Clear Sky Science · en
Lactate derived from macrophages drives skin dermal fibroblasts phenotypic remodeling via MCT1-primed histone H3 lysine 23 lactylation in hypertrophic scar
Why some scars grow thick and raised
Most cuts and burns eventually fade into thin, flat scars. But some wounds heal with thick, raised bands of tissue called hypertrophic scars that can itch, hurt, and limit movement. This study uncovers an unexpected driver of those stubborn scars: a common metabolic byproduct called lactate, produced by immune cells in stiff, healing skin, which reprograms nearby repair cells to behave in a more aggressive, scarring way.
From normal repair to problem scar
In healthy wound healing, skin cells work together to close the injury and rebuild the tissue. Fibroblasts, the main repair cells in the skin’s deeper layer, briefly switch into a stronger form that makes collagen, the protein scaffolding of skin. Once the wound is sealed, these cells quiet down again. In hypertrophic scars, however, fibroblasts stay stuck in this overactive state, laying down too much collagen in thick, disorganized bundles. The authors began by comparing normal skin to hypertrophic scar tissue and found a clear metabolic shift: the scar environment favored fast, sugar-burning chemistry that generates large amounts of lactate, along with high levels of a transport protein called MCT1 that ferries lactate into cells. 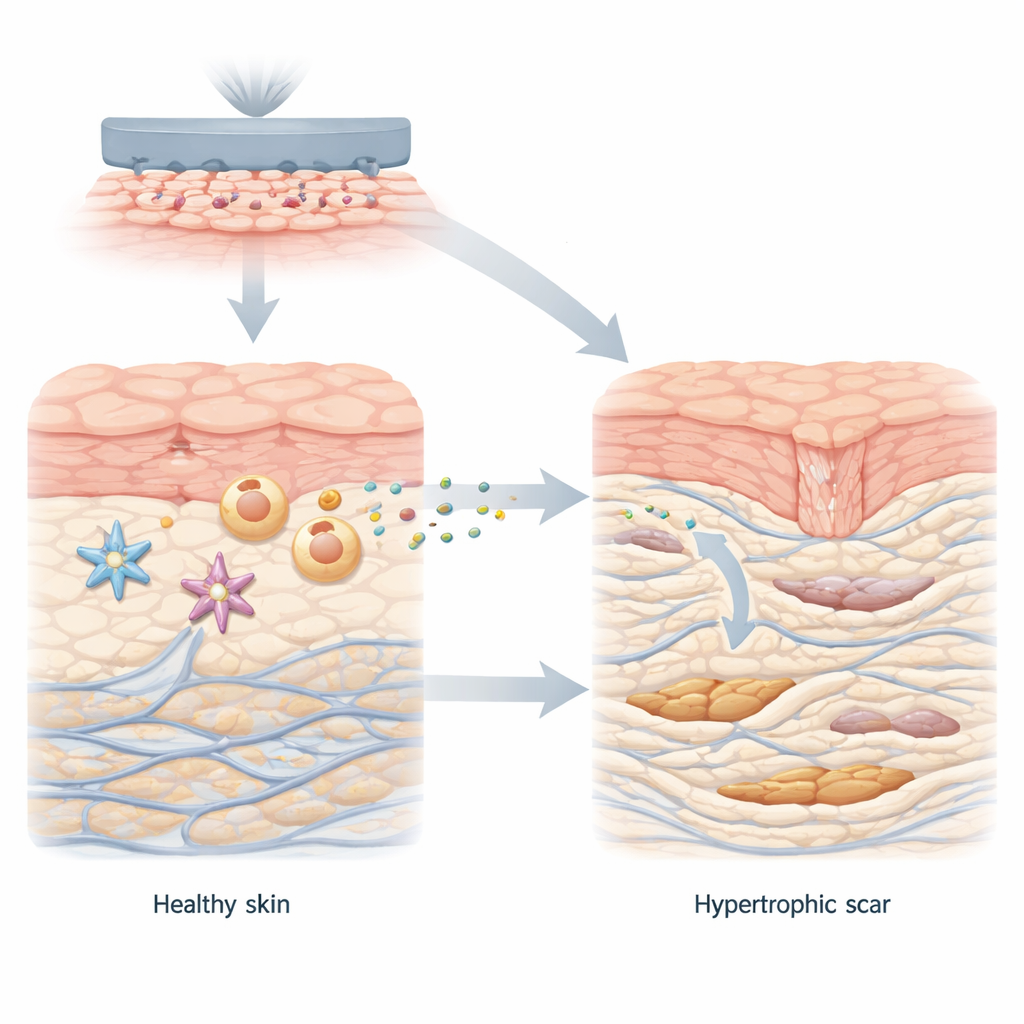
Immune cells as unexpected lactate factories
To discover where all this lactate comes from, the team examined different cell types from human skin and mouse models: macrophages (immune cells that clear debris and coordinate healing), blood vessel cells, and fibroblasts. They grew these cells on soft or stiff materials that mimic normal and scarred skin. On