Clear Sky Science · en
Tailoring sodium and oxygen mixed-ion conduction in the A-site non-stoichiometric NaNbO3-based ceramics
Why this ceramic story matters
As our phones, cars, and power grids lean more on rechargeable batteries and fuel cells, we need solid materials that can move charged particles quickly and safely. This paper explores a specially designed ceramic that lets two kinds of charged atoms, sodium and oxygen, slip through its crystal structure. By learning how to switch between different types of charge flow inside one material, researchers aim to build better solid-state batteries, fuel cells, and sensors that are more efficient and longer lasting.
Designing a smart atomic highway
The authors focus on a perovskite ceramic based on NaNbO3, a crystal structure already known for its rich electrical behavior. They subtly tweak the material by adding a small, fixed amount of calcium and zirconium, and then carefully vary how much sodium is present compared with the ideal formula. This “non-stoichiometric” strategy means the crystal never quite has the textbook ratio of elements. Instead, it holds either too few or too many sodium ions. Those tiny imbalances create defects—missing atoms or extra ones squeezed into small gaps—that reshape how other ions can move. 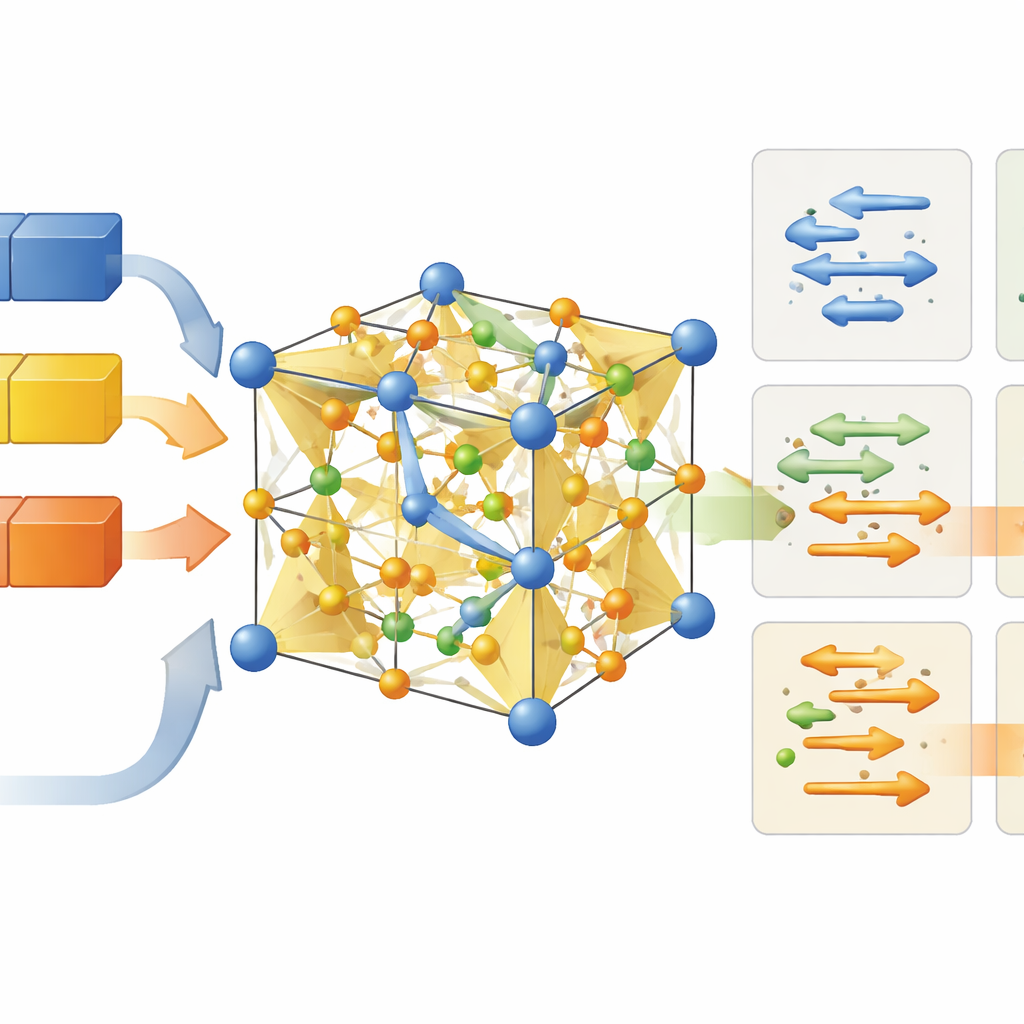
How tiny distortions reshape pathways
Using X-ray diffraction and electron microscopy, the team shows that all of these ceramics keep the same overall crystal framework, an orthorhombic perovskite. What changes is the local geometry. When sodium is lacking, the crystal develops vacancies at sodium and oxygen sites. These missing atoms pull nearby oxygen octahedra—clusters of six oxygen atoms around niobium—into a flattened, distorted shape. When sodium is in excess, extra sodium ions squeeze into the gaps between atoms, stretching and twisting these octahedra in a different way and slightly expanding the channels made from Na–O–Na and Na–O–Nb linkages. In simple terms, the atomic building blocks keep the same basic arrangement, but their angles and spacings flex enough to open or pinch off different pathways for ions to move.
