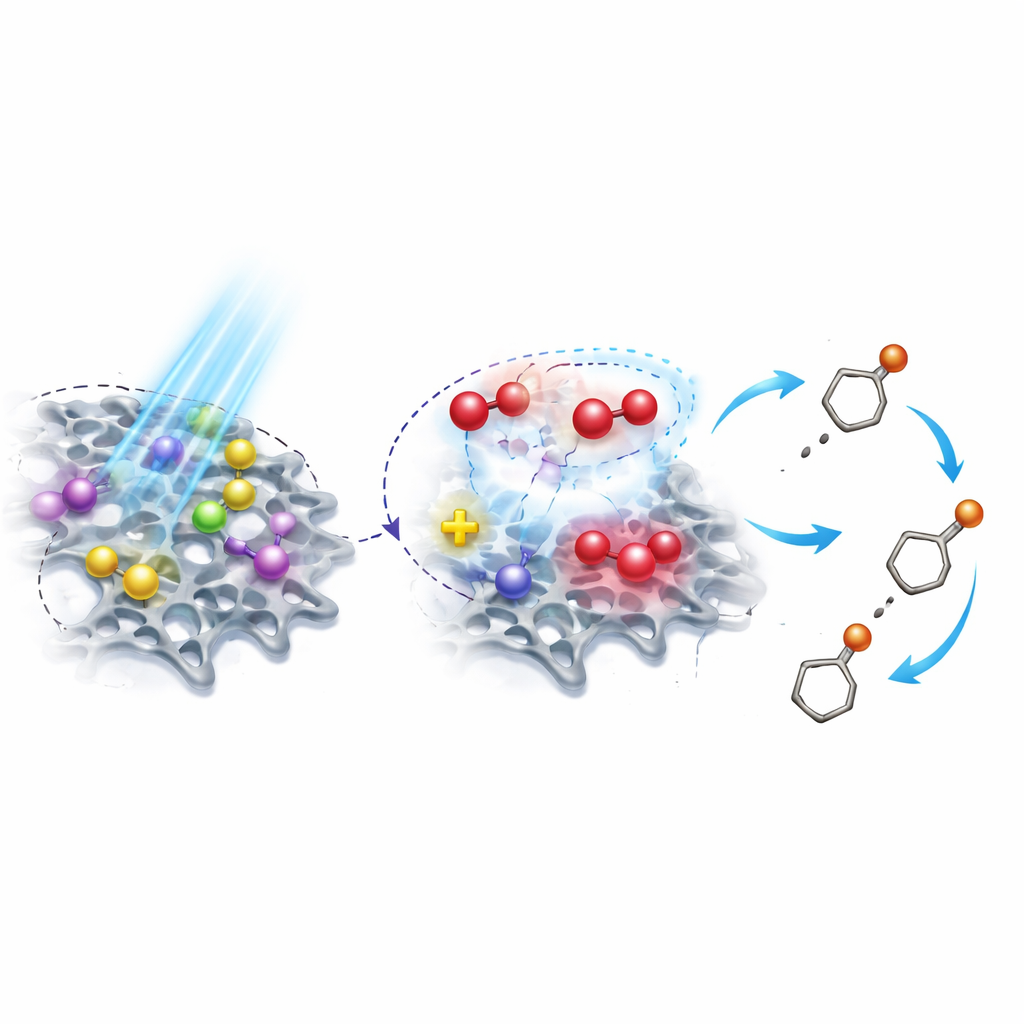
Using Light and Oxygen to Make Valuable Chemicals
To test what these materials can do, the team used them as photocatalysts for a benchmark reaction: turning styrene—a simple petrochemical—into benzaldehyde, an important ingredient in fragrances, flavorings, and fine chemicals. In water mixed with a small amount of an alcohol co-solvent and under blue LED light, the best halogenated polymer turned styrene into benzaldehyde with over 99% yield and selectivity, using nothing more than oxygen from the air. Control experiments showed that similar polymers without sulfone linkers or heavy halogens were far less active. Additional tests with chemical “traps,” spectroscopic probes, and spin-detecting techniques revealed that the key reactive species is singlet oxygen, a highly energetic form of O₂, aided by holes (positive charges) on the polymer. The heavy atoms bromine and iodine enhance the formation of long-lived excited states, making it easier for the polymer to hand energy to oxygen and keep electrons and holes apart long enough to do useful work.
What This Means for Future Clean Chemistry
In plain terms, this work shows how to use light and a thin sheet of bismuth to stitch together small organic molecules into robust, porous, and finely tuned plastics that then act as efficient, metal-free photocatalysts. By controlling where halogens and sulfone groups sit in the framework, the authors can dial in how these materials absorb light and generate reactive oxygen, allowing them to oxidize styrene cleanly to benzaldehyde through singlet oxygen rather than more wasteful pathways. The approach addresses long-standing challenges in making halogen-rich conjugated polymers while avoiding harsh conditions and precious metals, opening the door to a new generation of designer porous materials for green synthesis, solar-driven chemical production, and other technologies that rely on taming oxygen and light.
Citation: Ozer, M.S., Eroglu, Z., Koyuncu, S. et al. Strategically significant synthesis of conjugated porous organic polymers via retro diazotization chemistry. Nat Commun 17, 3008 (2026). https://doi.org/10.1038/s41467-026-69515-9
Keywords: conjugated porous polymers, photocatalysis, singlet oxygen, bismuthene, halogenated polymers