stiff surfaces, only macrophages flipped into a highly glycolytic, or sugar-hungry, mode and pumped out large amounts of lactate, both in lab dishes and in actual scar tissue. When macrophages were removed from healing mouse wounds, tissue lactate levels dropped and scar formation lessened. These results point to macrophages, especially those sensing a stiff environment, as the main lactate factories that shape the scar microenvironment.
Lactate as a signal that rewires fibroblasts
Next, the researchers asked how this excess lactate affects fibroblasts. When fibroblasts were bathed in lactate-rich fluid from stiff-cultured macrophages, they multiplied faster, moved more, and produced more collagen, all hallmarks of the aggressive myofibroblast state. Blocking the MCT1 transporter on fibroblasts, or reducing lactate production in macrophages, sharply reduced these changes. Inside fibroblasts, incoming lactate did more than fuel energy production: it triggered a specific chemical tag on histone proteins, which package DNA in the cell nucleus. This tag, called H3K23 lactylation, was much higher in scar-forming fibroblasts than in normal ones and acted like a switch that turned on two key genes, HEY2 and COL11A1, which together amplify scarring signals.
A self-reinforcing scarring loop
The proteins driven by this histone tag form a powerful feedback loop. HEY2 boosts the activity of a pathway involving YAP1 and SMAD2, which is known to push fibroblasts toward a more contractile, collagen-producing state. COL11A1, a collagen-related structural protein, physically interacts with the MCT1 transporter on fibroblasts, helping stabilize it and make lactate uptake more efficient. In other words, lactate entering the cell changes gene regulation in a way that further enhances lactate entry and collagen production, locking fibroblasts into a pro-scar identity. 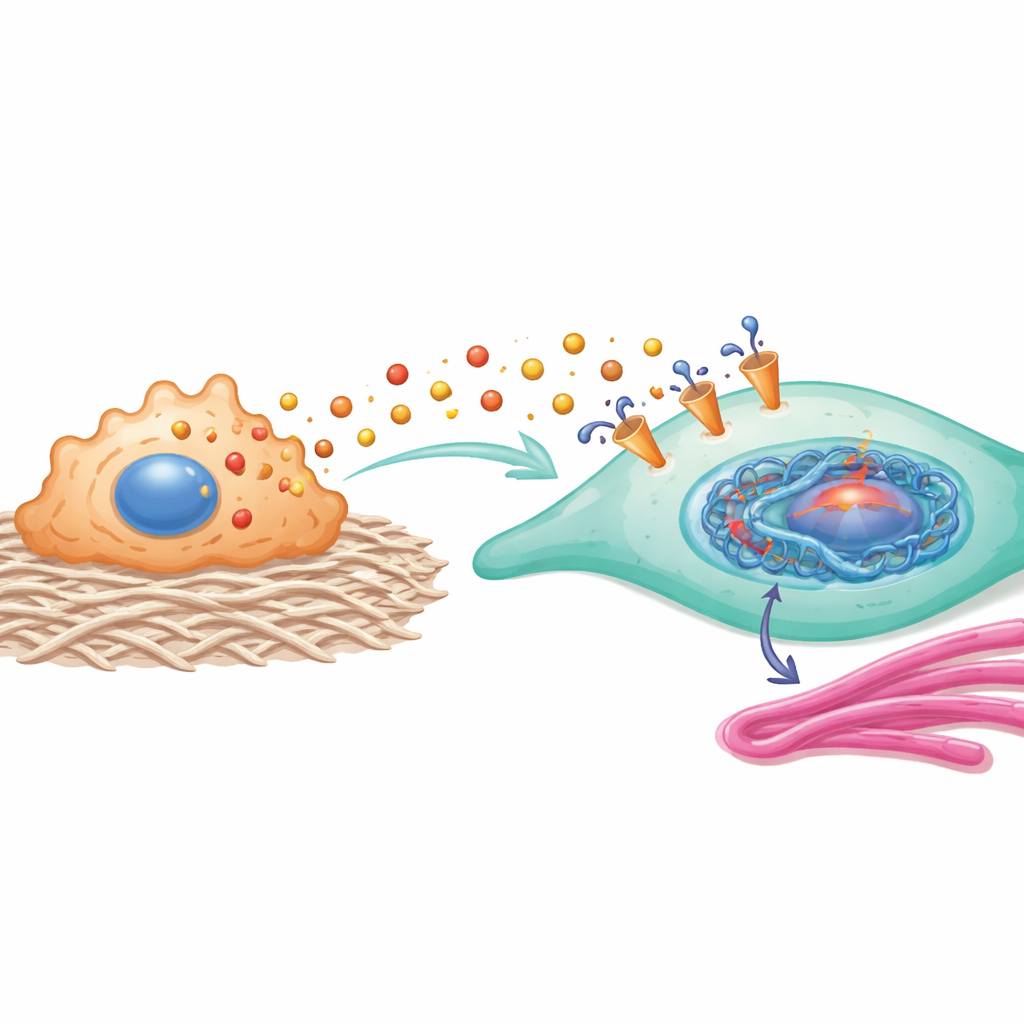
