Clear Sky Science · en
Dynamic photochromism in cocrystals and tri-state fluorescence switching in films for multilevel optical encryption
Light That Makes Crystals Jump and Messages Appear
Imagine a material that not only changes color when you shine ultraviolet (UV) light on it, but can also crack, jump, and switch its glow in carefully timed steps to hide or reveal secret information. This study introduces just such a smart material, built from organic molecules arranged in crystals and thin plastic films, that combines dramatic motion, vivid color changes, and tunable fluorescence for advanced anti-counterfeiting and optical encryption.
Building Smart Crystals from Simple Molecules
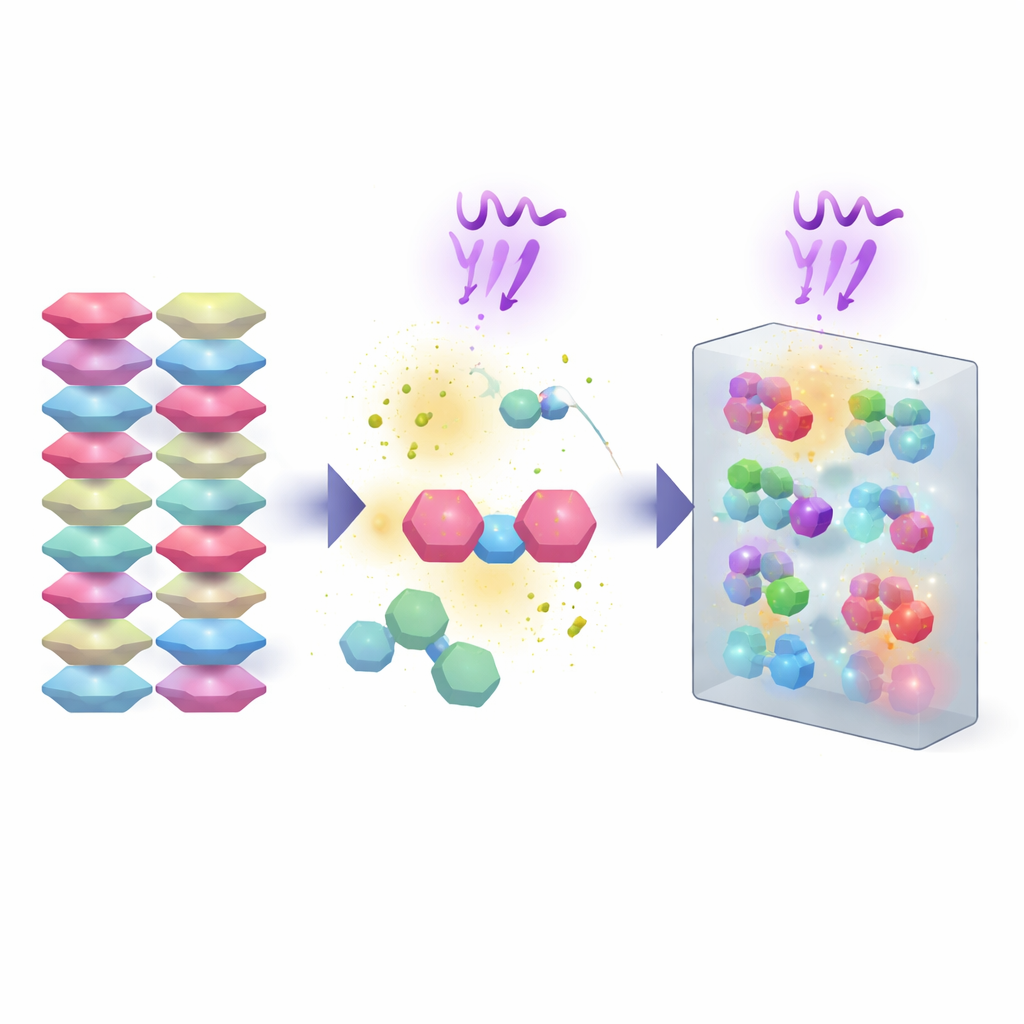
The researchers start by co-assembling two small organic molecules into a single crystal: one is an electron-rich building block (3-aminodibenzofuran), the other an electron-poor partner (octafluoronaphthalene). These molecules stack in an orderly, layered pattern so that donors and acceptors alternate closely, allowing electrons to move between them when excited by light. This careful packing is crucial; it creates a solid that looks like an ordinary transparent plate with blue fluorescence, but it is primed to respond in complex ways once UV light is turned on.
From Colorless Crystal to Jumping, Darkened Fragments
When the cocrystal is exposed to 365 nm UV light, its behavior is striking even to the naked eye. Within seconds, colorless crystals turn brown as their greenish glow fades, showing a strong photochromic response: the material remembers the light by changing color. At the same time, the crystals suddenly split and even leap from the surface, a phenomenon known as the photosalient effect. Detailed measurements show that UV light causes tiny shifts and rotations inside the crystal lattice and generates radical species—highly reactive, short‑lived intermediates. These radicals help two nitrogen atoms from neighboring molecules link together, forming a new azo compound while the fluorinated partner escapes as vapor. As this internal chemical makeover proceeds, built‑up mechanical stress is released as cracking and jumping.
Uncovering the Hidden Reaction Pathway
To understand what is happening inside, the team combines several structural and spectroscopic tools. Single‑crystal X‑ray data taken before and after short light exposures reveal small but directional distortions in the lattice, hinting at growing internal strain. After prolonged irradiation, the final product is isolated and identified as a specific azo molecule formed by coupling two of the original building blocks. Infrared spectra, nuclear magnetic resonance, mass spectrometry, and electron paramagnetic resonance all point to radicals forming under UV light, loss of the fluorinated guest, and the appearance of the new azo bond. Calculations support a stepwise path in which UV light promotes electron transfer between the two components, followed by deprotonation and nitrogen–nitrogen coupling that is energetically favorable, turning a carefully packed crystal into a different, more stable solid.
