Clear Sky Science · en
The photolyase/cryptochrome of Aspergillus nidulans senses oxidative stress and shuttles from nuclei to mitochondria
How Fungi Sense Light and Stress
Sunlight keeps plants and fungi alive, but it also brings dangers like DNA damage and harmful oxygen-based chemicals. This study explores how a common mold, Aspergillus nidulans, uses a single protein, called CryA, to sense both light and oxidative stress and to protect itself. Understanding this "dual sensor" not only deepens our knowledge of how microbes cope with harsh environments, but may also shed light on how cells in general coordinate signals between their nucleus and their energy-producing mitochondria.
A Blue-Light Repair Enzyme with a Hidden Role
CryA belongs to a family of proteins known for repairing DNA damage caused by ultraviolet light. These proteins, called photolyases and cryptochromes, use a light-absorbing molecule (a flavin) to harness blue light and fix broken DNA bases. The researchers confirmed that CryA has the classic architecture of a DNA repair enzyme, binds the usual light-harvesting cofactors, and clusters with known photolyases in evolutionary trees. On that basis alone, CryA would seem like a routine repair tool. Yet earlier work had hinted that it also influences how the fungus develops, behaving more like a light-regulated switch than a simple molecular mechanic.
A Master Dimmer for Light-Responsive Genes
To uncover CryA’s regulatory side, the team tracked where it sits in the cell and how changing its amount alters fungal growth. They found that under normal conditions CryA accumulates in the nucleus, where the DNA is stored. When they removed the cryA gene, the fungus produced more sexual structures; when they forced extra CryA to be made, it almost completely blocked the formation of the usual asexual spores, leaving pale, fluffy colonies. Gene expression tests showed that many light-activated and development-related genes switched on too strongly without CryA and too weakly when CryA was overproduced. Together, these results reveal CryA as a negative-feedback element: light raises cryA levels, CryA then moves into the nucleus and reins in light- and development-induced genes, preventing the response from running out of control. 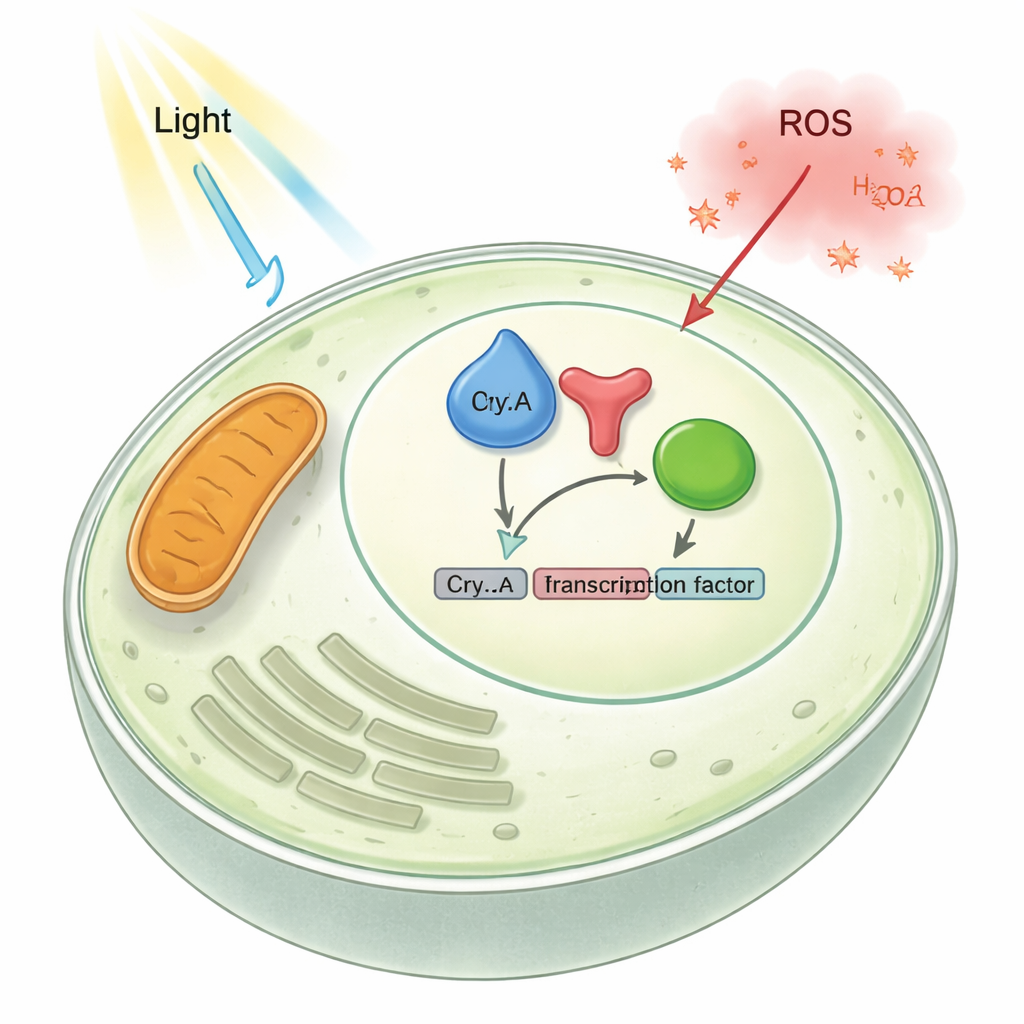
Crosstalk with the Main Light and Stress Pathways
The fungus already relies on another photoreceptor, a red-light sensor called phytochrome (FphA), and on a stress pathway ending in a transcription factor named AtfA. Using protein–protein interaction tests in living cells and purified proteins, the researchers showed that CryA physically binds both FphA and AtfA in the nucleus. When cryA was deleted, genes normally activated by red light through phytochrome became more strongly expressed; when cryA was overexpressed, they became harder to turn on. Chromatin experiments indicated that, in the absence of CryA, a key light-responsive gene carries more activating histone marks, suggesting that CryA normally dampens the chromatin-opening activity driven by phytochrome. In effect, CryA clamps down on both the light sensor and the downstream transcription factor, acting as a shared brake for light and stress signaling.
