Clear Sky Science · en
Mechanistic insights into the non-equilibrium thermodynamics of nitrogen fixation via acoustic cavitation
Turning Air into Useful Fertilizer with Sound
Nitrogen from the air is essential for fertilizers and food production, but converting this stubborn gas into useful forms usually demands huge factories, extreme heat, and high pressure. This study explores a very different approach: using intense sound waves to create tiny exploding bubbles in water that can "fix" nitrogen under far-from-equilibrium conditions. By watching and modelling what happens inside these fleeting hot spots, the researchers show how sound-driven bubbles could offer a new route to make nitrogen-based chemicals without traditional catalysts or giant reactors.
Why Fixing Nitrogen Is So Hard
Our atmosphere is mostly nitrogen gas, but its atoms are locked together by one of the strongest chemical bonds in nature. Breaking that bond efficiently is why the Haber–Bosch process relies on powerful equipment and consumes large amounts of energy worldwide. Conventional methods must strike an awkward balance: temperatures high enough to activate nitrogen, but not so high that the desired products fall apart or equilibrium pushes the reaction backward. This paper argues that instead of maintaining a steady temperature, it can be more effective to briefly overshoot—using ultrafast bursts of heat—and then cool so quickly that useful products are trapped before they decompose.
Using Ultrasound to Create Tiny Reaction Chambers
When powerful ultrasound passes through water, it makes microscopic gas bubbles that grow and then violently collapse, a phenomenon known as acoustic cavitation. Each collapsing bubble behaves like a miniature, short-lived reactor. In billionths of a second, the gas inside is squeezed to temperatures above 5000 kelvin and then cooled again at rates near 1012 kelvin per second. Under these conditions, nitrogen molecules in the bubble can split into reactive fragments, which then combine with oxygen-, hydrogen-, or water-derived fragments to form nitrite, nitrate, or ammonium. The new products are then shot into the surrounding liquid, where they accumulate over time while the next generation of bubbles forms and collapses. 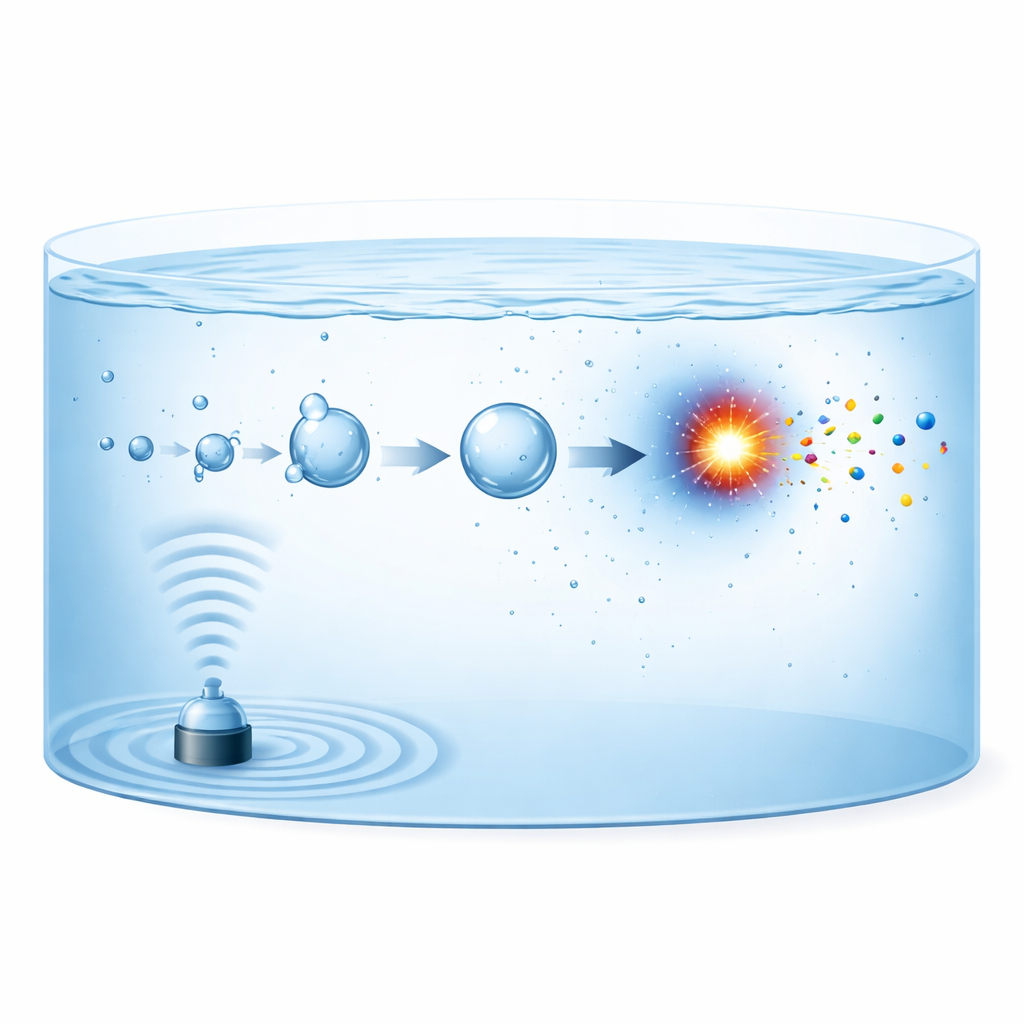
Tuning Bubbles to Choose Between Different Products
The team systematically varied the gases feeding the bubbles (nitrogen plus oxygen or hydrogen), the strength and frequency of the ultrasound, and the presence of solid particles that help bubbles form. With nitrogen–oxygen mixtures, the system mainly produced oxidized products such as nitrite and nitrate; with nitrogen–hydrogen mixtures, it favored ammonium. Small amounts of talc particles acted as bubble "seeds," lowering the threshold for cavitation and making the reactions more reproducible. By adjusting sound pressure and reaction time, the researchers could shift the balance between nitrite and nitrate, showing that part of the chemistry happens inside the collapsing bubble and part continues in the surrounding water as reactive fragments slowly convert nitrite into more oxidized nitrate.
