Clear Sky Science · en
Cell wall hydrolysis promotes a second wave of transpeptidation to achieve cell separation following septation in Bacillus subtilis
How Bacteria Finish Splitting in Two
Bacteria divide astonishingly fast, yet manage to build and rebuild their tough outer shell without bursting. This study uncovers a hidden final step in how the soil bacterium Bacillus subtilis finishes cell division. The work shows that after a new wall has already formed between two future daughter cells, the wall is first cut and then subtly re-stitched from within. Understanding this extra step helps explain how bacteria maintain their shape and toughness—and may point to new ways to disrupt harmful microbes.
Building a Wall, Then Letting Go
When a rod-shaped bacterium like Bacillus subtilis divides, it first builds a flat internal wall, called a septum, that separates the mother cell into two compartments. This wall is made of peptidoglycan, a mesh of sugar strands tied together by short peptides, forming a protective cage around the cell. In many bacteria with two membranes, construction and cutting of this wall happen at the same time. But in Bacillus subtilis, which has a single thick outer wall, the process is split in two: the septum is built completely, and only later is it opened to let the daughter cells separate into individual rods.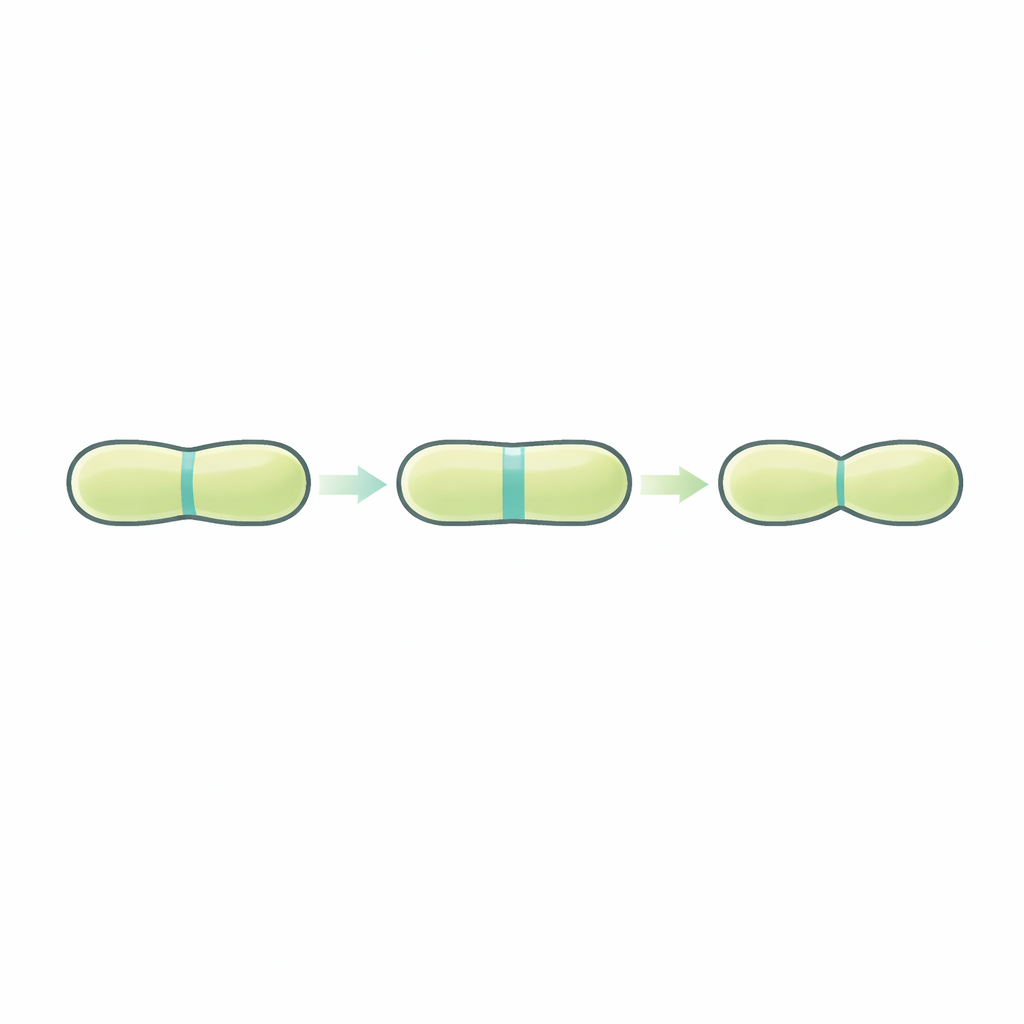
Tracking Hidden Construction Work
The authors used special fluorescent building blocks that mimic the natural components of the cell wall. These probes glow when enzymes attach them into the peptidoglycan mesh, allowing researchers to see where and when linking reactions occur. With high-resolution three-dimensional microscopy, they followed these signals in thousands of cells. They confirmed a first wave of activity as the septum grows inward from the cell’s outer edges to the center, forming a complete flat plate. Unexpectedly, they then observed a second, later wave of crosslinking activity that starts at the rim of the finished septum and travels inward as the cells begin to separate.
Remodeling Without Adding New Material
One key question was whether this late activity reflected the insertion of fresh cell wall material, or simply a rearrangement of what was already there. To find out, the team labeled a soluble precursor that must be used when new strands are added. They saw this precursor only during the early building of the septum, not during the later separation stage. By combining multiple fluorescent labels, they showed that the second wave of activity uses existing, previously unlinked peptide chains as raw material. In other words, once the septum is built, the cell does not thicken it further; instead, it cuts and then re-crosslinks the same mesh to strengthen the newly forming end poles of the daughter cells.
