Clear Sky Science · en
Hydrated metal charge density as a universal descriptor explaining mechanistic variations in periodate activation toward pollutant degradation
Cleaning Up Water with Smart Chemistry
Many medicines and industrial chemicals slip through conventional water treatment plants and end up in rivers and drinking water. This study explores a promising way to break down such stubborn pollutants using a common oxidizing agent, periodate, and shows that a simple property of metal ions in water can predict how well—and how safely—this chemistry will work. Understanding this rule could help engineers design cleaner, more efficient treatment systems for diverse real‑world waters.
Why Metal Choice Matters for Pollutant Breakdown
Advanced oxidation processes use powerful reactive species to tear apart organic molecules. Periodate is one such oxidant, and it can be “turned on” by dissolved metal ions. The authors compared three closely related metals—iron, ruthenium, and osmium—that belong to the same column of the periodic table and might be expected to behave similarly. All three can activate periodate and help destroy a test drug pollutant, carbamazepine. Yet experiments showed striking differences: ruthenium worked extremely fast, iron was slower, and osmium sat in the middle. Even more puzzling, osmium removed a wide variety of pollutants with little preference, while iron and ruthenium were choosy, attacking some compounds much more readily than others.
Two Very Different Chemical Pathways
To uncover what lay behind these differences, the team tracked which short‑lived oxidizing species appeared in each metal–periodate system. With iron and ruthenium, the dominant players were “metal–oxygen” units that act like precision tools, transferring an oxygen atom directly to specific sites on a pollutant. These pathways tended to form epoxy products—ring structures that add a single oxygen atom—showing that the reaction is quite selective. In the osmium system, however, the main actors were hydroxyl radicals, extremely reactive fragments that attack almost any nearby organic molecule, leading to hydroxylated products and small ring‑opened fragments. Tests with chemical probes and spin‑trapping spectroscopy confirmed that hydroxyl radicals and related reactive oxygen species, not high‑valent osmium–oxygen units, drive most of the pollutant destruction when osmium is present.
How Water Conditions Shape the Reactions
