Clear Sky Science · en
Lack of MDA5 delays hematopoietic aging by modulating inflammaging and proteostasis in mice
Why keeping blood young matters as we age
As we grow older, the system that makes our blood cells slowly wears down. Stem cells in the bone marrow that normally replenish red cells, immune cells, and platelets become more prone to inflammation and more likely to produce certain cell types at the expense of others. This shift contributes to anemia, weakened immunity, and higher cancer risk in old age. The study summarized here explores a surprising player in this process: an antiviral sensor called MDA5. By turning off this single molecule in mice, the researchers found they could keep blood-forming stem cells functionally younger for longer.
A sensor built to fight viruses backfires with age
MDA5 is an innate immune sensor that detects double-stranded RNA, a molecular pattern common in viruses but also generated from our own genomes, especially from mobile genetic elements whose activity increases with age. When MDA5 detects such RNA, it triggers a cascade of inflammatory signals and antiviral responses. The team asked whether this constant low-level activation in older animals might drive “inflammaging” – the persistent, smoldering inflammation that accompanies aging – in the bone marrow. Comparing normal mice to mice lacking MDA5, they measured many immune signaling molecules in bone marrow fluid. Older mice without MDA5 had markedly lower levels of key inflammatory cytokines, including interferon-β and interleukin-1, and showed reduced activation of inflammatory DNA regions in stem cells, indicating a quieter, less inflamed environment.
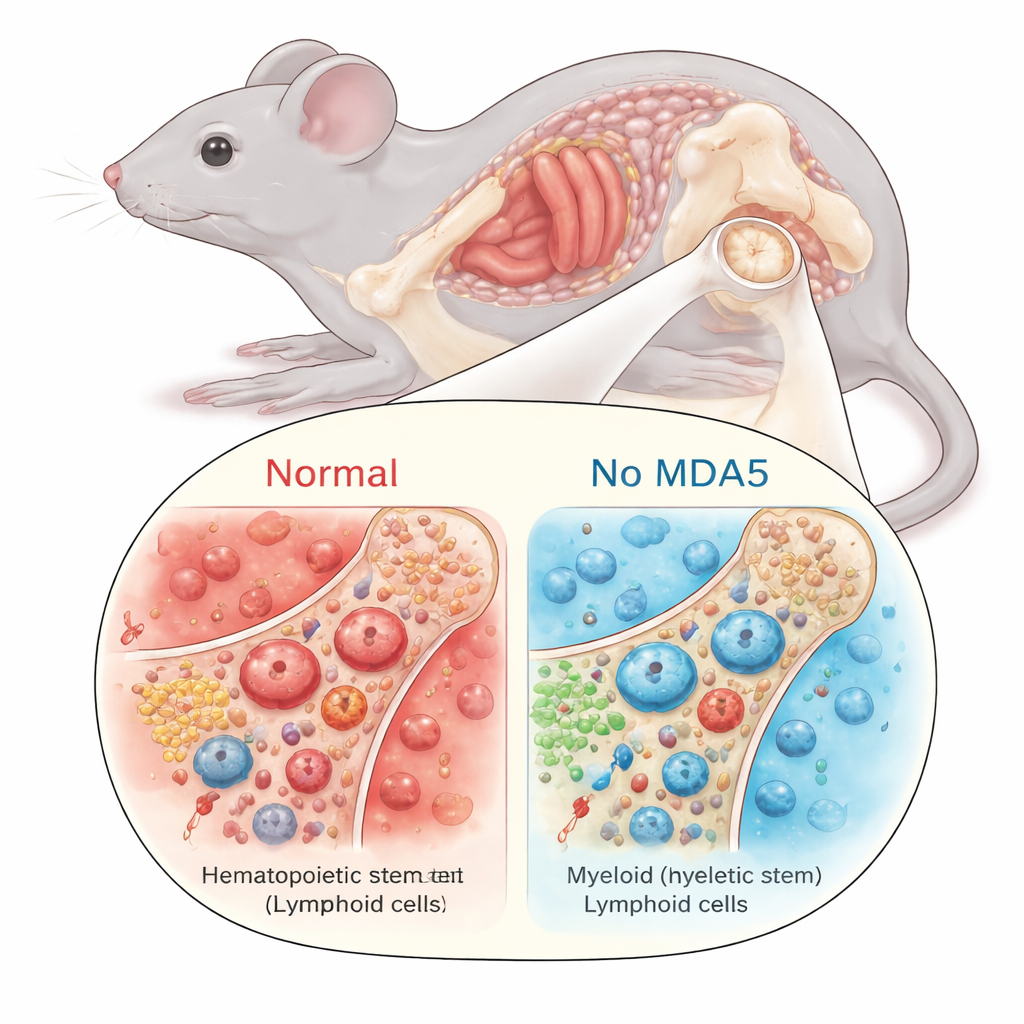
Younger behavior from aging blood stem cells
