Clear Sky Science · en
Negative feedback regulation of STING signaling by TAX1BP1-directed Golgiphagy
How Cells Avoid an Immune Overreaction
Our immune system must walk a tightrope: it has to recognize invading viruses and damaged DNA quickly, but it also needs to shut that alarm down before runaway inflammation harms healthy tissues. This study uncovers how a little-known cellular helper, TAX1BP1, acts as a built-in brake on a powerful alarm pathway called cGAS–STING. By explaining how cells dismantle this alarm once it has done its job, the work sheds light on autoimmune disease, antiviral defense, and possible cancer therapies.
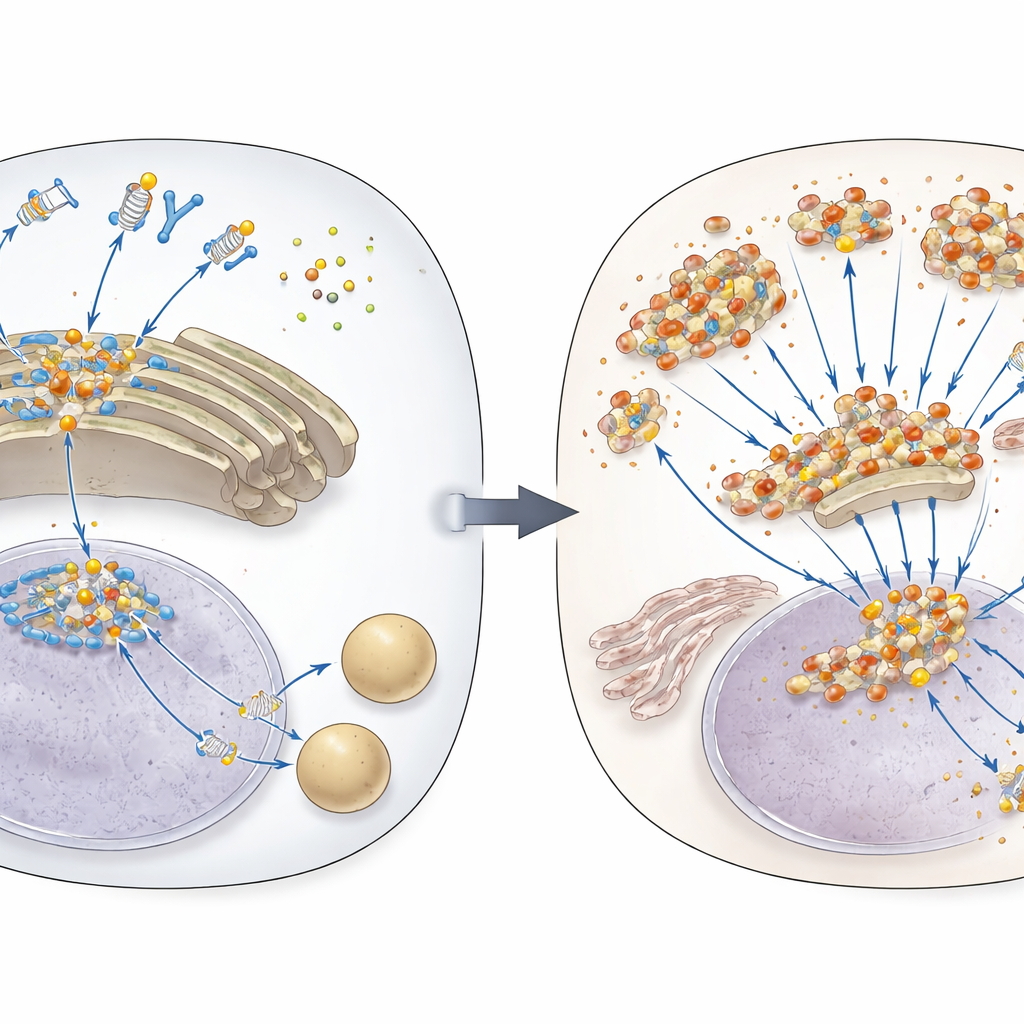
The Cell’s DNA Alarm System
When foreign or misplaced DNA appears in the fluid inside our cells, a sensor protein called cGAS detects it and produces a small messenger molecule. That messenger flips on STING, a switch-like protein that lives on internal membranes. STING then travels to a cellular shipping hub called the Golgi, where it clusters and triggers the production of antiviral molecules known as type I interferons, along with other inflammatory signals. Because this response is so potent, cells normally degrade STING after a short burst of activity, routing it into cellular recycling centers called lysosomes. Until now, how STING was targeted for this shutdown step had only been partly understood.
A Cellular Brake with Many Jobs
TAX1BP1 was previously known as a multitasker that helps the cell’s waste-disposal system, autophagy, recognize unwanted material. It can bind both to cargo tagged for removal and to the forming sacs that will engulf that cargo. The authors asked whether TAX1BP1 might also help restrain the cGAS–STING alarm. Using human immune-like cells and mouse macrophages lacking TAX1BP1, they found that activating cGAS or STING produced far more interferon and inflammatory cytokines than in normal cells. These TAX1BP1-deficient cells were also more resistant to infection by herpes simplex virus, consistent with their supercharged antiviral signaling.
Cleaning Up STING and a Stressed Golgi
Diving deeper, the researchers showed that without TAX1BP1, STING stayed active for longer and was broken down more slowly. Microscopy and biochemical tests revealed that active STING forms large aggregates and that these clumps accumulate when TAX1BP1 is missing. At the same time, the Golgi structure swells and fragments into pieces. The team discovered that TAX1BP1, together with another recycling helper called p62, targets these damaged Golgi fragments and the STING clusters on them for destruction in lysosomes—a specialized form of clean-up the authors refer to as “Golgiphagy.” This process is distinct from the classic, bulk form of autophagy and instead uses more selective pathways to grab the overloaded Golgi pieces.