The team also examined how pH and natural water ingredients influence these processes. For osmium, pollutant removal became stronger as the water grew more alkaline, where coordination of extra hydroxide ions around the metal makes it easier for electrons to move and radicals to form. At high metal or oxidant doses, however, the osmium system “self‑quenched” as the newly formed radicals were mopped up by excess chemicals rather than by pollutants. Natural organic matter, which often interferes with oxidation reactions, had surprisingly little impact on osmium‑driven degradation, suggesting that the radical pathway remains robust in realistic waters. In contrast, iron and ruthenium worked best under acidic conditions, and natural organic matter strongly suppressed their activity by soaking up the key metal–oxygen oxidants.
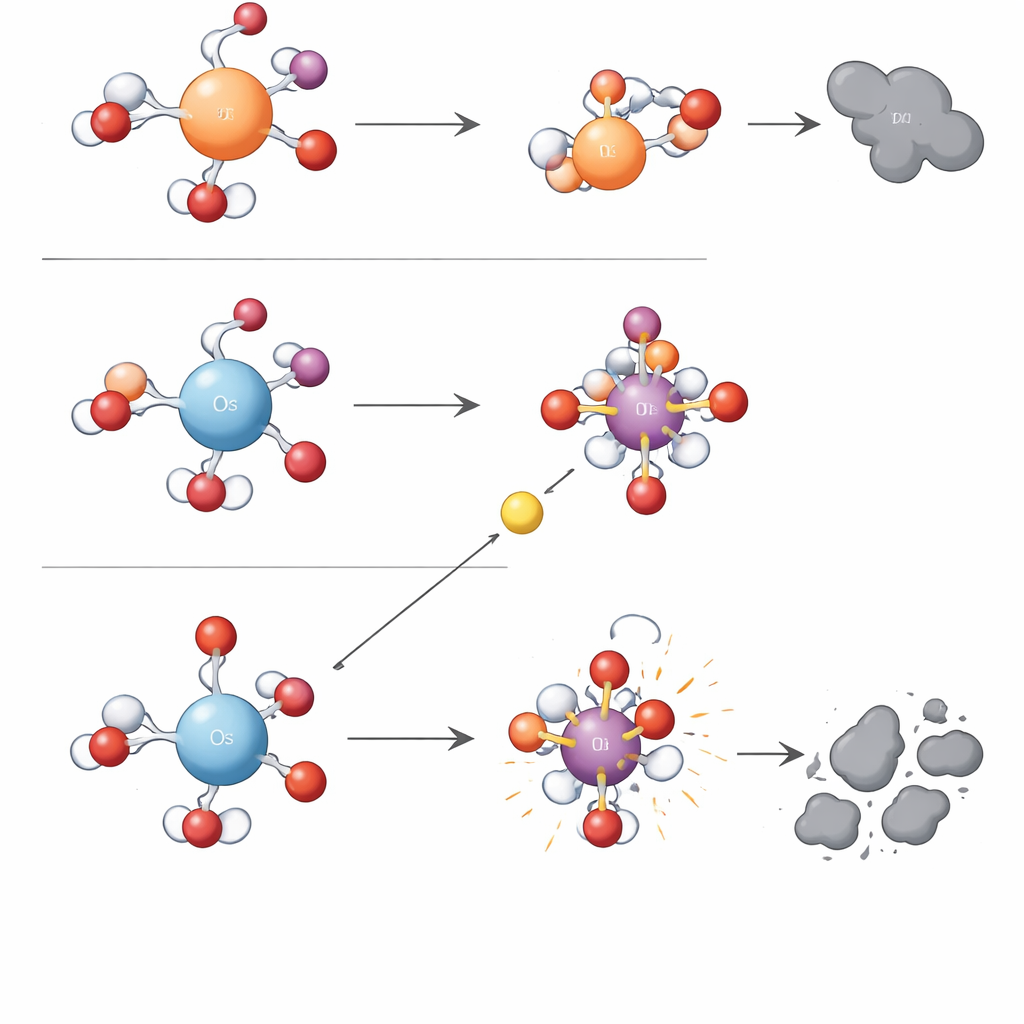
A Simple Charge Rule Behind Complex Chemistry
To move beyond trial‑and‑error, the researchers turned to quantum‑chemical calculations. They found that a single electronic feature—the positive charge density of the hydrated metal center—could explain the split between the two reaction types. Osmium in water carries a higher concentrated positive charge than iron or ruthenium. This strong positive center clings tightly to its bound oxygen atom, making its metal–oxygen unit stable but sluggish toward direct oxygen transfer. At the same time, the strong pull on nearby water and ions lowers the energy barrier for single‑electron steps that ultimately snap an oxygen bond in periodate and release hydroxyl radicals. Metals with lower charge density, like iron and ruthenium, hold electrons more tightly, favoring the formation and use of the selective metal–oxygen oxidants instead of free radicals. The authors call this organizing idea the “Charge Density Control Mechanism,” and show that it also rationalizes trends reported for other metals such as manganese.
Designing Better Water Treatment from a Single Knob
By linking a difficult set of observations to one simple descriptor—the charge density of a metal surrounded by water—this work offers a roadmap for tailoring advanced oxidation processes. High‑charge‑density metals should favor broad, radical‑based breakdown of complex pollutant mixtures, especially in alkaline or organic‑rich waters. Lower‑charge‑density metals are better suited when selective, gentler oxidation is desired, for instance to transform specific contaminants without over‑reacting with everything else present. In practical terms, this means chemists and engineers can screen or design catalysts for water purification by tuning how tightly metal centers hold charge in their watery environment, rather than optimizing each system by trial alone.
Citation: Qian, Y., Sun, Y., Xu, J. et al. Hydrated metal charge density as a universal descriptor explaining mechanistic variations in periodate activation toward pollutant degradation. Nat Commun 17, 2683 (2026). https://doi.org/10.1038/s41467-026-69496-9
Keywords: advanced oxidation processes, periodate activation, water treatment, transition metal catalysis, hydroxyl radicals