Clear Sky Science · en
Synergistic electrode design for efficient CO2 electrolysis to multicarbon products at elevated temperatures
Turning Waste Heat into Useful Chemistry
Factories that convert carbon dioxide into useful fuels and chemicals may sound like science fiction, but they are already being built. As these devices grow larger and more powerful, they heat up—much like a laptop working hard. This study shows that instead of fighting that heat with costly cooling systems, smart electrode design can harness higher temperatures to turn CO2 more efficiently into energy-rich, multi‑carbon products such as ethylene and alcohols.
Why Hotter Reactors Are a Double-Edged Sword
Industrial CO2 electrolysis runs electricity through water and CO2 to make new molecules. Scaling these systems up increases electrical resistance and poor heat removal, which in turn drives the cell temperature well above room temperature. Higher temperature speeds up chemical reactions and lowers energy barriers—good news in principle—but it also creates serious problems. Copper, the workhorse metal that helps link carbon atoms together, changes its surface structure when hot. Gas diffusion electrodes, which carefully balance gas, liquid, and solid regions, begin to flood with water vapor. At the same time, key CO‑based reaction intermediates detach from the surface too early, and the system instead produces hydrogen and simple one‑carbon products, wasting both electricity and CO2.
Finding the Weak Links in the Hot Cell
The researchers systematically heated a flow‑cell reactor from room temperature to 75 °C and watched how copper-based electrodes behaved. Using a suite of structural probes, they found that bare copper quickly oxidizes and subtly reshapes at higher temperature, shifting products from valuable two‑carbon molecules toward methane and hydrogen. A more stable form, cuprous oxide (Cu2O) nanocubes, held its structure better but still performed poorly when hot. The culprit turned out not only to be the catalyst itself, but also the surrounding environment: increased water vapor pressure flooded the gas diffusion electrode, choking off CO2 access and enlarging the area where only hydrogen could form. Even when flooding was controlled, higher temperature made CO intermediates more likely to desorb before they could couple into multi‑carbon products.
Building a Smarter, Water‑Repelling Electrode
To turn this hostile hot environment into an advantage, the team redesigned the cathode as a layered “tandem” structure. First, they mixed the Cu2O catalyst with tiny particles of polytetrafluoroethylene (PTFE)—a highly water‑repellent material—to stabilize the delicate gas–liquid–solid interface and prevent flooding, even at high temperatures and high current. Next, they added a silver layer that excels at converting CO2 into CO, feeding a steady stream of CO intermediates toward the Cu2O. Finally, they decorated the Cu2O surface with isolated palladium atoms, which bind CO more strongly and keep it on the surface long enough for carbon–carbon bonds to form. Together, these layers manage water, local gas concentration, and intermediate binding strength so that the extra thermal energy lowers the barrier for carbon‑carbon coupling instead of simply speeding up side reactions.
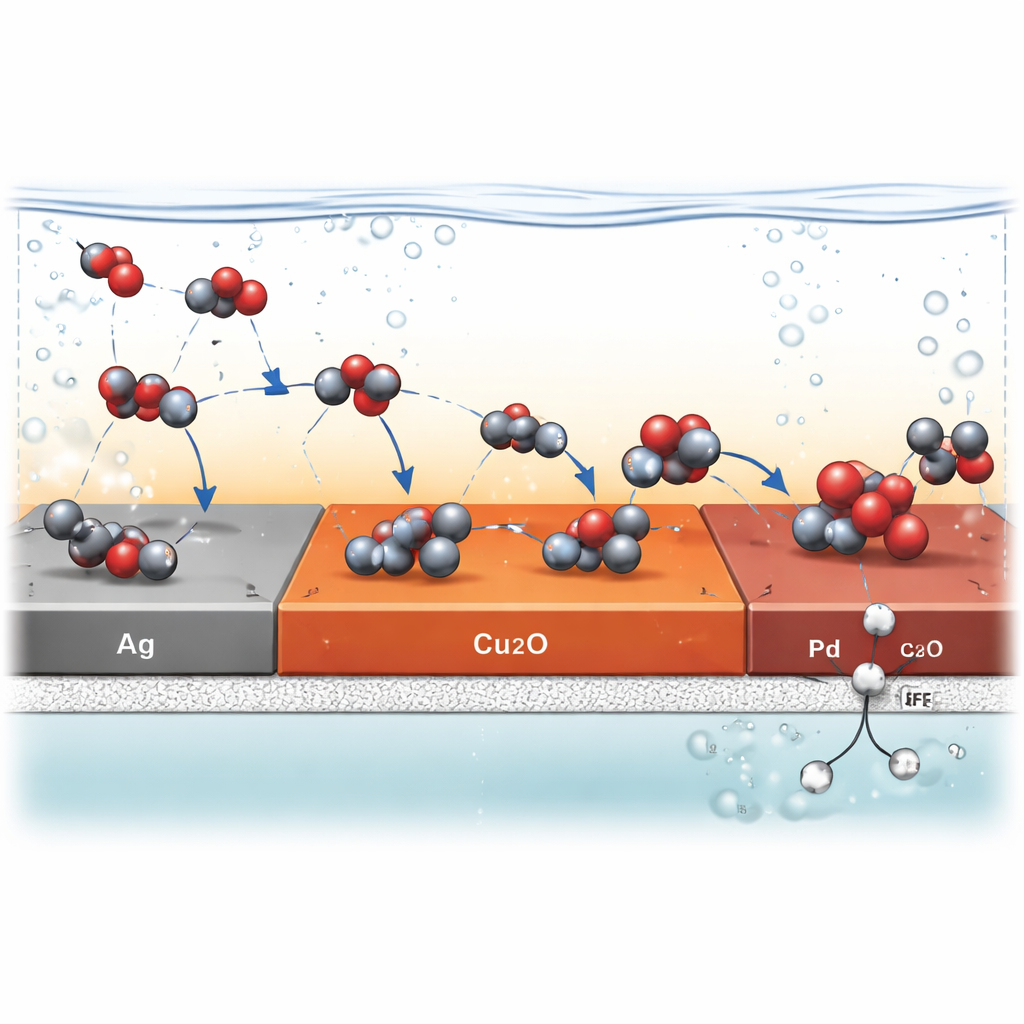
Turning Heat from Foe to Friend
With this synergistic electrode design, the reactor achieved more than 70% Faradaic efficiency for multi‑carbon products across industrially relevant current densities at 75 °C, all while operating stably for many hours. The hotter cell not only produced more desirable products, it also used electricity more efficiently: the energy efficiency toward multi‑carbon products improved by about 30% compared with room‑temperature operation. A preliminary cost analysis indicated that running hot and eliminating active cooling could cut nearly 15% of operating costs related to temperature control. In simple terms, the study shows that waste heat in large CO2‑to‑chemicals plants can be transformed from a reliability headache into a powerful ally—if the electrode is carefully engineered to control water, gas access, and how strongly reaction intermediates stick to the surface.
Citation: Hu, L., Yang, Y., Wang, J. et al. Synergistic electrode design for efficient CO2 electrolysis to multicarbon products at elevated temperatures. Nat Commun 17, 2684 (2026). https://doi.org/10.1038/s41467-026-69506-w
Keywords: CO2 electrolysis, multicarbon fuels, electrocatalysis, industrial decarbonization, flow cell reactors