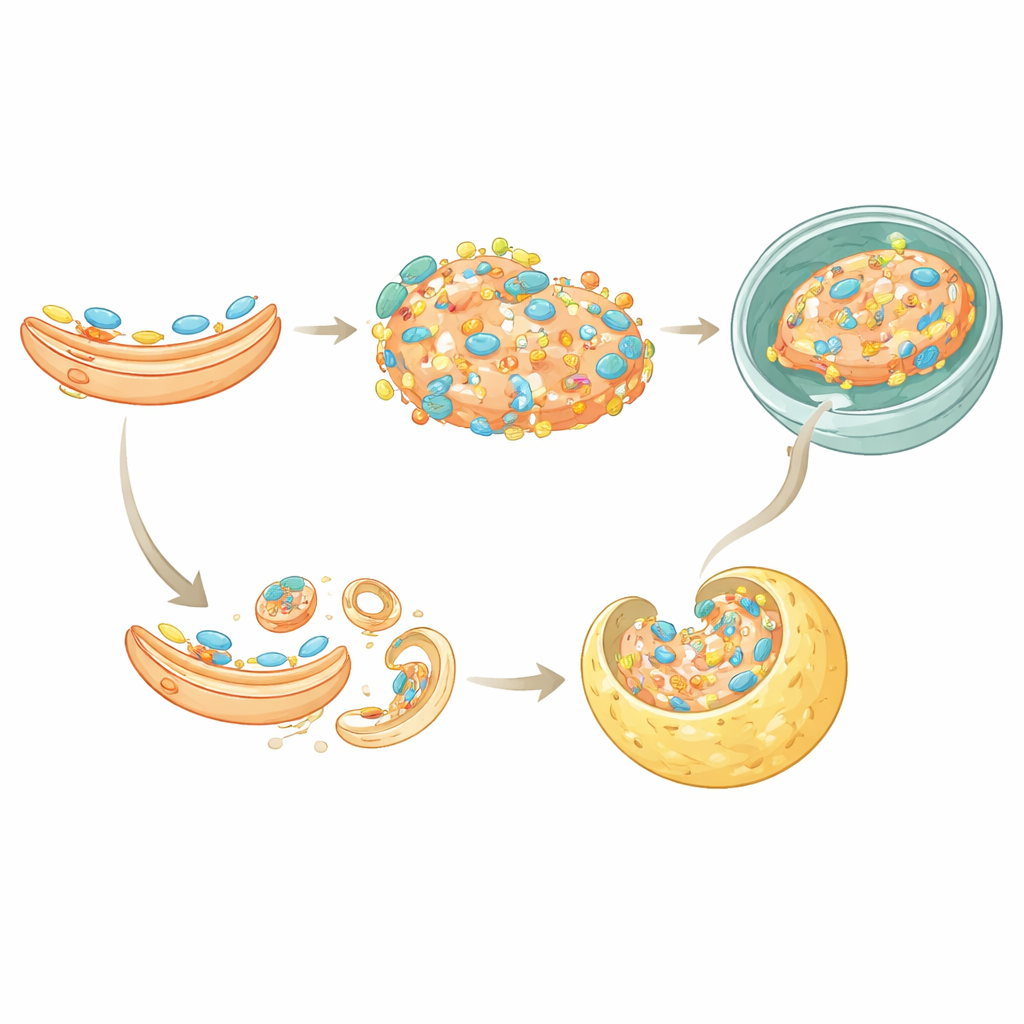
Two Routes to Turning Off the Alarm
The study also uncovered a second way TAX1BP1 helps silence STING. Normally, once STING has signaled at the Golgi, it is packaged into small vesicles that bud off and are recognized by a protein-sorting system called ESCRT, which then delivers STING into lysosomes. TAX1BP1 binds directly to STING and helps it interact with a key ESCRT component, HGS. When TAX1BP1 was removed, STING’s handoff to this sorting machinery was less efficient, and its degradation slowed. However, some breakdown still occurred, indicating that TAX1BP1 works alongside p62 in this microautophagy route, giving cells partially redundant ways to ensure that STING does not linger too long.
Why This Matters for Health and Disease
To test the importance of this brake in living animals, the researchers engineered mice lacking TAX1BP1 specifically in certain immune cells. When stimulated with a STING-activating drug, these mice had higher levels of interferon and inflammatory cytokines in their blood than normal mice. When infected with a poxvirus, they carried less virus, showing enhanced resistance. Together, these findings suggest that TAX1BP1 couples the moment of STING activation at the Golgi to a timed shutdown through targeted destruction of both STING and damaged Golgi membranes. For a layperson, the takeaway is that cells not only sound an alarm when they detect dangerous DNA, they also actively dismantle parts of their own internal wiring to be sure the alarm switches off. Understanding and potentially tuning this internal brake could help in designing treatments for chronic inflammatory diseases, improving antiviral defenses, or modulating immune responses in cancer.
Citation: Suklabaidya, S., Mohanty, S., Reider, I.E. et al. Negative feedback regulation of STING signaling by TAX1BP1-directed Golgiphagy. Nat Commun 17, 2762 (2026). https://doi.org/10.1038/s41467-026-69422-z
Keywords: innate immunity, STING pathway, autophagy, Golgi apparatus, antiviral response