Clear Sky Science · en
Photosynergetic ligand-exchange for modular synthesis of copper nanoclusters
Building Tiny Copper Worlds
Copper is cheap, abundant, and already central to our energy and electronics infrastructure. This study shows how scientists can now build copper particles made of just a few dozen atoms with almost Lego-like control, using light to swap out the molecules on their surface. Such precision could lead to more efficient catalysts, sensors, and light-harvesting materials that are both affordable and easier to customize than today’s high-end metals like gold and silver.
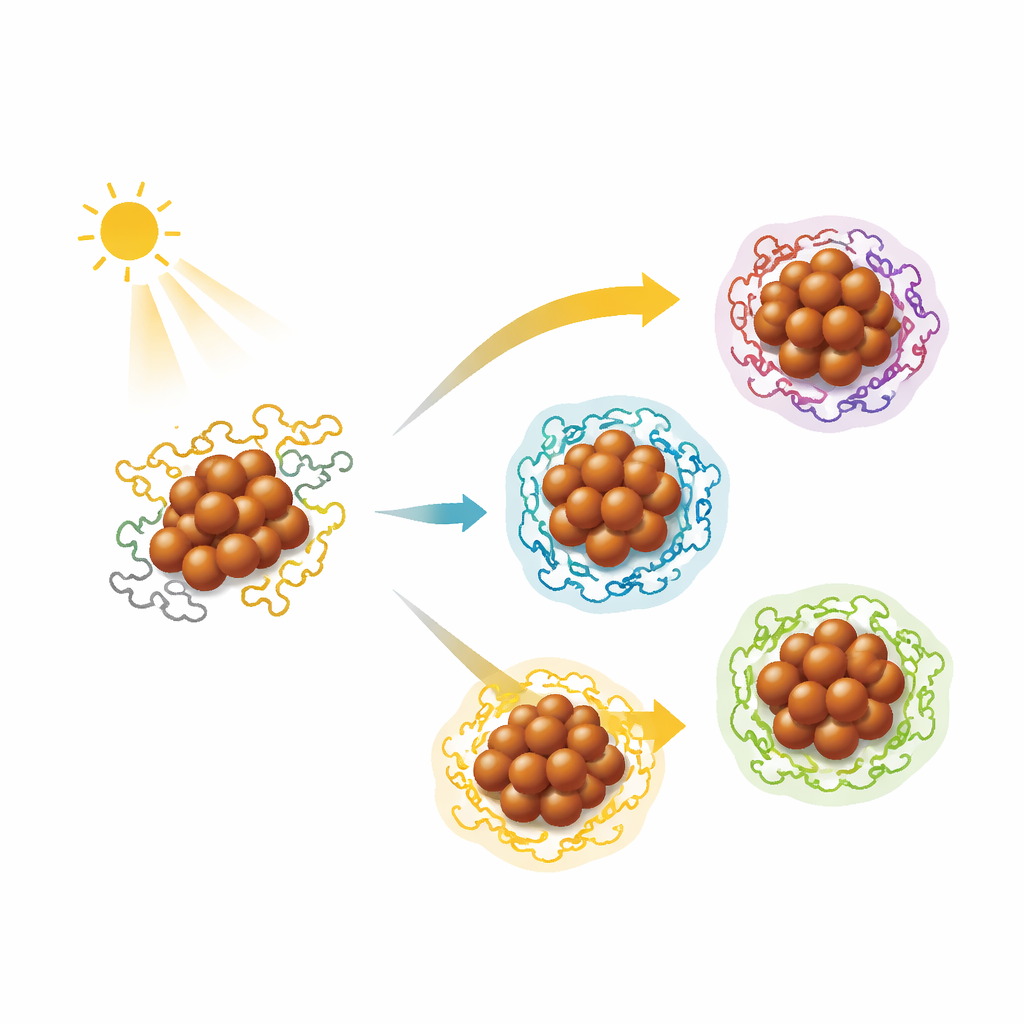
Why Small Copper Clusters Matter
At the nanoscale, metals behave less like bulk solids and more like giant molecules. Clusters of tens of atoms can have sharply defined electronic and optical properties that depend sensitively on their exact size and the molecules bound to their surface. For gold and silver, chemists have learned to make such clusters with atomic precision and to relate their structures to their behavior. Copper clusters promise similar or even broader functionality at far lower cost, but they have been harder to control, particularly when it comes to making families of related structures in a predictable, modular way.
Limitations of Older Ways to Grow Clusters
Traditional methods to make copper nanoclusters either grow them from individual atoms or try to gently modify preformed clusters. In the first route, a chemical or physical trigger causes copper ions to assemble into clusters in the presence of stabilizing molecules. This approach can generate interesting structures but often gives broad size distributions and offers limited freedom to vary the attached molecules. In the second route, known as ligand exchange, chemists start from a well-defined parent cluster and attempt to replace the surface molecules with new ones. For copper, this has proven difficult: exchanges tend to be incomplete, the clusters can fall apart, and the products are often hard to purify and analyze.
Using Light as a Smart Tool
The authors introduce a different strategy they call photosynergetic ligand-exchange. They begin with a robust copper cluster made from 14 copper atoms surrounded by selenium- and phosphorus-containing molecules. This parent cluster is stable in the dark, but when illuminated it partially falls apart into a mixture of tiny copper units, selenium, and organic fragments. Crucially, this breakdown is not random destruction: under light, the cluster becomes reactive enough that when new phosphorus-based molecules are present, the pieces can recombine into new, well-defined copper clusters rather than just decomposing. By carefully tuning the conditions and the added molecules, the team can steer this reassembly toward specific outcomes.