Cutters and Stitchers Working Together
The study pinpoints two main players in this remodeling. The enzyme LytF acts as a cutter, slicing bonds in the wall to open the septum. Another enzyme, PBPH, is a transpeptidase that stitches peptide chains together. Using mutants lacking different wall-cutting enzymes, the authors showed that when LytF’s activity is removed or disabled, the second wave of crosslinking largely disappears and cells remain linked in long chains. Likewise, when PBPH is missing, cells show strong defects in separation and almost no late crosslinking at complete septa. Imaging of fluorescently tagged proteins further revealed that LytF arrives at the septum only after an earlier division scaffold has left, and that PBPH’s continued presence at separating sites depends on LytF’s cutting action. Together, these observations support a model in which carefully timed wall breakdown by LytF exposes or rearranges peptide chains that PBPH can then crosslink to reinforce the nascent poles.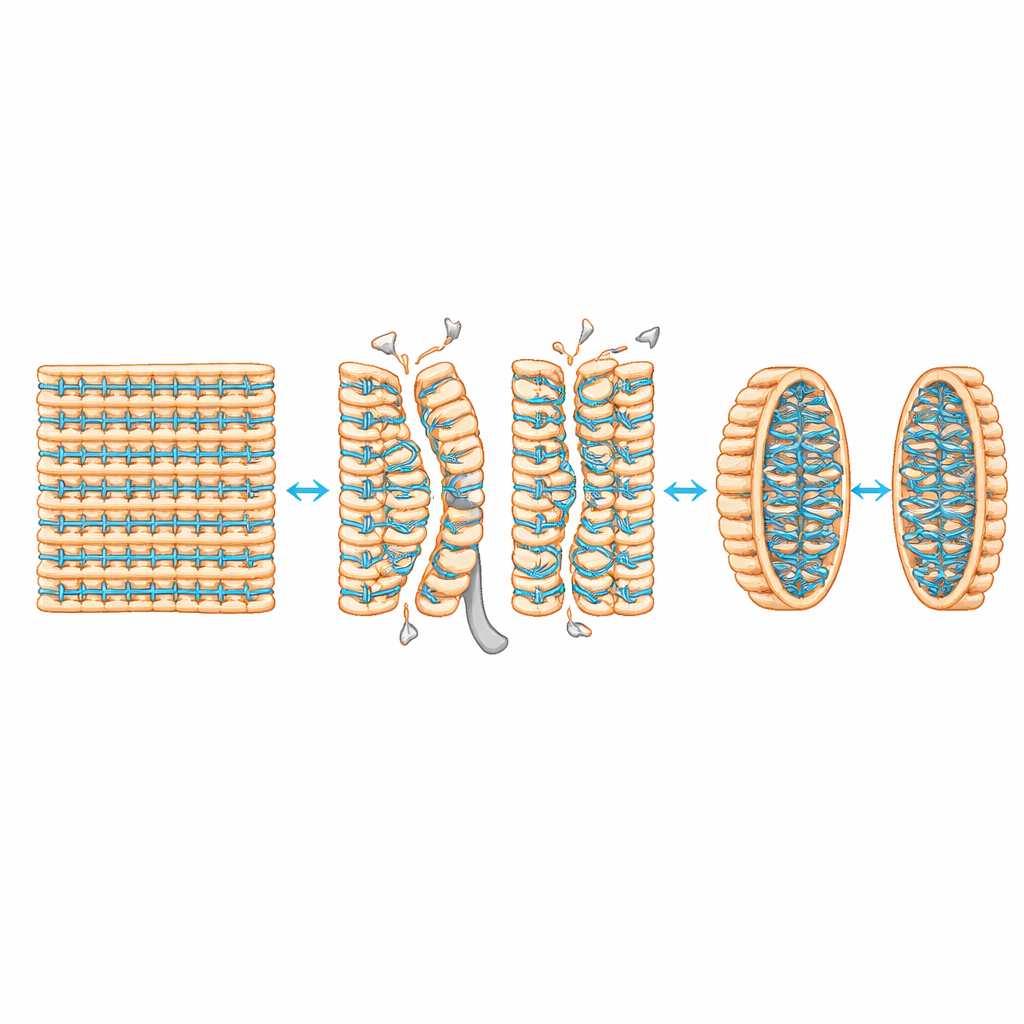
Why the Second Wave Matters
As the flat septal wall is reshaped into two rounded poles, the mechanical load on the peptidoglycan changes dramatically: instead of being pressed from both sides, the new poles must now withstand internal pressure from within the daughter cells. The authors propose that the second wave of crosslinking stiffens and stabilizes the wall just as it is being cut and curved, preventing rupture. Because many other Gram-positive bacteria divide in a similar two-step fashion—first completing a septum, then separating—this remodeling wave may be a general feature of how these microbes safely finish division. By exposing this hidden final step, the study refines the textbook picture of bacterial cell division and suggests fresh targets for antibiotics that aim to weaken the bacterial wall at its most vulnerable moment.
Citation: Patel, V., Hsu, YP., Debnath, M. et al. Cell wall hydrolysis promotes a second wave of transpeptidation to achieve cell separation following septation in Bacillus subtilis. Nat Commun 17, 2689 (2026). https://doi.org/10.1038/s41467-026-69404-1
Keywords: bacterial cell division, peptidoglycan, Bacillus subtilis, cell wall remodeling, transpeptidase