Clear Sky Science · en
Interfacial stress decoupling enables stable palladium-based hydrogen sensing
Why safer hydrogen matters
Hydrogen is gaining attention as a clean fuel for factories, vehicles, and energy storage. But the same gas that promises a low‑carbon future is also highly flammable, demanding fast and reliable leak detection long before concentrations become dangerous. Many existing hydrogen sensors are sensitive yet wear out quickly, especially when their active materials repeatedly swell and shrink during use. This article reports a new way to build tiny hydrogen sensors that stay both ultrasensitive and mechanically robust, paving the way for long‑lived, low‑power detectors that can be made across entire wafers and built into portable safety devices. 
The weakness at the joining line
Most electrical gas sensors rely on a thin “sensing” film attached to a solid support with metal electrodes. For hydrogen, palladium is a favorite: it soaks up hydrogen atoms, forming a hydride and changing its electrical resistance in a way that can be read out as a signal. However, every absorption and release cycle makes the palladium lattice expand and contract, building up stress where it meets the underlying substrate. Over time this leads to cracks, dislocations, and ultimately peeling or fracture at the interface, which degrades the signal or kills the device. Traditional tricks to strengthen adhesion—roughening the surface, adding sticky polymers, or inserting rigid buffer layers—often clamp the palladium so tightly that hydrogen can no longer move freely, slowing the response and reducing sensitivity.
A floating bridge between metal layers
To escape this trade‑off, the authors designed a “floating‑structure” hydrogen sensor in which the active palladium layer is linked to the gold bottom electrode by a very thin molecular bridge: a self‑assembled monolayer (SAM) of dithiol molecules. Each molecule has sulfur atoms at both ends that bind strongly to gold and palladium, while its carbon chain forms a flexible backbone. This creates a dual interface—palladium–SAM and SAM–gold—instead of a single rigid junction. The SAM behaves like a molecular shock absorber: as hydrogen enters the palladium and causes it to swell, the carbon chains bend and stretch, relieving both sideways and vertical stress while keeping the metals firmly joined. Calculations confirm that the sulfur–metal bonds are stronger than a direct palladium–gold contact, and that the SAM‑based interface is mechanically tougher, failing at higher strain and in a more ductile, damage‑tolerant manner. 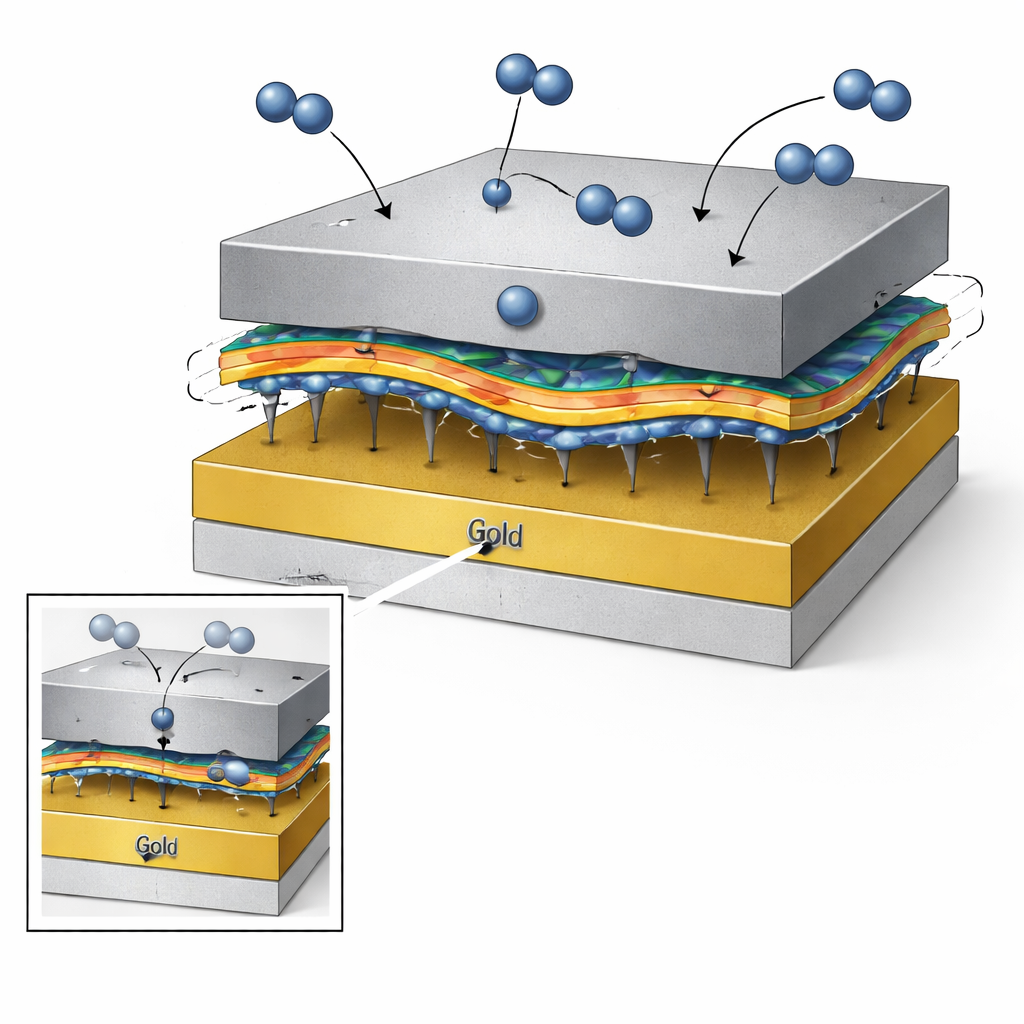
How the new structure boosts sensing
The team built sensors where the palladium film and gold electrode are stacked vertically with the SAM sandwiched in between, leaving the palladium exposed all around for gas access. High‑resolution electron microscopy and elemental mapping show a uniform, roughly two‑nanometer‑thick molecular layer bridging the metals. Electrical tests reveal that adding the SAM slightly lowers conductivity but still allows efficient charge transport. More importantly, hydrogen sensing at room temperature shows a dramatic improvement: compared with a conventional planar device and a floating design without the SAM, the full floating‑SAM architecture delivers a much larger change in resistance, faster response and recovery, and reliable operation at hydrogen levels up to 4 percent by volume. Modeling of hydrogen uptake kinetics indicates that the SAM greatly weakens the “clamping” effect of the substrate, allowing hydrogen to diffuse into the palladium more rapidly—about an order of magnitude faster than without the molecular layer.
