Clear Sky Science · en
Myoferlin is a component of late-stage vRNP trafficking vesicles for enveloped RNA viruses
Why this matters for everyday health
Seasonal flu and other respiratory infections may feel routine, but together they kill millions of people worldwide each year. Many of these viruses, including influenza A and respiratory syncytial virus (RSV), already dodge vaccines and antiviral drugs by rapidly mutating. This study looks instead at something viruses cannot easily change: the human cell machinery they hijack to escape from infected cells. By uncovering a shared weak spot in that machinery, the work points toward future medicines that could blunt a whole range of respiratory viruses at once.
A cellular delivery route that viruses exploit
Our airway cells constantly reuse bits of their outer membrane through a process called recycling. Small membrane bubbles, or vesicles, shuttle cargo from inside the cell back to the surface, guided by a family of proteins known collectively as Rab11. Influenza A virus, RSV and a related virus called Sendai virus all exploit this route late in infection. They load their genetic cargo, packaged as ribonucleoprotein complexes, onto Rab11-containing vesicles that carry them to the top (airway-facing) side of the cell, where new virus particles bud off and spread. Until now, Rab11 was the only host factor clearly shared by these trafficking vesicles, leaving a big gap in our understanding of how they form and function.
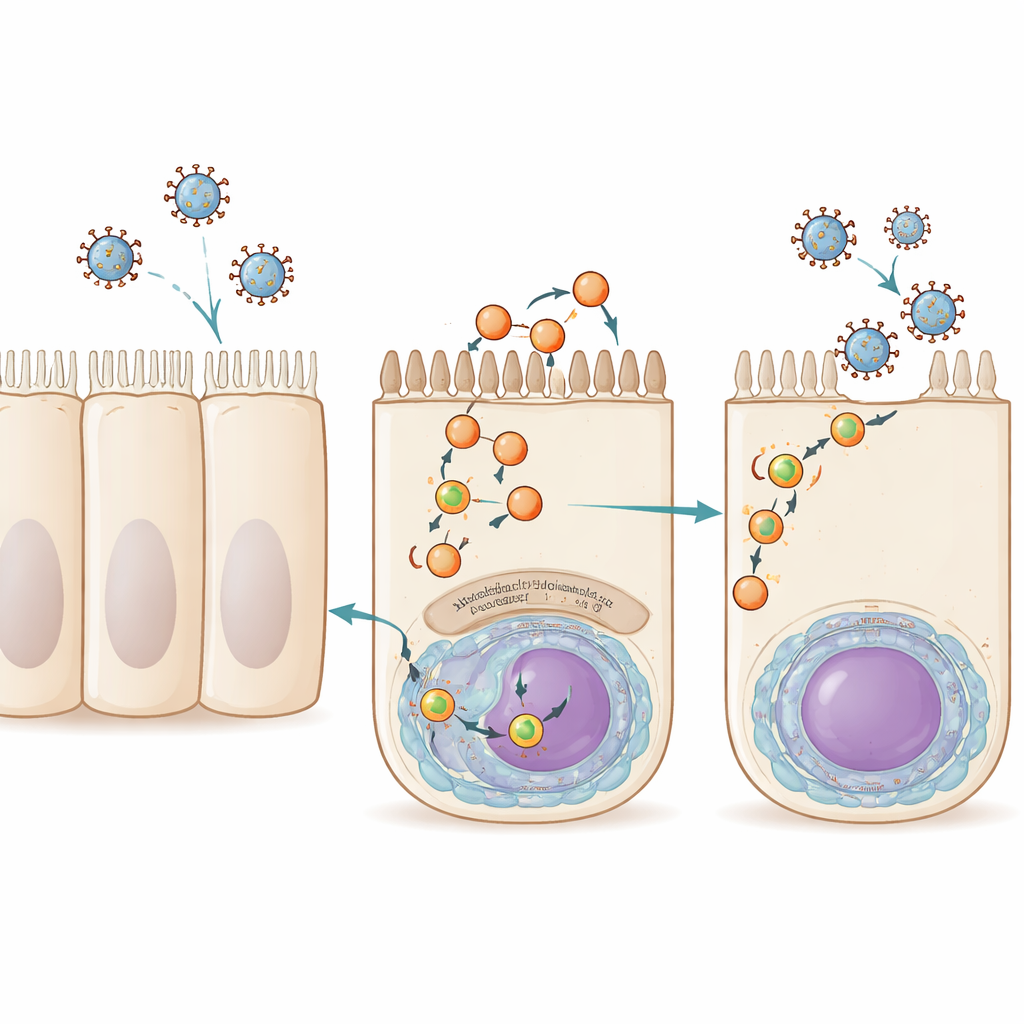
Finding a new player: myoferlin
The researchers began by asking which human proteins physically associate with influenza’s genome-packaging machinery at different times during infection. They engineered an influenza A virus whose polymerase carried a molecular tag, then used mass spectrometry to catalogue human proteins bound to this complex early and late in infection. Among hundreds of candidates, one stood out at late time points: myoferlin, a large membrane-associated protein previously linked to muscle development, endocytosis and membrane