With less inflammation in the marrow, the blood-forming stem cells behaved differently. In normal aging, these hematopoietic stem cells (HSCs) tend to accumulate in number but decline in quality: they divide more often, lose their long-term regenerative capacity, and develop a bias toward producing myeloid cells (such as certain white blood cells) rather than lymphoid cells that support adaptive immunity. In aged mice lacking MDA5, this typical pattern was blunted. Their HSCs expanded less, showed a reduced myeloid bias, and stayed more quiescent – spending more time in a resting state rather than cycling. When the researchers transplanted equal numbers of stem cells into irradiated recipient mice, aged MDA5-deficient HSCs were better able to rebuild the blood system in non-competitive settings, indicating superior functional reserves compared with age-matched normal HSCs.
Metabolism and protein quality control stay more youthful
Beyond cell counts and behavior, the researchers examined the inner workings of these stem cells. Using a combination of gene expression, chromatin accessibility, metabolite profiling, and single-cell analyses, they found that loss of MDA5 preserved a more youthful metabolic and protein-maintenance profile. Aged MDA5-deficient HSCs had healthier redox markers, such as lower levels of oxidized glutathione and higher levels of NAD and NADP, molecules linked to cellular resilience. Inside these cells, misfolded and unfolded proteins – a hallmark of aging and stress – were reduced, while markers of autophagy, the process by which cells recycle damaged components, were increased. Protein synthesis was dialed down, and amino acids accumulated, consistent with a shift toward careful protein quality control rather than rapid, error-prone production.
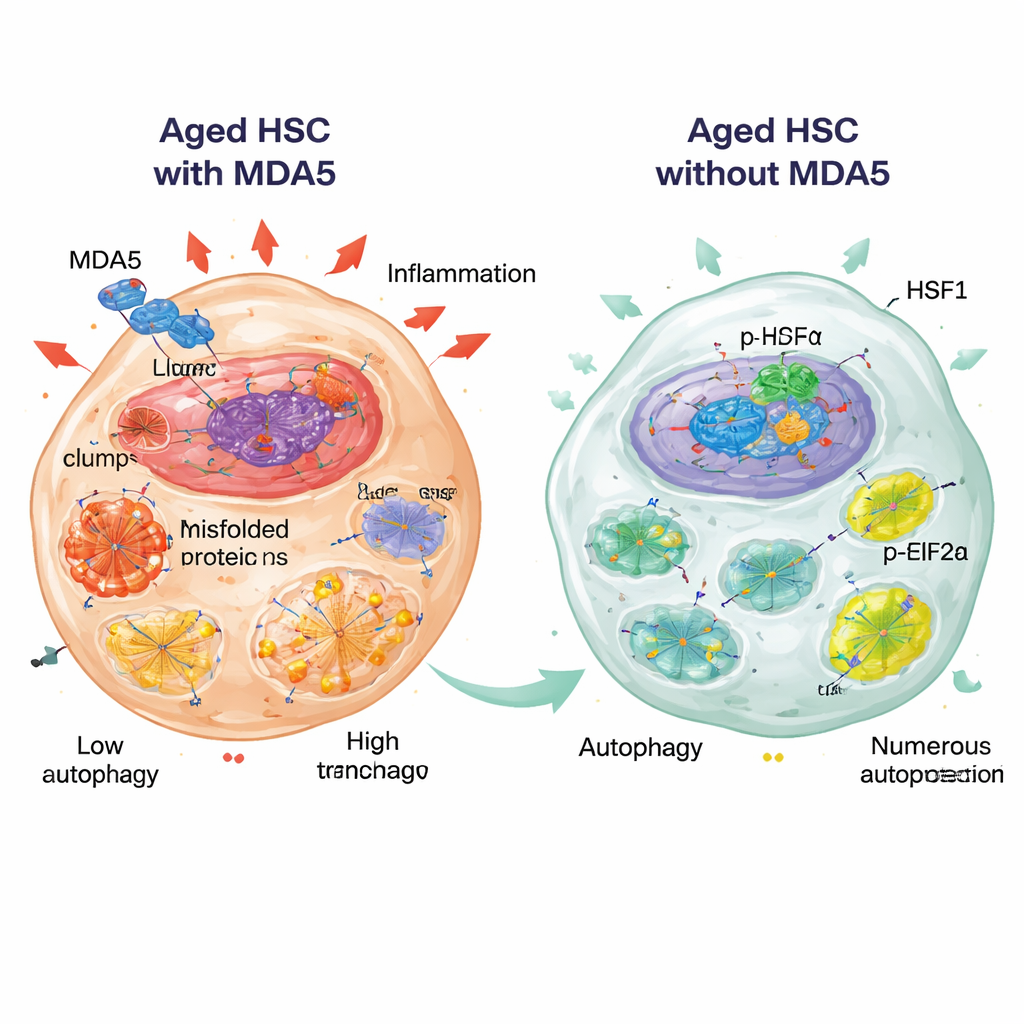
HSF1 and eIF2α: guardians of proteostasis