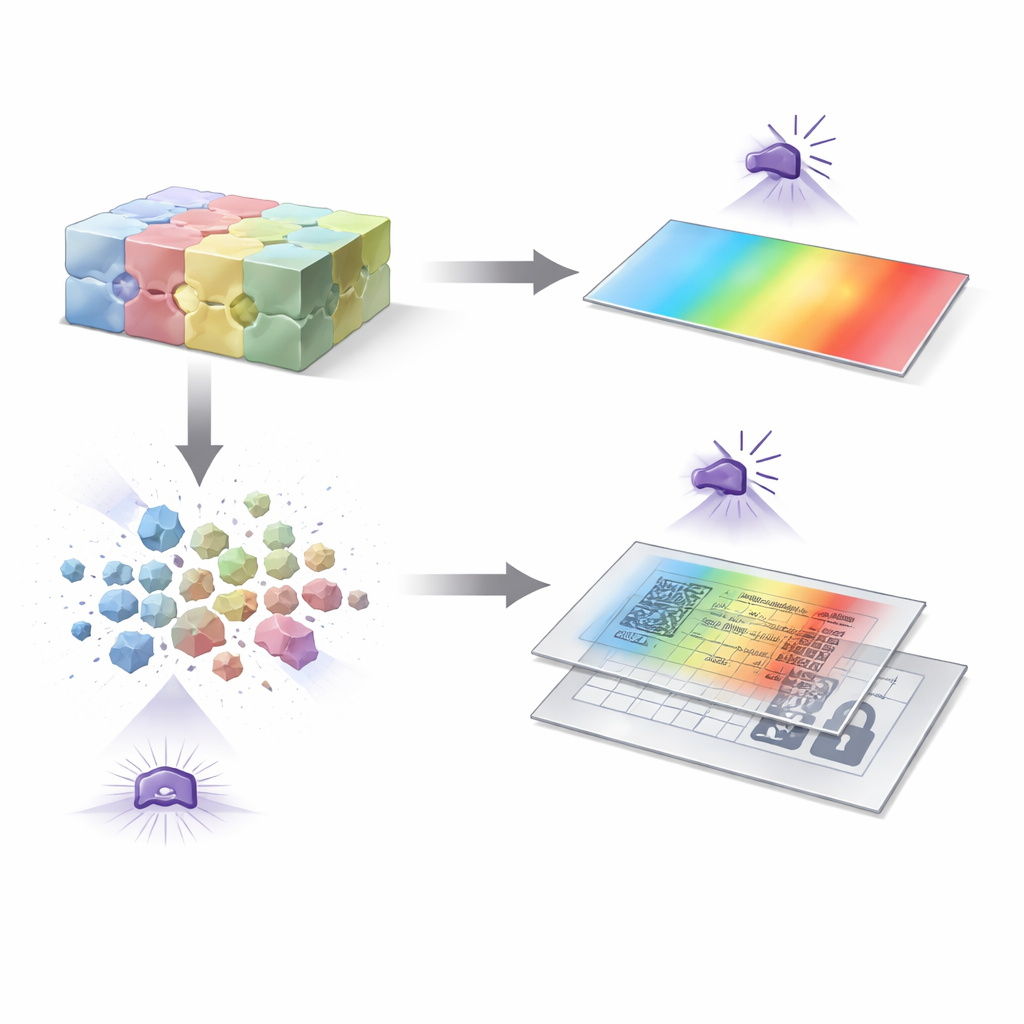
Films That Glow, Shift Color, and Then Go Dark
The same light-sensitive chemistry becomes even more versatile when the photoactive powder is embedded in clear plastic films, such as polymethyl methacrylate (PMMA). In these films, the initial fluorescence is weak, but a short burst of UV light causes it to brighten dramatically as molecular motion and aggregation are subtly rearranged. Next, the film shows excitation‑wavelength‑dependent emission: by changing the color of the light used to excite it, the emitted glow can be tuned smoothly from blue to red. With continued UV exposure, radicals and color centers accumulate, the film darkens, and the fluorescence gradually quenches. This sequence—dim to bright to quenched—provides a built‑in “tri‑state” optical switching scheme in a single material platform.
Hiding and Revealing Information with Light
By patterning UV exposure through masks and controlling irradiation time, the authors demonstrate multi-level optical encryption. A film can look blank in daylight and under UV before activation, then reveal bright fluorescent patterns after a specific exposure, and later show visible color images as photochromism progresses. With further irradiation, both glow and color can be erased, resetting the system. They even design a simple numeric code where an apparent password seen in daylight is misleading, while the true code appears only under UV after the correct exposure window, and is later overwritten by a uniform interference pattern. In short, this work shows how a thoughtfully engineered crystal–polymer system can translate invisible light-triggered chemistry into coordinated color, glow, and motion—offering new routes to secure labels, smart displays, and streamlined synthesis of azo dyes.
Citation: Li, S., Xing, M., Xu, X. et al. Dynamic photochromism in cocrystals and tri-state fluorescence switching in films for multilevel optical encryption. Nat Commun 17, 2556 (2026). https://doi.org/10.1038/s41467-026-69434-9
Keywords: photochromic crystals, light-responsive materials, fluorescence switching, optical data encryption, azo compound synthesis