repair. When the team reduced myoferlin levels in lung-derived cells using small interfering RNAs or blocked it with a small-molecule inhibitor, the cells still made viral RNA and viral proteins normally, but released far fewer infectious influenza particles. This indicated that myoferlin is not needed for copying the viral genome, but is crucial for the later stages when new viruses are assembled and exit the cell.
Myoferlin travels with viral cargo
High-resolution imaging revealed where myoferlin sits during infection. In uninfected cells, myoferlin largely overlaps with Rab11-positive recycling vesicles, both near the nucleus and out toward the cell edge, and loss of either protein disrupts the normal distribution of the other. During influenza infection, the Rab11 network is dramatically remodeled into enlarged, irregular vesicles that carry viral ribonucleoproteins. Myoferlin is retained in these remodeled vesicles and shows strong co-localization with the viral genome and Rab11. When the team used a drug that forces viral genome complexes and their carrier vesicles to clump together, myoferlin was pulled into the same aggregates, confirming that it is a built-in component of these late-stage trafficking structures and not just nearby by chance.
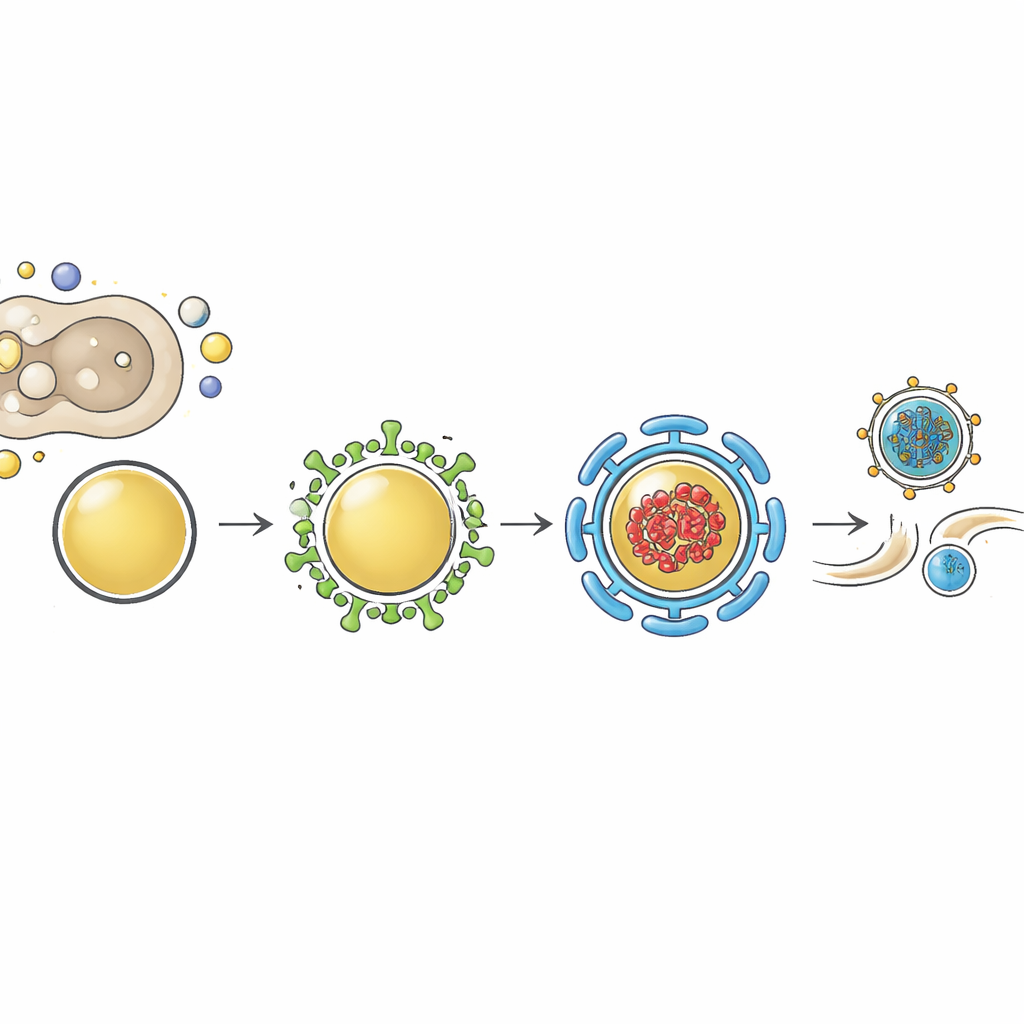
A shared hub for multiple respiratory viruses
The study then asked whether this dependence on myoferlin is unique to influenza or more broadly shared. Knocking down myoferlin in lung cells infected with RSV or Sendai virus again caused a strong drop in infectious virus released, without lowering viral RNA levels. Single-molecule imaging showed the genomes of both viruses clustering together with myoferlin and Rab11 in cytoplasmic spots that likely represent transport hubs rather than replication factories. This pattern supports a unifying picture: different enveloped respiratory RNA viruses, despite diverse lifestyles, converge on the same Rab11–myoferlin vesicle system when it is time to move their completed genomes toward the cell surface for packaging and exit.
