Clear Sky Science · en
Positive cooperativity between RAS-binding and cysteine-rich domains regulates RAF membrane binding kinetics via lateral rebinding
Why this tiny molecular dance matters
Inside our cells, life-and-death decisions about growth, division, and survival are often made at the surface of the cell membrane. A key player in these decisions is a protein called RAF, which helps relay growth signals and is frequently miswired in cancer. This study uncovers, in unprecedented kinetic detail, how RAF latches onto cell membranes, how long it stays there, and why crowded patches of another protein, RAS, can keep RAF active for longer than expected.
Signals meeting at the cell surface
RAF works in a major signaling highway known as the MAPK pathway, which links outside cues such as growth factors to changes in gene activity. In its resting form, RAF floats in the cell interior in a folded, self-inhibited shape. It is switched on only when it meets RAS, a small molecular switch anchored to the inner surface of the cell membrane. RAS must be in its “on” state and embedded in the right kind of lipids for RAF to be recruited and activated. Because RAF molecules are relatively scarce compared with many other signaling components, exactly how they engage the membrane and avoid premature release strongly influences whether a cell will respond weakly or strongly to a growth signal.
Two hands for grabbing the membrane
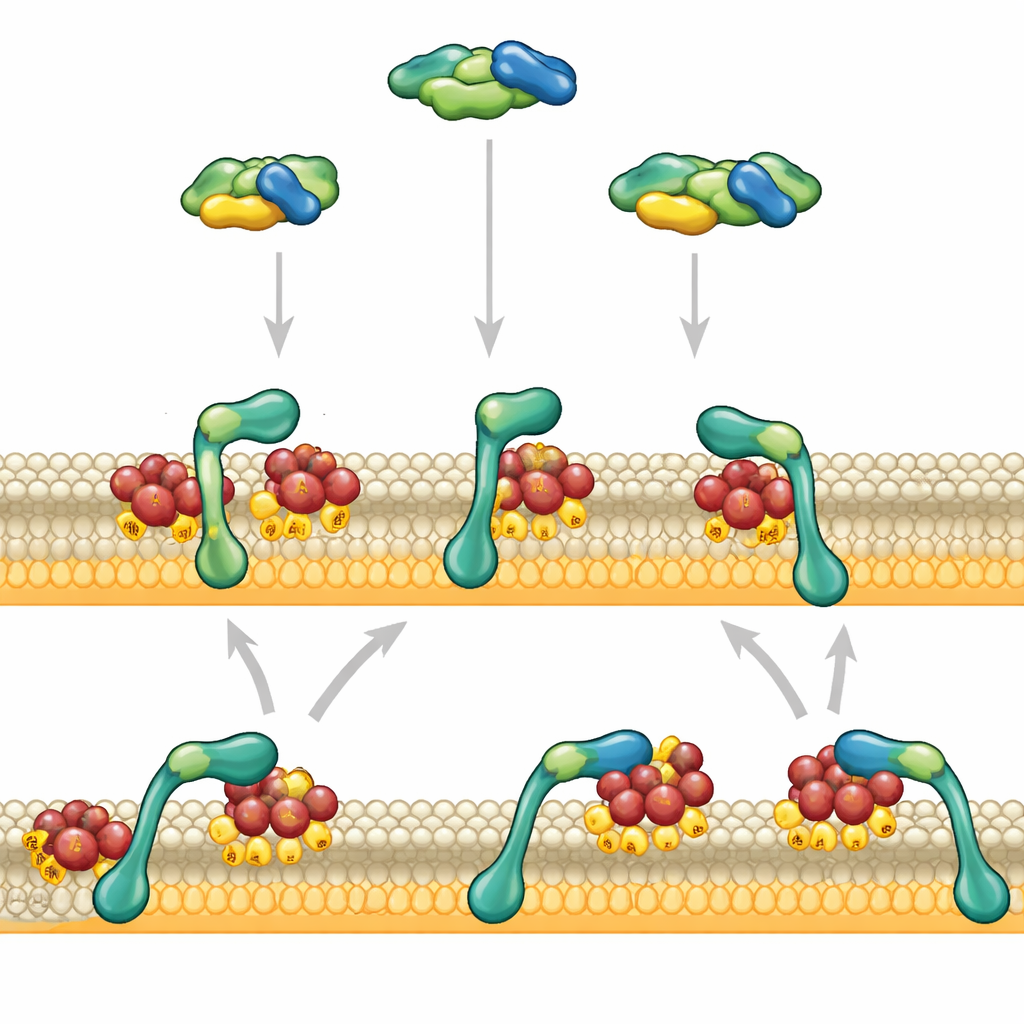
The authors focused on two regions at RAF’s front end: one that grips RAS and one that prefers negatively charged membrane lipids. Using artificial membranes and purified proteins, they watched individual RAF fragments binding to RAS-decorated surfaces with advanced microscopy. When the RAS-binding region acted alone, it touched the membrane briefly and let go within about a second. The lipid-loving region, by itself, barely stuck at all. But when these two regions were linked together, they behaved very differently: RAF now attached strongly and lingered on the membrane for tens of seconds, especially when the membrane carried many negatively charged lipids similar to those in real cells.
