Clear Sky Science · en
Plasmodium ARK1 regulates spindle formation during atypical mitosis and forms a divergent chromosomal passenger complex
How Malaria Parasites Divide in Surprising Ways
Malaria parasites spend their lives shuttling between humans and mosquitoes, multiplying furiously in blood and in the mosquito gut. To do this, they divide their cells in ways that look very different from the textbook diagrams of human cell division. This study uncovers how a key enzyme, called ARK1, controls these unusual division processes and why it could be an attractive target for new antimalarial drugs.
Two Very Different Ways to Multiply
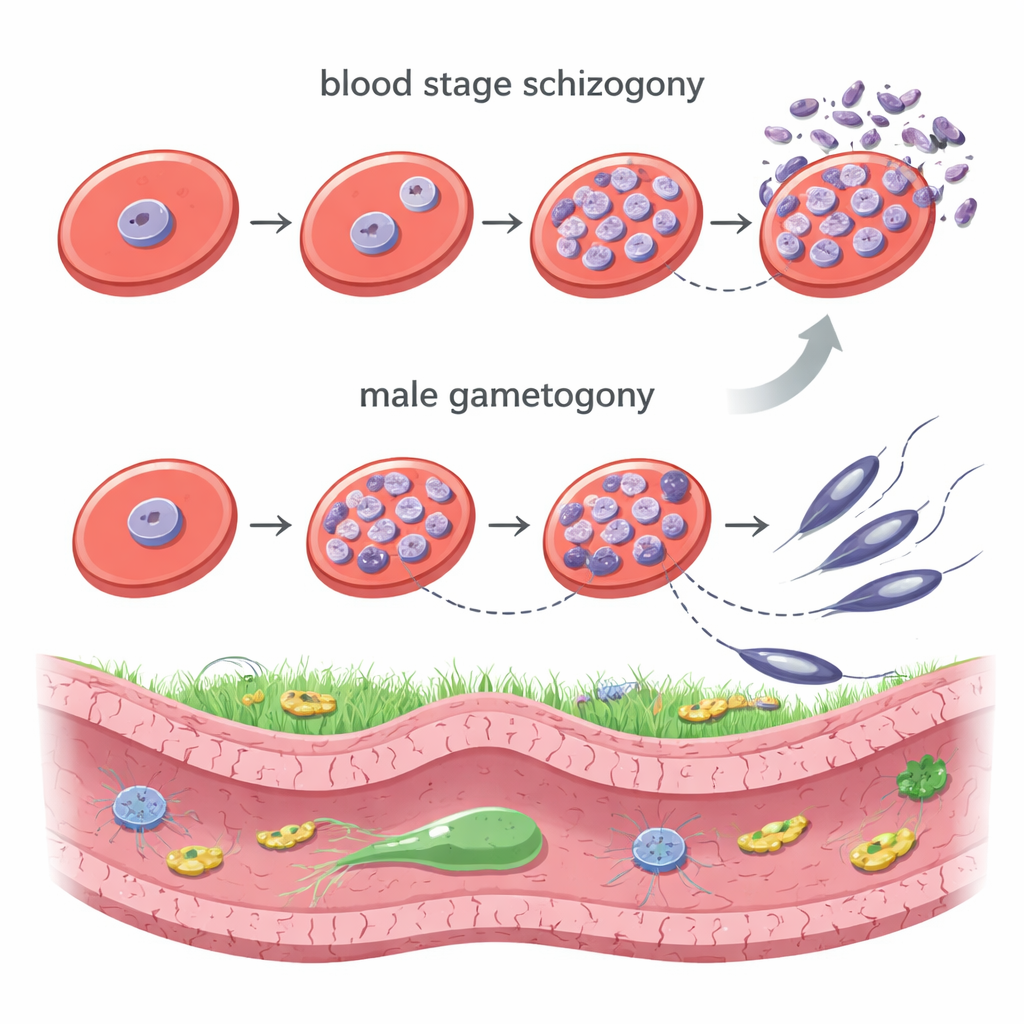
Malaria parasites of the genus Plasmodium use at least two strikingly different styles of cell division. Inside human red blood cells, they undergo schizogony: one parasite nucleus divides many times without the cell splitting, creating a bag full of nuclei that later bud off as dozens of new parasites. In the mosquito, male sex cells go through a lightning-fast division called male gametogony, where the DNA is copied three times in just a few minutes to form an eight-fold genome, and then rapidly packaged into eight sperm-like, flagellated cells. Both forms of division take place inside an intact nucleus and rely on a specialized structure known as a microtubule organizing center (MTOC), which builds the tiny fibers that pull chromosomes apart.