How myoferlin shapes virus-carrying vesicles
Myoferlin is built from multiple "C2" domains that can bind membranes and partner proteins. Earlier work showed that one of these domains engages a family of membrane-remodeling proteins called EHDs. In this study, fluorescently tagged EHD1 and EHD2 were found within the same remodeled vesicles that carry influenza genomes and Rab11, especially when these vesicles were experimentally clustered. Reducing EHD2 levels, like reducing myoferlin, lowered influenza yields without affecting genome replication. Moreover, removing myoferlin caused EHD2 protein levels to drop, suggesting that myoferlin helps stabilise or correctly position EHD2 on membranes. The authors propose that myoferlin sits on Rab11-positive vesicles and recruits EHD proteins to sculpt their membranes, completing the formation of specialised "irregularly coated vesicles" that are optimised for transporting viral cargo to budding sites.
What this means for future antiviral strategies
Taken together, the findings position myoferlin as a central organiser of a host recycling route that many enveloped respiratory viruses rely on at a critical late step. Because targeting viral proteins alone often leads to rapid resistance, host factors that are reused by multiple viruses are attractive drug targets. Interfering with myoferlin’s function, or its partnership with Rab11 and EHD proteins, could in principle slow or block the spread of diverse pathogens, from influenza and RSV to other Rab11-dependent viruses such as some coronaviruses. Much work remains to translate this concept into safe therapies, but the study provides a detailed map of a shared viral escape route—and spotlights myoferlin as a promising choke point along that path.
Citation: Bonazza, S., Turkington, H.L., Sukumar, S. et al. Myoferlin is a component of late-stage vRNP trafficking vesicles for enveloped RNA viruses. Nat Commun 17, 2507 (2026). https://doi.org/10.1038/s41467-026-69386-0
Keywords: influenza, respiratory viruses, vesicular trafficking, host factors, myoferlin