Clear Sky Science · en
Photochemical thiocarbonyl difluoride generation enables azetidine synthesis
A New Light-Driven Shortcut to Useful Drug Building Blocks
Chemists are constantly searching for gentler, cleaner ways to build the complex molecules found in modern medicines. This paper describes a light-powered method to turn simple nitrogen-containing compounds into highly versatile building blocks under mild conditions, avoiding some of the toxic and wasteful ingredients used today. The work could make it easier to explore new drug candidates and to fine-tune existing ones by adding fluorine-rich features prized by pharmaceutical chemists.
Turning Simple Amines into Powerful Tools
Many drugs contain nitrogen atoms, and one common trick in medicinal chemistry is to temporarily “activate” nitrogen so it can be reshaped or adorned with new groups. A family of compounds called thiocarbamoyl fluorides is especially attractive for this purpose, because they can be transformed into a variety of useful products and can be converted directly into nitrogen–trifluoromethyl (N–CF3) groups, which often improve a molecule’s stability and behavior in the body. Unfortunately, making the key intermediate, thiocarbonyl difluoride, has typically required harsh, high‑temperature processes and moisture‑sensitive fluorine reagents, limiting its broader use.
Reimagining a Familiar Reagent with Light
The authors revisited an old workhorse of fluorine chemistry, N‑trifluoromethylthiophthalimide (often shortened to Phth–SCF3), usually employed to bolt a trifluoromethyl–sulfur unit onto molecules. They discovered that under visible light, in the presence of a simple organic “sacrificial” reducing agent, this reagent behaves in a completely different way. Instead of attaching itself directly to the target, it breaks apart through a single‑electron process, giving rise to short‑lived sulfur‑containing radicals. These radicals pair up and then shuffle atoms in a sequence of steps that ultimately produces thiocarbonyl difluoride in the reaction mixture, right where it is needed, without ever isolating this reactive gas. 
Unlocking Strained Rings to Build Azetidines
With this in‑situ source of thiocarbonyl difluoride in hand, the team turned to a family of highly strained, tiny ring systems called azabicyclo[1.1.0]butanes. These “spring‑loaded” structures store a great deal of energy in their compact shape. When the newly generated thiocarbonyl difluoride interacts with them, it opens the ring in a polar fashion and triggers a semipinacol rearrangement—a controlled shift of atoms that relieves strain and forges a new, four‑membered nitrogen ring known as an azetidine. In a single, light‑driven step, simple starting materials are converted into complex “spiro” azetidines and fluorinated thiocarbamoyl fluorides, motifs that are otherwise difficult to access directly. 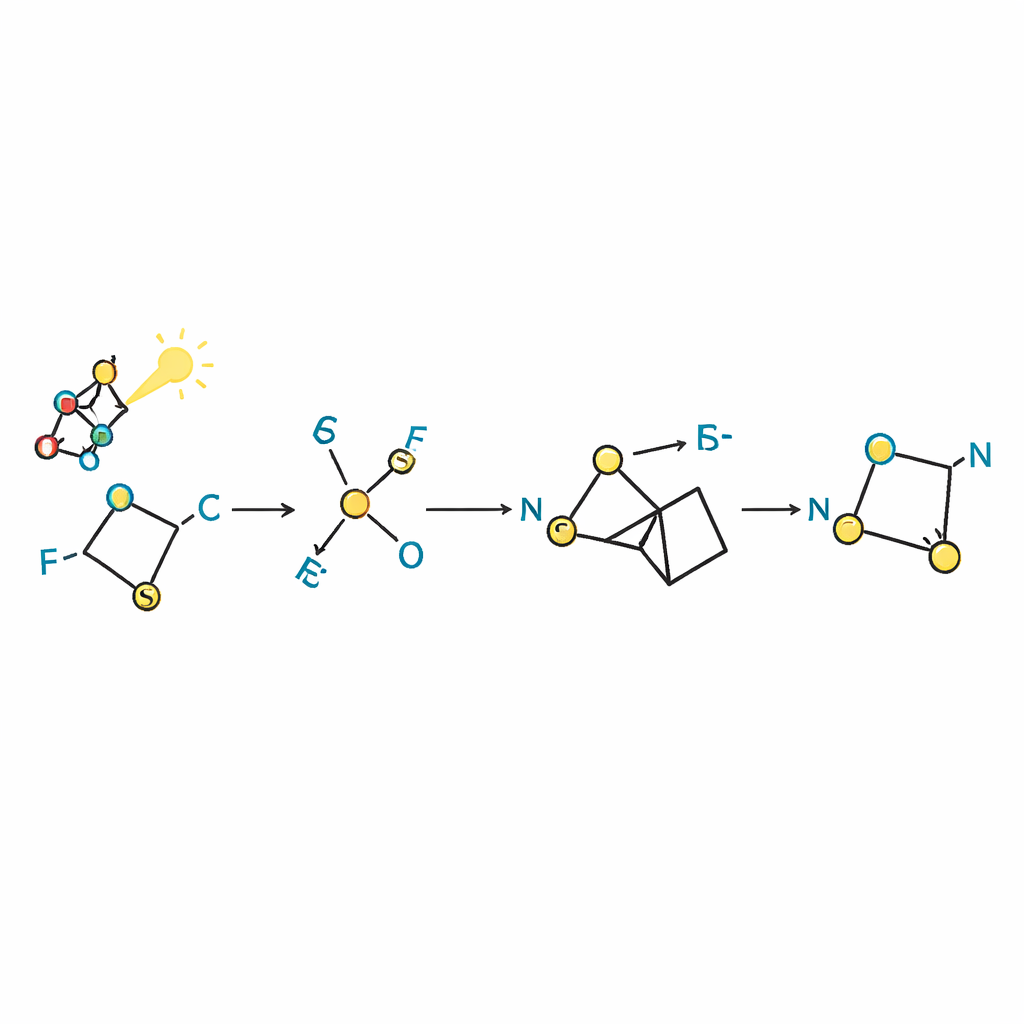
