Clear Sky Science · en
A macrophage-induced subpopulation of mesenchymal cells expressing Fcer1g contributes to wound-induced fibrosis
Why some wounds leave lasting scars
Most of us carry scars from cuts, surgeries, or accidents, but why some wounds heal smoothly while others leave thick, raised marks has remained a puzzle. This study digs into that question by zooming in on the cells that rebuild damaged skin. The researchers uncover a previously unrecognized group of scar‑making cells that switch on early after injury, under the influence of immune cells called macrophages. Understanding this hidden partnership could point the way to treatments that let skin heal with less scarring while still closing wounds efficiently. 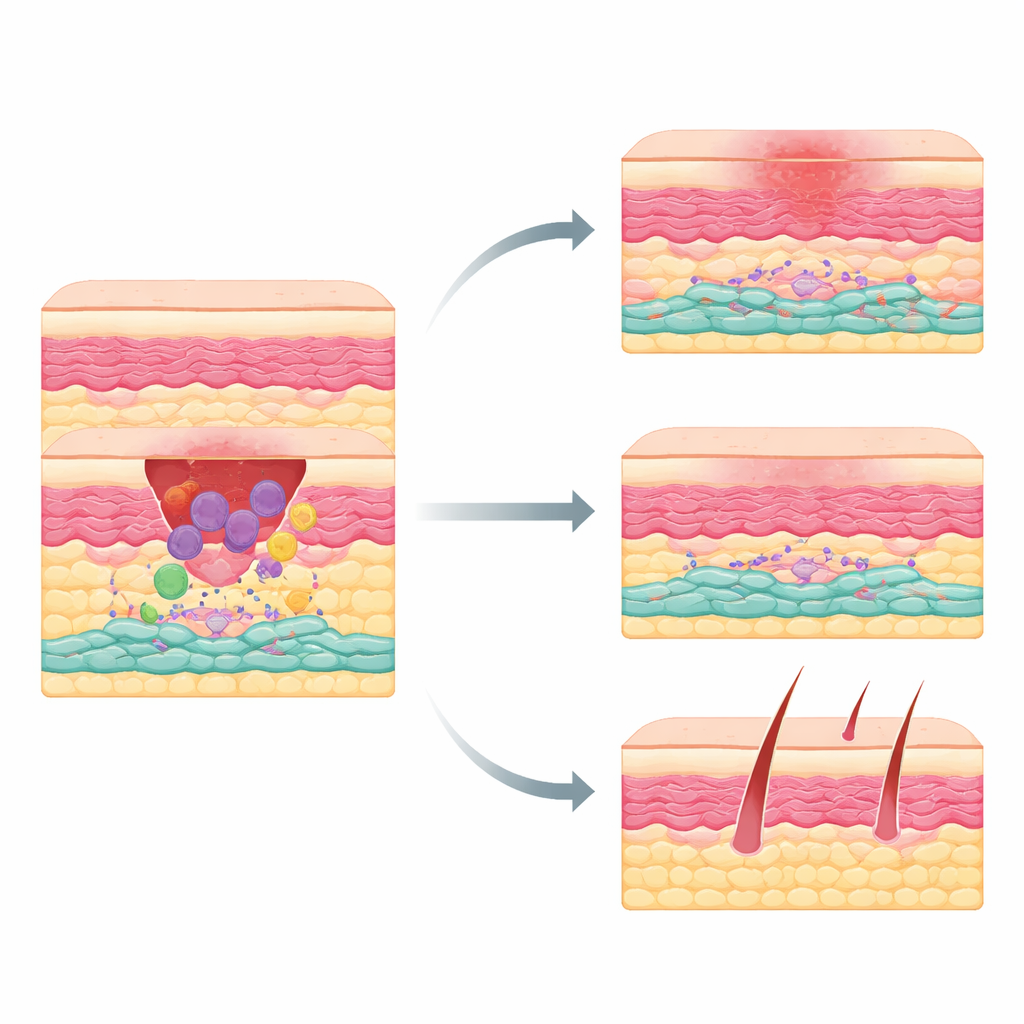
The hidden players inside healing skin
When skin is cut, the body moves through a choreographed sequence: first inflammation to clean up damage, then tissue growth, and finally remodeling. Macrophages arrive early to clear debris and send chemical signals that rally other cells. Among the responders are mesenchymal cells—fibroblast‑like builders that lay down collagen and other support material. These builders are not all the same: some favor regeneration, while others tend to overproduce scar tissue. Until now, scientists had not clearly identified which adult skin mesenchymal cells were the main culprits behind thick scars.