Stability under real‑world stress
Durability tests highlight the advantage of engineering the interface rather than just the sensing material. When repeatedly cycled between nitrogen and hydrogen, sensors with the SAM show nearly unchanged performance over at least 50 cycles, even at high hydrogen concentrations that cause large volume changes in palladium. Devices without the SAM, in contrast, lose more than half their response or fail outright under the same conditions. The floating SAM design also handles varying humidity with only modest impact on performance, distinguishes hydrogen from other gases such as nitrogen dioxide and hydrogen sulfide, and operates at extremely low power—on the order of a few microwatts at small applied voltages. Over more than three months of testing, the sensors maintain stable signals, suggesting lifetimes compatible with long‑term monitoring.
From wafer to handheld detector
Because the structure is compatible with standard microfabrication methods, the authors produced dense arrays of these sensors across 4‑inch wafers and showed that individual chips have very similar baseline resistance and hydrogen response. Packaged devices behave like their unpackaged counterparts, confirming that they can be integrated into commercial‑style housings. The team then built a complete detection platform by combining a sensor with a Wheatstone bridge, low‑noise amplification, and wireless electronics on a circuit board, and by embedding the system in a handheld unit with its own micro‑pump. This portable detector can pick up hydrogen leaks down to one part per million, transmit readings in real time, and trigger alarms inside settings such as hydrogen cylinder cabinets. Its performance rivals or exceeds that of a commercial detector, particularly in speed.
What this means for future sensors
For non‑specialists, the key message is that the “weakest link” in many sensors is not the sensing material itself but the seam where it meets the rest of the device. By inserting a tailored molecular bridge that is both strongly bonded and mechanically forgiving, this work shows it is possible to keep palladium‑based hydrogen sensors extremely sensitive while preventing them from tearing themselves apart over time. The result is a tiny, low‑power chip that can be mass‑produced, built into portable monitors, and trusted to watch over hydrogen systems for months or years—an important step toward making hydrogen a safer, more practical part of everyday energy infrastructure.
Citation: Gao, R., Zhang, G., Wang, X. et al. Interfacial stress decoupling enables stable palladium-based hydrogen sensing. Nat Commun 17, 2665 (2026). https://doi.org/10.1038/s41467-026-69499-6
Keywords: hydrogen sensing, palladium sensor, self-assembled monolayer, gas leak detection, sensor reliability