A Master Switch for Building the Division Machinery
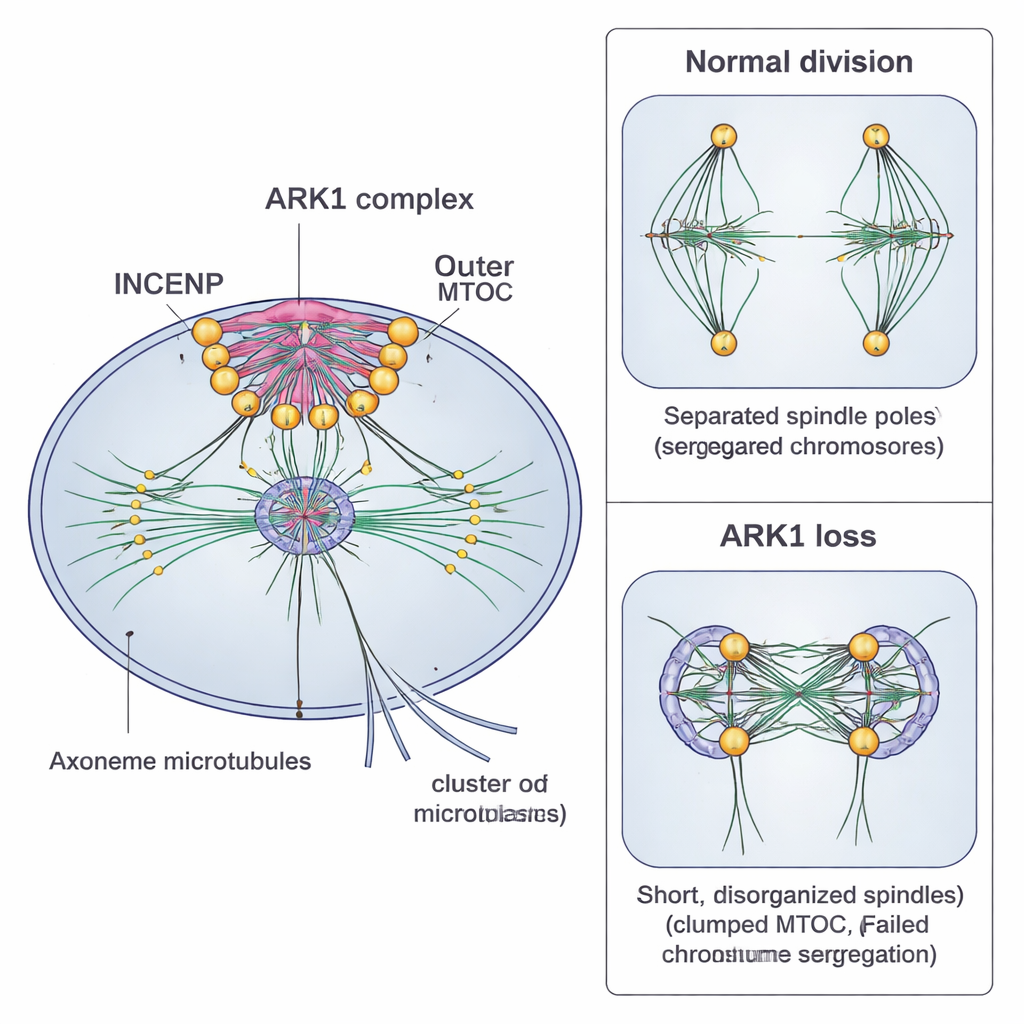
The authors focused on an enzyme called Aurora-related kinase 1 (ARK1), part of a family of proteins that in many organisms act as master switches for cell division. Using genetic tricks in two malaria species—Plasmodium falciparum, which infects humans, and Plasmodium berghei, which infects rodents—they tagged ARK1 with fluorescent markers to see where it goes, and then partially or completely removed it to see what breaks. High-resolution live imaging and expansion microscopy showed that ARK1 appears only when nuclei are actively dividing. It concentrates at the inner part of the MTOC and along the mitotic spindle—the fiber bundle that separates chromosomes—rather than being spread throughout the cell.