A Library of Designer Copper Clusters
Using this light-assisted pathway, the researchers built a family of 18 different copper nanoclusters, all structurally determined at the atomic level. Many contain 32 or more copper atoms arranged in layered, sandwich-like frameworks stabilized by selenium atoms and various phosphorus-containing ligands; others are smaller or larger variants formed when specialized ligands reshape the metal framework. One striking demonstration is the creation of chiral copper clusters, which exist as left- and right-handed mirror images. By introducing chiral ligands under light, the team coaxed the metal core itself into a twisted arrangement, producing clusters that interact differently with circularly polarized light—an ability that could be useful for advanced optics and sensing.
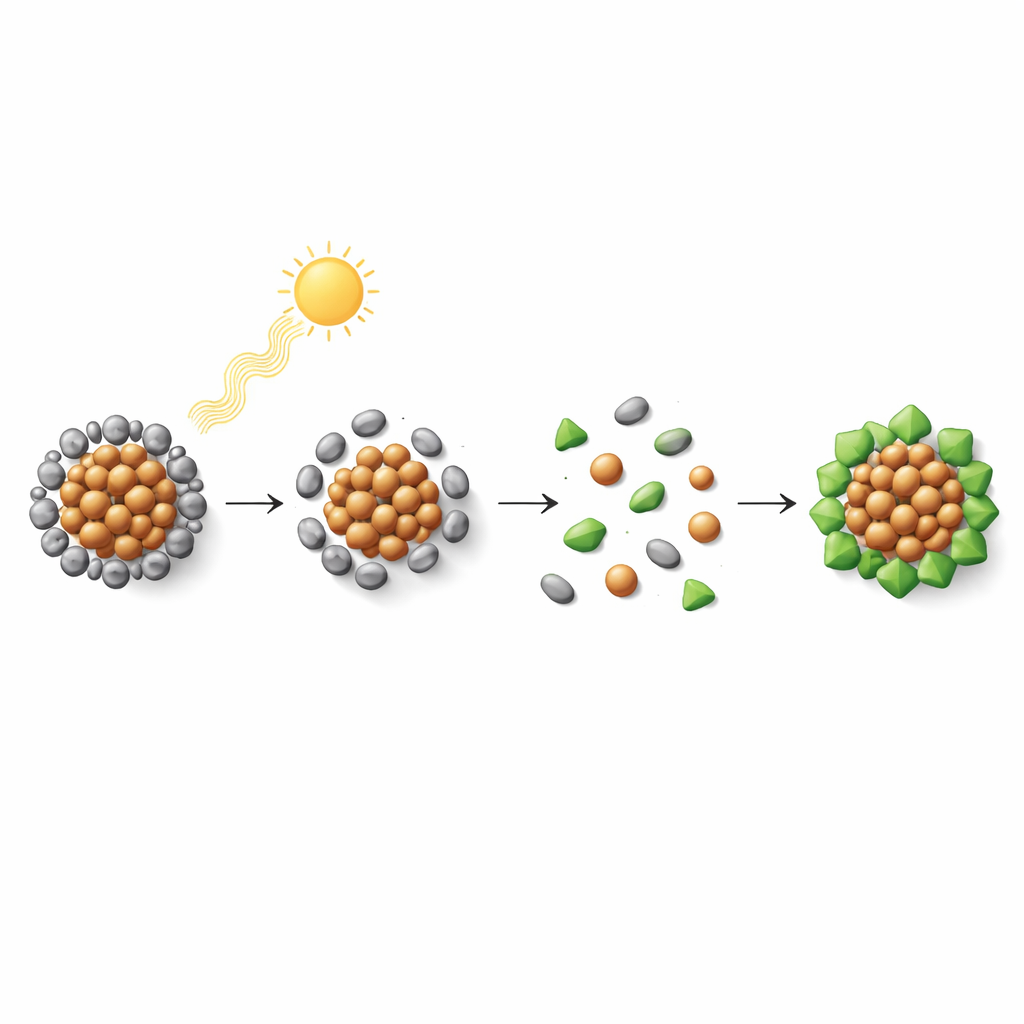
How the Process Unfolds
To understand what light actually does in this system, the authors followed the reaction in real time using a suite of techniques. Ultraviolet–visible spectroscopy showed that the parent cluster’s optical fingerprint vanished and new features appeared as illumination progressed. Mass spectrometry revealed a sequence of intermediate fragments, from partly stripped parent clusters to small copper–selenium units that eventually disappeared as the final products formed. Electron spin measurements confirmed the presence of short-lived radical species produced when light cleaves bonds in the original surface molecules. Piecing these clues together, the team proposes a stepwise pathway in which light first loosens and removes outer ligands, exposes the metal core, breaks it into modular pieces, and then allows these pieces to reassemble around the newly added ligands into stable, redesigned clusters.
What This Means Going Forward
In plain terms, this work turns a single copper cluster into a flexible “starter kit” for building many others. Light acts like a remote control that makes the cluster temporarily malleable, while the choice of surrounding molecules dictates what new structure emerges. Because the starting material is easy to make in bulk and the method tolerates many ligand types, this photosynergetic strategy offers a practical route to tailor copper nanoclusters for specific tasks. The same principles could be extended to other metals, helping chemists design next-generation catalysts, optical components, and energy materials with atomic-level precision at realistic cost.
Citation: Yang, M., Li, Q., Xie, Z. et al. Photosynergetic ligand-exchange for modular synthesis of copper nanoclusters. Nat Commun 17, 2596 (2026). https://doi.org/10.1038/s41467-026-69444-7
Keywords: copper nanoclusters, photochemical synthesis, ligand exchange, chiral nanomaterials, modular nanosynthesis