Cooperation that slows the exit
This dramatic change arose not because RAF found the membrane faster, but because it left more slowly. First, RAF’s RAS-binding segment recognizes activated RAS and docks RAF at the membrane. Only after this first handshake does the lipid-binding region fully engage surrounding lipids, slowing RAF’s sideways motion and anchoring it more firmly. This second contact in turn stabilizes the original RAS–RAF interaction, creating a positive feedback loop between protein–protein and protein–lipid contacts. Experiments altering the short linker that connects the two regions showed that their tight spatial coordination is crucial: making the linker more flexible or longer weakened RAF’s ability to stay on the membrane.
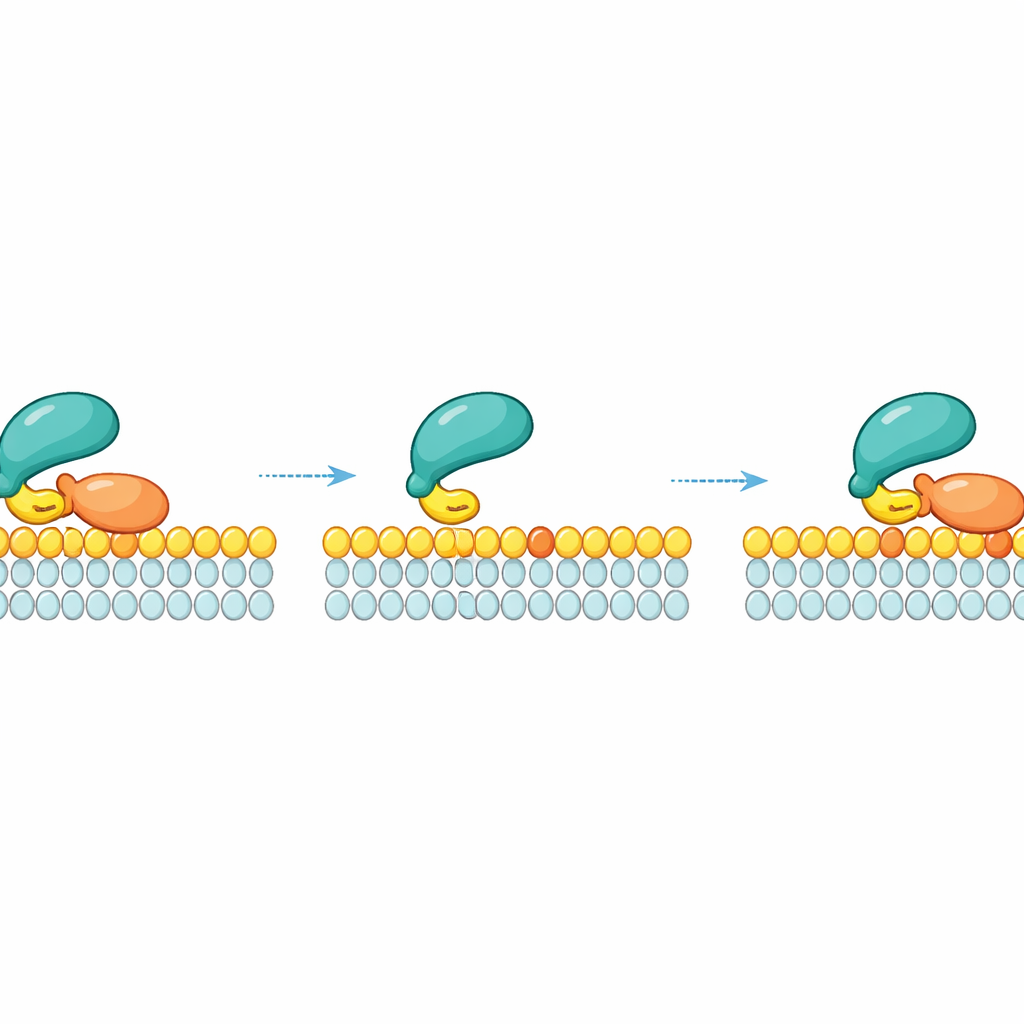
Lateral rebinding: sliding instead of falling off
A key insight of the work is that RAF does not simply detach into the surrounding fluid once it lets go of a particular RAS molecule. Instead, after RAS releases, RAF can remain briefly tethered to the membrane through its weak lipid contact and slide sideways. While in this transient state, it can “regrab” a neighboring RAS molecule on the same membrane patch. This lateral rebinding creates a kinetic safety net: at high local RAS density—such as in nanoclusters where RAS molecules pack tightly—RAF has many chances to re-engage RAS before drifting away. Measurements and simulations showed that the higher the density of active RAS on the membrane, the longer RAF remains membrane-bound due to these repeated local reattachments.
From prolonged visits to reliable activation
The extended time that RAF spends on the membrane has important consequences. RAF activation is not a single event but a multistep sequence that includes shape changes, removal of inhibitory tags, and pairing of two RAF molecules into an active dimer. The study suggests that only RAF molecules that dwell on the membrane long enough can complete this sequence, a form of “kinetic proofreading” that helps cells avoid accidental activation from fleeting, weak signals. By revealing how the interplay between RAS binding, lipid engagement, and lateral rebinding sets RAF’s membrane residency, the work outlines a general strategy by which cells may use many small, weak interactions on membranes to finely tune powerful signaling pathways.
Citation: Jimenez Salinas, A., Tevdorashvili, K., Grim, J. et al. Positive cooperativity between RAS-binding and cysteine-rich domains regulates RAF membrane binding kinetics via lateral rebinding. Nat Commun 17, 2593 (2026). https://doi.org/10.1038/s41467-026-69437-6
Keywords: RAS, RAF kinase, cell membrane signaling, lateral rebinding, kinetic proofreading