Reading charge flow from electrical fingerprints
To find out which particles are actually moving, the researchers probe the ceramics with impedance spectroscopy, a method that measures how the material responds to an alternating electrical signal over a range of frequencies and temperatures. They combine this with an analysis called the distribution of relaxation times, which helps separate contributions from grains, grain boundaries, and electrodes. By running the tests in nitrogen, air, and pure oxygen, they can tell whether sodium ions, oxygen ions, or electrons dominate under each condition. They also build “sandwich” samples that include a known oxygen-ion conductor to block sodium transport and isolate oxygen motion. Together, these techniques let them chart how conductivity and activation energy change with sodium content and temperature.
Switching between oxygen and sodium highways
The measurements reveal a clear pattern. When the ceramic is sodium-poor, oxygen ions are the main movers, especially once the structure transforms into a high-symmetry cubic phase at elevated temperatures. The flattened oxygen octahedra and abundant oxygen vacancies provide low-energy channels for oxygen ions to hop through. Near the ideal sodium content, the material conducts a mix of oxygen ions and intrinsic electrons, giving a blended conduction mode. When sodium is in excess, however, oxygen motion becomes relatively unimportant. The extra sodium ions expand Na–O–Na and Na–O–Nb networks, widening the “bottlenecks” that sodium must pass through and lowering the barrier for sodium motion. In this regime, sodium ions dominate the conductivity while oxygen-ion transport plays only a minor role. 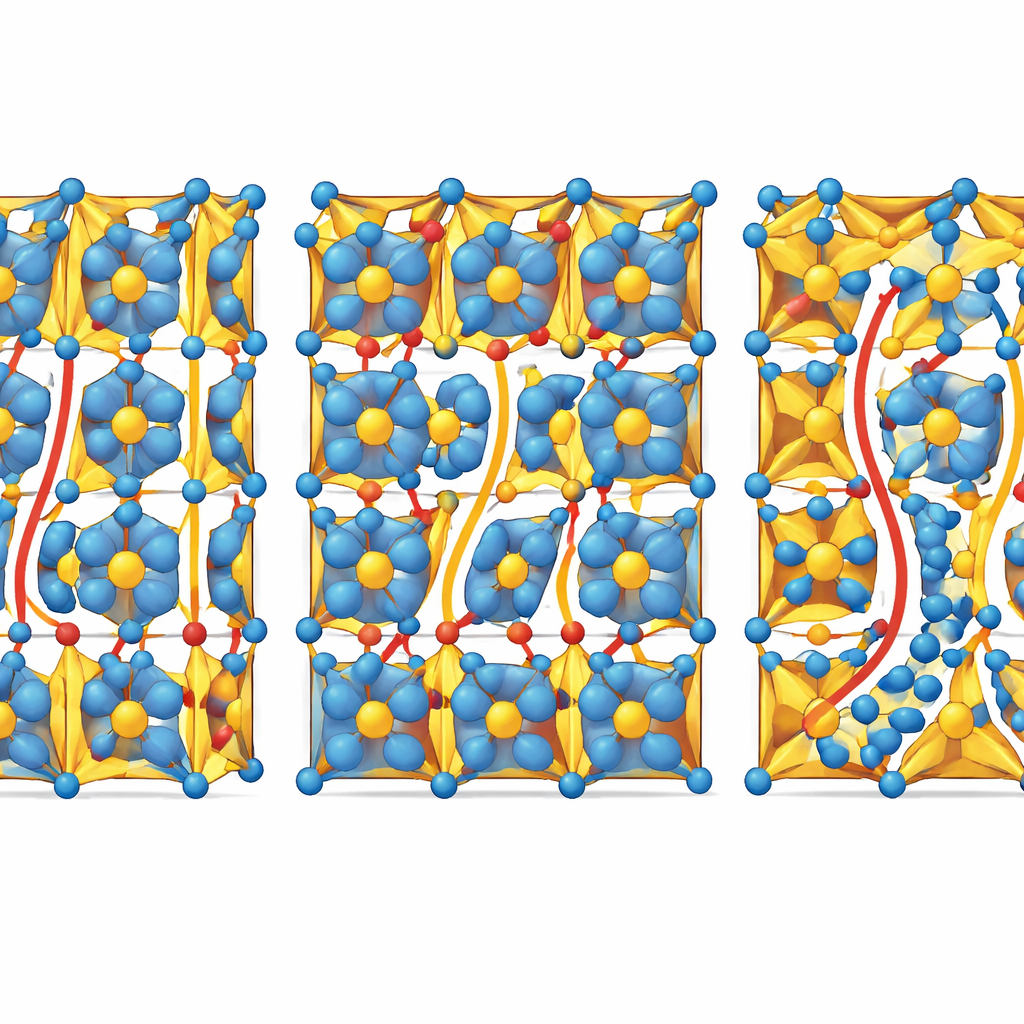
What this means for future energy devices
For a non-specialist, the key takeaway is that tiny, controlled imbalances in a crystal’s composition can be used like a dial to switch which ions move most easily. By understanding how missing atoms, extra atoms, and subtle twists of oxygen cages affect the ease of motion for sodium and oxygen ions, the authors show how to design ceramics that can be tuned for specific jobs—favoring oxygen-ion transport for fuel cells, sodium-ion transport for solid-state batteries, or mixed behavior where both are useful. This work provides a roadmap for engineering ion highways inside perovskite materials, helping guide the search for safer, more versatile solid electrolytes.
Citation: Liu, Z., Xiang, C., Ren, P. et al. Tailoring sodium and oxygen mixed-ion conduction in the A-site non-stoichiometric NaNbO3-based ceramics. Nat Commun 17, 2545 (2026). https://doi.org/10.1038/s41467-026-69428-7
Keywords: solid-state electrolyte, sodium ion conduction, oxide ion conductor, perovskite ceramics, energy storage materials