Two molecular regulators emerged as central to this protective state: HSF1, a master transcription factor that boosts the production of chaperones and other protein-protective factors, and eIF2α, a translation factor whose phosphorylated form (p-eIF2α) slows down protein synthesis during stress. In aged MDA5-deficient HSCs, HSF1 protein levels and nuclear localization were elevated, and p-eIF2α was increased, consistent with an active stress-protection program. The authors found that MDA5 can come into close proximity with HSF1 and, when overexpressed, can retain HSF1 in the cytoplasm, suggesting that active MDA5 may limit HSF1’s beneficial nuclear functions. Importantly, when they pharmacologically activated HSF1 in aged normal HSCs using a small molecule, these cells became more quiescent and better maintained their ability to form colonies over repeated rounds in culture, partially mimicking the youthful features seen in the absence of MDA5.
What this means for healthy aging of the blood
Together, these findings paint a picture in which chronic activation of an antiviral sensor, MDA5, gradually pushes blood-forming stem cells toward inflammation, metabolic stress, and protein damage. Removing MDA5 in mice interrupts this cycle: inflammatory signals in the marrow are dampened, HSCs remain more rested and less biased, and their internal protein quality-control systems stay robust. While directly disabling MDA5 in people would be risky because of its role in fighting infections, this work highlights the broader principle that targeting age-related inflammation and bolstering proteostasis could help preserve blood stem cell function in later life. In the long term, safer ways to modulate MDA5-related pathways or to boost HSF1 and eIF2α-driven stress responses may offer strategies to extend the healthy span of our blood and immune systems.
Citation: Bergo, V., Bousounis, P., To Vu, G. et al. Lack of MDA5 delays hematopoietic aging by modulating inflammaging and proteostasis in mice. Nat Commun 17, 1645 (2026). https://doi.org/10.1038/s41467-026-69424-x
Keywords: hematopoietic stem cells, inflammaging, MDA5, proteostasis, aging blood