A Rapid Stress Sensor that Jumps to Mitochondria
Oxidative stress—an excess of reactive oxygen species like hydrogen peroxide—poses a constant threat to cells. The authors found that such stress, like light, boosts cryA expression. Strikingly, when hydrogen peroxide was added, CryA moved from the nucleus to the mitochondria in less than a minute. This jump required a short, flexible extension at the protein’s N-terminus and, in particular, a single cysteine amino acid within it. When that cysteine was changed to another residue, CryA could no longer leave the nucleus upon stress. Truncating the N-terminal tail forced CryA to live permanently at mitochondria. These engineered strains responded differently to oxidants: nuclear-only and mitochondrial-only versions of CryA altered the fungus’s resistance to hydrogen peroxide and menadione and reshaped expression of antioxidant genes. The findings suggest that CryA does more than sense stress—it may help coordinate communication between mitochondria and the nucleus so that antioxidant defenses are tuned to the type and level of damage. 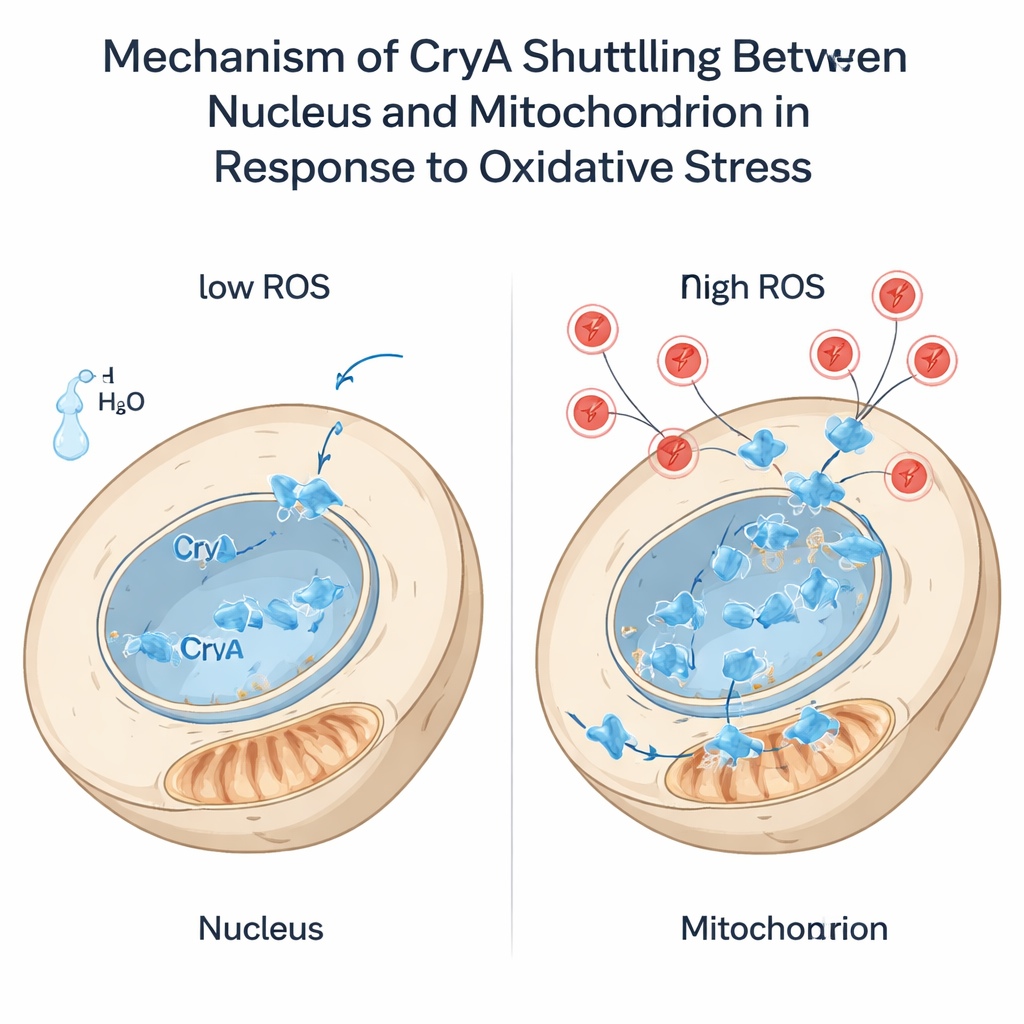
Why This Matters
To a non-specialist, CryA can be thought of as a cellular traffic cop that watches both the light on the outside and the oxidative stress on the inside, then decides when to slow down growth, development, and gene activity. By shuttling between the nucleus and mitochondria and tapping into major signaling routes, it keeps the fungus from overreacting to light or stress while still allowing a fast protective response. Similar proteins and mechanisms exist in many organisms, so this work offers a window into how living cells integrate environmental cues with internal damage signals to survive in a changing world.
Citation: Landmark, A., Rudolf, T., Hundshammer, K. et al. The photolyase/cryptochrome of Aspergillus nidulans senses oxidative stress and shuttles from nuclei to mitochondria. Nat Commun 17, 1483 (2026). https://doi.org/10.1038/s41467-026-69403-2
Keywords: light sensing, oxidative stress, cryptochrome, mitochondria, fungal development