Immune cells that quietly set the stage for scarring
To explore how macrophages influence scarring, the team used mice in which macrophages can be selectively removed during the first few days after wounding. When these early macrophages were depleted, the wounds still closed but formed much smaller scars, with less dense collagen. Single‑cell RNA sequencing—a way to profile gene activity cell by cell—showed that most mesenchymal subgroups were unchanged by losing macrophages. One exception stood out: a distinct subset of PDGFRα‑positive mesenchymal cells that turned on a gene called Fcer1g. This subset nearly vanished when macrophages were missing, suggesting that macrophages are required to create or maintain these cells.
A fast‑growing, scar‑driving cell population
Further experiments mapped where and when these Fcer1g‑positive mesenchymal cells appear. In unwounded skin they were rare, but after injury they rapidly expanded, first around the wound edges and then concentrating in the wound bed, where they made up most PDGFRα‑positive cells by day seven. These cells divided more rapidly than other mesenchymal cells and showed low migration, indicating that local proliferation fuels their rise. When the researchers used a clever genetic system to selectively eliminate cells that expressed both PDGFRα and Fcer1g, wound closure proceeded normally—but scars shrank by more than 20 percent, collagen content dropped by about 30 percent, and the repaired skin’s collagen network looked more like unwounded skin. Hair follicles were more abundant in these depleted wounds, hinting at more regenerative healing. 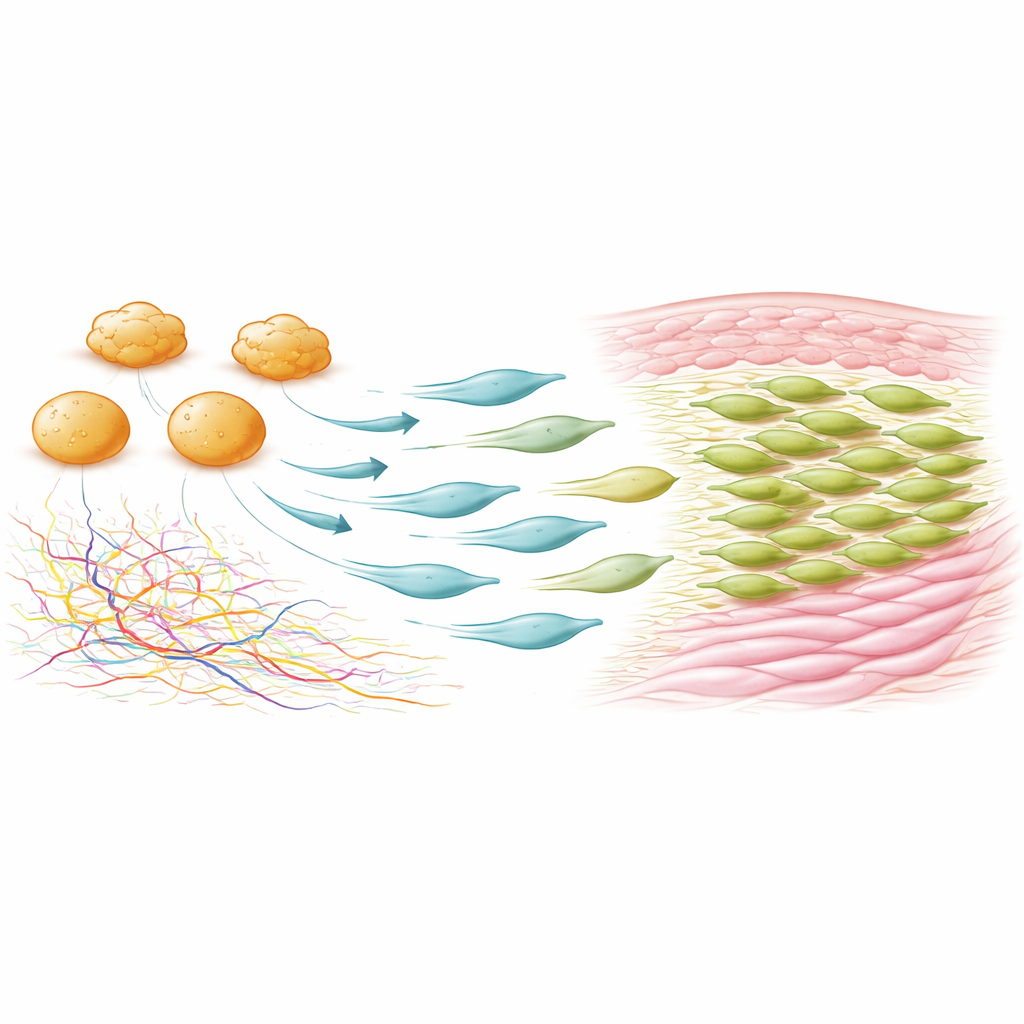
How macrophages signal builders to over‑scar
To understand how macrophages spark this scar‑prone state, the team analyzed likely communication routes between cell types. A leading candidate was fibronectin, a sticky protein that forms part of the temporary matrix in fresh wounds. Macrophages in early wounds were a major source of fibronectin, and its levels fell sharply when macrophages were removed. In mice engineered to lack a wound‑associated version of fibronectin (the EDA form), Fcer1g‑positive mesenchymal cells were greatly reduced and scars were milder. In culture, direct contact with macrophages was enough to convert ordinary dermal fibroblasts into Fcer1g‑positive, PDGFRα‑positive cells, underscoring that macrophages can directly induce this state.