Peering Inside the Nano-Scale Heat Pulse
To understand why such extreme, fleeting conditions still yield stable products, the authors combined measurements with detailed simulations and quantum-chemical calculations. These show that, at very high temperatures, nitrogen can break apart directly in the gas phase, opening pathways that are normally inaccessible. But the same calculations also reveal that keeping the gas hot would make the final products unstable. The key is rapid quenching: the bubble’s temperature spike activates nitrogen, then the near-instant cooling stabilizes intermediate fragments and finished molecules like ammonia and nitrous acid before they can fall apart or revert to nitrogen. Modelling individual bubbles, especially when doped with argon that boosts collapse temperatures, confirmed that higher peak temperatures shift the product mix and raise overall fixation rates. 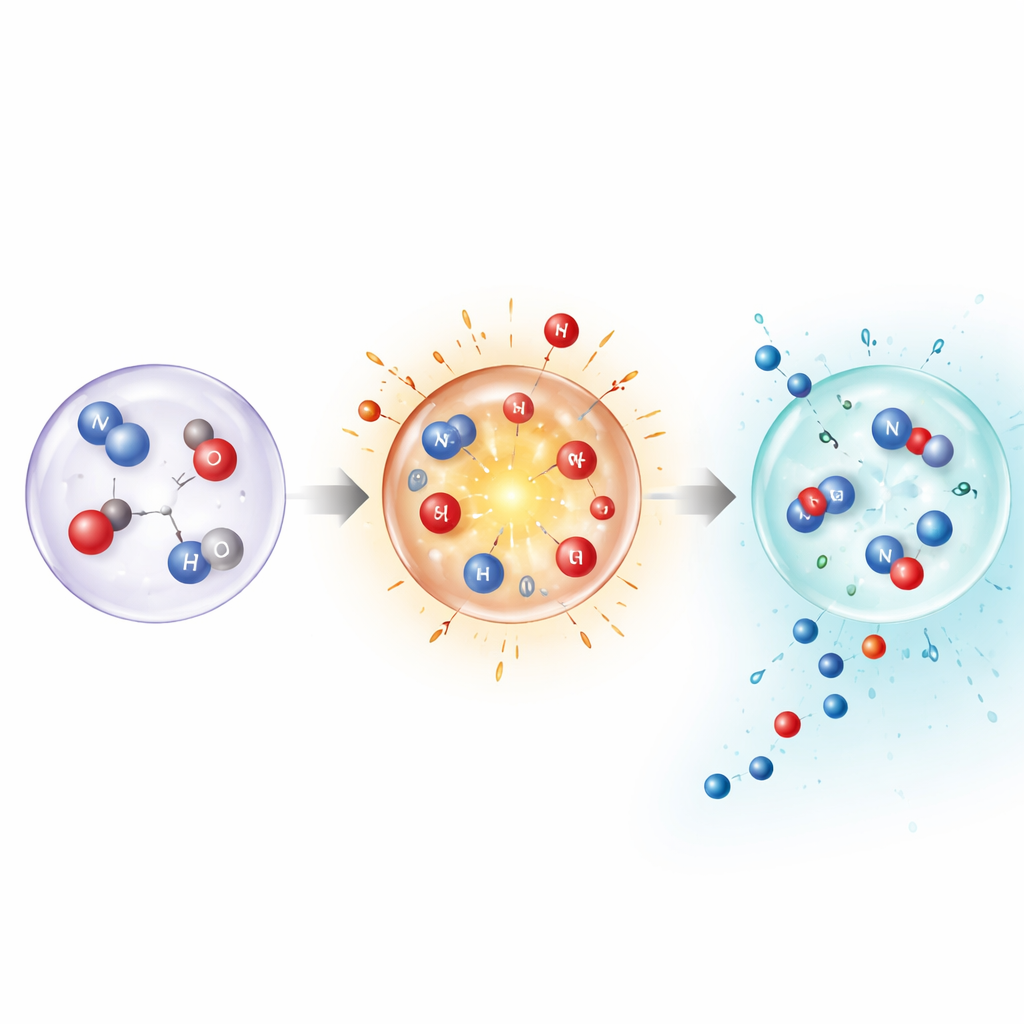
Energy Use and Future Possibilities
Although this sound-driven method is not yet as energy-efficient as the best industrial processes, its performance already rivals historic electric-arc approaches and some modern plasma systems, all while operating at ambient overall conditions and without solid catalysts. Importantly, the same cavitation events also split water, releasing hydrogen, oxygen, and hydrogen peroxide—energy-rich by-products that might be harvested alongside fixed nitrogen. The authors emphasise that their setup is designed to reveal mechanisms rather than to maximize output, but the work establishes acoustic cavitation as a distinct way to fix nitrogen by exploiting extremely rapid thermal cycling in microscopic bubbles. For non-specialists, the takeaway is that carefully controlled sound can turn ordinary water and air into fertilizer ingredients through a sequence of tiny, invisible explosions, hinting at greener and more flexible routes to produce vital nitrogen chemicals in the future.
Citation: Pan, X., Preso, D.B., Liu, Q. et al. Mechanistic insights into the non-equilibrium thermodynamics of nitrogen fixation via acoustic cavitation. Nat Commun 17, 2682 (2026). https://doi.org/10.1038/s41467-026-69466-1
Keywords: nitrogen fixation, acoustic cavitation, sonochemistry, fertilizer production, ultrasound chemistry