Turning down the loop to improve healing
To test whether interrupting this loop can improve healing, the team used mice in which MCT1 could be selectively removed from fibroblasts, as well as a drug that blocks MCT1. In both cases, wounds closed faster, the newly formed skin looked more organized, and collagen fibers were thinner and better aligned, with fewer features of hypertrophic scarring. The specific histone tag and its downstream genes also dropped. Depleting macrophages, the main lactate source, or lowering lactate chemically had similar anti-scar effects. These experiments show that the stiff-wound–macrophage–lactate–fibroblast chain is not just a correlation but a key driver of pathological scarring.
What this means for future scar treatments
This work reframes lactate from a simple metabolic waste product into a powerful messenger that links mechanics, metabolism, and gene control in wound healing. By showing how macrophage-derived lactate, transported through MCT1, changes histones and switches on a self-reinforcing scarring program in fibroblasts, the study highlights several promising therapeutic targets. Drugs that modulate lactate production, block MCT1, or interfere with the specific histone modification could one day help wounds heal more quickly and with flatter, less noticeable scars.
Citation: Yuan, Y., Xiao, Y., Zou, J. et al. Lactate derived from macrophages drives skin dermal fibroblasts phenotypic remodeling via MCT1-primed histone H3 lysine 23 lactylation in hypertrophic scar. Nat Commun 17, 2694 (2026). https://doi.org/10.1038/s41467-026-69388-y
Keywords: hypertrophic scar, lactate signaling, macrophage–fibroblast crosstalk, histone lactylation, wound healing therapy