A shared scar program in mice and humans
To see whether this cell type matters in people, the researchers re‑examined existing human skin‑wounding datasets at single‑cell and whole‑tissue resolution. They found a matching mesenchymal subgroup in human wounds that expressed FCER1G and a similar gene signature, peaking about a week after injury—the same timing seen in mice. Across more than one hundred human samples that included normal skin, acute wounds, chronic wounds, and raised scars, the FCER1G‑linked gene program clearly separated injured and scarred tissue from uninjured skin and was strongest early after wounding. This cross‑species similarity suggests that the newly defined cell state is an evolutionarily conserved driver of fibrotic repair.
Toward gentler healing with fewer scars
Taken together, the findings reveal a previously unappreciated chain of events: early‑arriving macrophages enrich fibronectin in the wound, fibronectin helps convert local mesenchymal cells into a rapidly dividing FCER1G‑positive state, and this specialized subset then lays down excess collagen that becomes scar tissue. Because removing these cells in mice lessened scarring without slowing wound closure, they represent an appealing target for future therapies. Drugs or biologics that interrupt the signals that create or sustain this cell state—perhaps by modulating macrophage behavior or the fibronectin environment—could one day help patients heal skin injuries with minimal, less visible scars.
Citation: Ma, X., Wang, E., Puviindran, V. et al. A macrophage-induced subpopulation of mesenchymal cells expressing Fcer1g contributes to wound-induced fibrosis. Nat Commun 17, 2686 (2026). https://doi.org/10.1038/s41467-026-69449-2
Keywords: wound healing, skin fibrosis, macrophages, fibroblasts, scar formation