What Happens When ARK1 Is Taken Away
When the researchers shut down ARK1 in blood-stage parasites, early development continued, but problems emerged when the parasites tried to divide their nuclei and segment into individual daughter cells. Spindles were stunted or disorganized, nuclei failed to separate correctly, and the resulting clusters of would-be daughter parasites often remained fused or malformed. Under the microscope, key surface and membrane structures that should outline tidy, grape-like bunches of new parasites instead appeared patchy and chaotic. Parasite numbers dropped sharply in the next round of infection, showing that ARK1 is required for efficient growth in blood.
Blocking Transmission Through the Mosquito
The team also reduced ARK1 levels during sexual development in the mosquito. In male gametocytes, which normally produce flagellated sperm-like cells within about 15 minutes after activation, ARK1 knockdown caused multiple failures at once. The inner and outer parts of the MTOC clumped instead of separating, spindles remained short, and the long, whip-like axonemes that power movement were poorly formed. As a result, very few functional male gametes emerged, fewer fertilized eggs developed into ookinetes and oocysts, and far fewer infectious sporozoites reached the mosquito salivary glands. Most mosquitoes carrying these weakened parasites failed to transmit the infection to new mice.
A Rewired Control Complex and New Drug Opportunities
To understand how ARK1 is guided, the researchers pulled it down from parasites and identified partner proteins by mass spectrometry. They discovered that ARK1 forms the core of an unusual “chromosomal passenger” complex with two scaffold proteins, named INCENP-A and INCENP-B. In many other organisms, this complex also includes two additional subunits, Survivin and Borealin, which help target it to chromosomes. Plasmodium and related parasites appear to have lost these components and instead duplicated the INCENP scaffold, rearranging the complex around ARK1 and the inner MTOC. Comparative genome analyses suggest that this rewiring happened repeatedly in different parasite lineages, highlighting how flexible the cell division toolkit can be over evolution.
Why This Matters for Fighting Malaria
For non-specialists, the key message is that malaria parasites divide using control systems that are both essential and different from those in humans. ARK1 sits at the heart of this parasite-specific division machinery, coordinating how chromosomes are separated and how infectious stages are produced in both people and mosquitoes. Because blocking ARK1 disrupts parasite growth in blood and nearly shuts down transmission through mosquitoes, drugs aimed at the ARK1–INCENP complex could, in principle, act at multiple stages of the life cycle. This makes ARK1 a promising candidate for future antimalarial strategies that not only treat disease but also reduce spread.
Citation: Nagar, A., Yanase, R., Zeeshan, M. et al. Plasmodium ARK1 regulates spindle formation during atypical mitosis and forms a divergent chromosomal passenger complex. Nat Commun 17, 1598 (2026). https://doi.org/10.1038/s41467-026-69460-7
Keywords: malaria cell division, Aurora kinase ARK1, Plasmodium mitosis, chromosomal passenger complex, antimalarial drug targets