Peering Under the Hood of the Reaction
To understand how this transformation unfolds, the researchers combined time‑resolved spectroscopy, electron‑paramagnetic resonance, and electrochemistry. Their data show that the photocatalyst first transfers an electron to the Hantzsch ester, an organic dye‑like compound that then reduces Phth–SCF3, causing it to fragment into a phthalimide anion and a trifluoromethylthiyl radical. Two of these radicals couple to form a disulfide, which in turn reacts with phthalimide to regenerate Phth–SCF3 and release a trifluoromethanethiolate anion. Loss of fluoride from this anion furnishes thiocarbonyl difluoride, which immediately reacts with the strained nitrogen ring. Subtle changes—such as turning the photocatalyst on or off, or changing the solvent—shift the balance between pathways, allowing the team to favor products bearing either a single fluorine atom or an SCF3 group on the ring.
From Lab Curiosity to Broad Synthetic Platform
Using this platform, the authors prepared dozens of azetidines that contain fluorine or SCF3 groups and often a challenging “quaternary” center, where a carbon is bonded to four different partners. They demonstrated that these thiocarbamoyl fluorides can be further transformed into N–CF3 azetidines using standard fluorination, offering a practical route to a class of compounds with very few existing syntheses. The same light‑driven strategy also works on more conventional amines, converting them directly into thiocarbamoyl fluorides and, from there, into thioureas and other useful derivatives. Because the key reagent Phth–SCF3 is easy to make and handle in air, the overall method avoids many of the safety and waste issues associated with traditional fluorine chemistry.
Why This Matters for Future Medicines
In essence, this work uncovers a new personality for a familiar reagent, revealing that under the influence of light it can quietly generate a powerful, elusive activator for nitrogen chemistry. By taming thiocarbonyl difluoride and channeling it into a mild, modular process, the authors provide a general route to fluorinated azetidines and related compounds that are of high interest in drug discovery. For non‑specialists, the key message is that careful control of electrons and light can turn a once‑limited laboratory curiosity into a practical tool, opening new avenues for building the next generation of medicines.
Citation: Rodríguez, R.I., Paut, J., Armellin, G. et al. Photochemical thiocarbonyl difluoride generation enables azetidine synthesis. Nat Commun 17, 2631 (2026). https://doi.org/10.1038/s41467-026-69464-3
Keywords: photoredox chemistry, fluorinated azetidines, thiocarbonyl difluoride, amine activation, medicinal chemistry